Article Content
INTRODUCTION (PDF)
The electrochemical carbon dioxide reduction reaction (CO2RR) has garnered significant interest for recycling carbon dioxide as an energy source under ambient temperatures and pressures.1–7 Catalysts play a crucial role in enhancing the conversion efficiency of the CO2RR to hydrocarbon and alcohol products, such as ethylene, methane, and ethanol.8–12 In the conversion of CO2 into value-added chemicals, the progression of multiple reactions in multicomponent catalysts mutually supports each reaction step. In particular, the local reaction sites around the hetero-interface in such catalysts have been considered influential in catalytic activity; consequently, reaction mechanisms have recently been studied using various composite catalysts.13–20 However, the changes in local reaction sites and catalytic structures induced by electrochemical reactions remain insufficiently understood. This is because in conventional composite catalyst fabrication processes, the local reaction sites and micro-/nano-structures cannot be accurately and arbitrarily designed. Such uncontrollable characteristics prevent direct observation of hetero-interfaces and tiny areas such as nanoparticle surfaces.21–26
Microfabrication technology has great potential to address these issues because it enables the artificial production of miniature structures on the micrometer scale and smaller. Recent literature has increasingly highlighted phenomena at the hetero-interface using microfabrication technology.27–33 For example, Lum and Ager demonstrated that micropatterned Cu catalysis allows control of the ratio of oxygenates to ethylene by tuning the distance between Cu- and CO-producing catalysts in aqueous CO2 reduction.27 Similarly, Larrazábal et al. showed that the high CO evolution activity of Cu–In catalysts is associated with indium-poor bimetallic phases formed under reaction conditions near Cu–In interfaces.28 Dong et al. investigated the effect of grain boundary oxidation in Cu–Ag thin films on the selectivity of CO and CH4 production. They found that Cu thin films with smaller grain sizes are susceptible to spontaneous oxidation, which degrades the Faradaic efficiency of production.29 However, further investigations are required to fully understand the mechanisms of electrochemical reduction reactions and the structural changes at hetero-interfaces.
In the present study, we investigated the changes in the hetero-interfacial reaction sites and surface structures of Cu–Ag catalysts before and after electrochemical CO2RR. We prepared micropatterned Cu–Ag catalysts consisting of periodic Ag square arrays on single-crystal Cu substrates using microfabrication technologies such as photolithography. These artificially patterned catalysts enabled the quantitative evaluation of changes in catalyst surfaces and reaction sites during CO2RR. Local analysis using energy-dispersive x-ray spectroscopy (EDS) directly revealed increased oxygen adsorption on the Cu surface near the Cu–Ag hetero-interface after CO2RR. In addition, unlike nanoparticle surfaces, the scalability and flatness of the micropatterned catalysts allowed quantitative evaluation of the mean and variance of surface roughness on Cu and Ag using atomic force microscopy (AFM) before and after the reaction. Our approach provides a tool for capturing detailed catalyst properties, thereby facilitating a better understanding of the CO2RR mechanism at hetero-interfaces.
II. MATERIALS AND METHODS
A. Preparation of the micro-pattered Cu–Ag catalyst
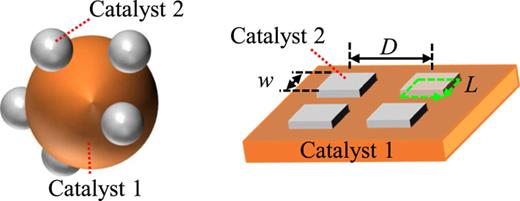
We studied the variation in the hetero-interfacial and structural characteristics of a Cu–Ag-based catalyst before and after electrochemical reactions using a micropatterned catalyst instead of a conventional composite catalyst. Figure 1 shows schematics of a conventional composite catalyst (left panel) and a micropatterned catalyst (right panel) consisting of two types of catalytic materials (catalysts 1 and 2). In composite catalysts, randomly scattered nanoparticles prevent us from analyzing their structural characteristics, such as surface roughness, and observing local reaction sites, such as hetero-interfaces. On the other hand, microfabrication technology enables us to artificially design the position and scale of the reaction sites by controlling the structural parameters, for example, the side length w of the square patterns, pattern spacing D, and hetero-interface length L, as shown in Fig. 1. In this study, we fabricated Ag periodic square patterns on a Cu(100) substrate using a microfabrication technology that is familiar with semiconductor device fabrication.
Schematics of a conventional composite catalyst and micro-/nano-patterned catalyst consisting of two catalyst materials. The distance between periodic patterns D, side length of the square-shaped pattern w, and hetero-interface length L are examples of artificially controllable parameters by micro-/nano-fabrication technology.
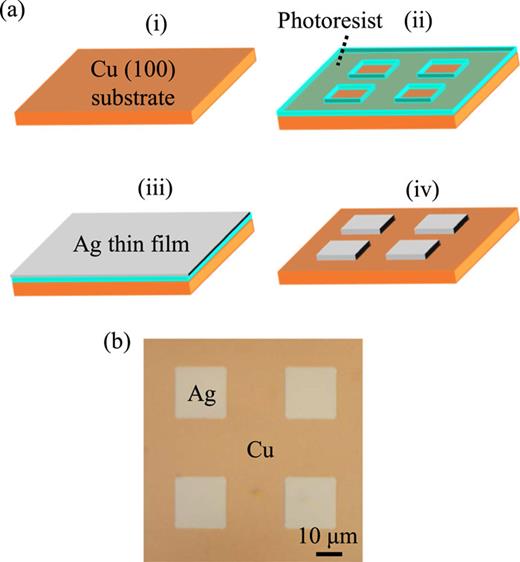
The steps for the fabrication of the micropatterned Cu–Ag catalyst are summarized in Fig. 2(a). We prepared a Cu substrate with a plane orientation of (100) (Shanghai Institute of Optics and Fine Mechanics, Chinese Academy of Sciences), which is 15 × 25 × 0.5t mm. The Cu substrate was cleaned using acetone, isopropyl alcohol (IPA), and deionized (DI) water with ultrasonic cleaning for 5 min each [Fig. 2(a-i)]. Using photolithography, we drew a pattern of a periodic Ag square array on a Cu substrate [Fig. 2(a-ii)]. To enhance the adhesion between the substrate and the photoresist, hexamethyldisilazane (Tokyo Ohka Kogyo Co. Ltd., OAP) was spin-coated onto the substrate at 5000 rpm for 50 s. Subsequently, a photoresist (Tokyo Ohka Kogyo Co. Ltd., THMR-iP3100 MM) was spin-coated under the same conditions as the OAP and pre-baked at 90 °C for 90 s. The substrate was inserted into a mask aligner (EV Group, EVG620) and patterned by illuminating ultraviolet light in contact with the photomask. After lithography, we dipped the substrate in the developer (Tokyo Ohka Kogyo Co. Ltd., NMD-W 2.38%) for 3 min and rinsed it with DI water for 1 min. We obtained a substrate with a resist pattern by post-baking at 110 °C for 90 s. Using electron beam evaporation equipment (Canon Anelva Corporation, EVD-500B), a Ag thin film with a thickness of 50 nm was fabricated on a substrate with a resist pattern at a pressure of 1.8 × 10−4 Pa and a deposition rate of 0.2 nm/s [Fig. 2(a-iii)]. A lift-off process was then used to remove the Ag from the Cu substrate, with the photoresist in the remover for 24 h. After ultrasonic cleaning in acetone, IPA, and DI water for 5 min each, we obtained a complete Cu–Ag catalyst with a pattern consisting of periodic Ag squares on the Cu substrate [Fig. 2(a-iv)]. Figure 2(b) shows a microscopic image of the fabricated catalyst with (w, D) = (20 μm, 40 μm). A 20 × 10 mm2 patterned area on the Cu–Ag catalyst was used for the CO2RR.
(a) Schematics of fabrication process for Cu–Ag micropatterned catalyst. The fabrication step is in the order of (i) to (iv). (b) Microscopic image of the completed micropatterned Ag squares with (D, w) = (40 μm, 20 μm) on a Cu(100) substrate. Here, D and w represent the periodic distance and side length of the square-shaped Ag pattern, respectively, as shown in Fig. 1.
B. Characterization
We used a scanning electron microscope (SEM) (Hitachi High-Tech Corporation, S-4800) to observe the surface of the patterned catalyst before and after CO2RR. We observed the catalyst, which was allowed to electrochemically react for 1 h under each of five voltage conditions, i.e., a total of 5 h, as the sample of post-CO2RR. We also conducted EDS (Oxford Instruments, UltimMax65) to investigate the elemental distributions on the surface and hetero-interface of the patterned catalyst. Ag and Cu surface conditions were analyzed by AFM (Hitachi High-Tech Corporation, E-sweep). Crystallinity of the Cu substrate and the deposited Ag film was analyzed using x-ray diffraction (XRD) (Rigaku Holdings Corporation, RINT-TTR).
C. Electrochemical measurement and product analysis
Electrochemical measurements were conducted in an original H-type cell separated using a bipolar membrane (Fumatech GmbH, Fumasep FBM) with a three-electrode system with a potentiostat/galvanostat (Meiden Hokuto Corp., HZ-Pro). The bipolar membrane prevents the alcohol products such as C2H5OH and C3H7OH from crossing over to the anode, as shown in the literature.34,35 An Ag/AgCl reference electrode (EC Frontier Co. Ltd., RE-11A) was inserted between the fabricated Cu–Ag catalyst and the bipolar membrane, and the counter electrode was a Pt mesh. The electrolyte used for these measurements was 0.1M KHCO3. CO2 gas was directly introduced into the electrolyte solution from near the patterned catalyst during the CO2RR. As a preparation of CO2RR, CO2 bubbling to the electrolyte was carried out for 30 min before electrochemical reaction, similar to the literature.27–29 The applied reversible hydrogen electrode [VRHE (V)] was calculated from the potential in the cell (VAg/AgCl) (V) by the Nernst equation considering the potential drop due to the resistance in our cell, i.e., VRHE = VAg/AgCl + 0.2 + 0.059 × pH + IAveR. Here, IAve is the average measured current and R is the resistance obtained from the impedance measurement. Note that the macroscopic position of Cu and Ag and flow direction of raw material such as CO2 become significant parameters under high current density,36 while the effect of them is negligible small under low current density.31–33
The gas products produced by CO2RR were analyzed using a gas chromatograph (SRI Instruments, 8610C). The liquid products were analyzed using a gas chromatograph equipped with a headspace sampler (Agilent Technologies Inc., 7890A). All measurements were performed under ambient conditions and at room temperature.
III. RESULTS AND DISCUSSION
A. Direct observation of hetero-interface
The micropatterned catalyst has great potential as a platform to analyze the variance in local reaction sites via electrochemical reactions. We evaluated the Cu–Ag hetero-interface before and after CO2RR using a micropatterned catalyst.
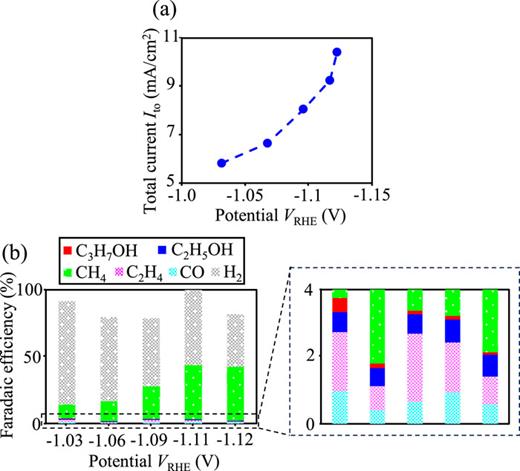
We confirmed that the micropattern functioned as a catalyst for the CO2RR. Figures 3(a) and 3(b) show the measured total current Ito and Faradaic efficiency, respectively, as functions of the applied potential VRHE using the patterned catalyst. Electrochemical measurements with the micropatterned catalyst produced hydrocarbons and alcohols, in addition to hydrogen, while there is unresolved future work that the reason why methane is a dominant product, such as the case for Cu(111).24,37 Ag (Cu) decomposes CO2 (CO) to produce CO (hydrocarbons and alcohols).7,38–46 Thus, the obtained products indicate that a series of reduction reactions from CO2 to hydrocarbons and alcohols occurred on the micropatterned Cu–Ag catalyst, similar to the reactions on conventional Cu–Ag composite catalysts. These results confirm that the micropattern functioned as a CO2-reduction catalyst. Considering from the Faradaic efficiency, there may be undetected products due to the insufficient resolution of the instrument or incompatible products for the instrument. However, such minor and small amounts of products are outside the scope of this study. Note that CO2 and OH− mass transfer is one of the factors affecting the reaction products under relatively high current density, e.g., .36 On the other hand, for low current density, the effect of mass transport is conventionally small enough to be ignored.
Using SEM-EDS observations, we evaluated the elemental distributions on the catalyst for pre- and post-CO2RR. First, the elemental distribution of the patterned catalyst was macroscopically observed. Figures 4(a)–4(e) [Figs. 4(f)–4(j)] show SEM images and elemental maps of Cu, Ag, C, and O, respectively, before (after) CO2RR. The elemental distribution of Cu [Figs. 4(b) and 4(g)] and Ag [Figs. 4(c) and 4(h)] indicates that the patterned structures remained without drastic changes, such as losses and deformations through CO2RR. Carbon [Figs. 4(d) and 4(i)] and oxygen [Figs. 4(e) and 4(j)] were preferentially adsorbed on Cu and Ag, respectively, regardless of CO2RR.
(a)–(j) Top-views of SEM images [(a) and (b)] and elemental mappings for Cu [(b) and (g)], Ag [(c) and (h)], C [(d) and (i)], and O [(e) and (j)] before and after CO2RR. (k)–(n) Top-views of SEM images around hetero-interface between Ag and Cu [(k) and (m)], and one-dimensional elemental distributions along the magenta lines [(l) and (n)] before and after CO2RR.
Second, for the microscopic analysis of the local reaction sites, we conducted a one-dimensional (1D) EDS analysis around the Cu–Ag hetero-interface. Figure 4(k) shows the SEM image of the hetero-interface before CO2RR and a scanning line colored by magenta. Transitions in the elemental distributions of Cu (blue) and Ag (red) [Fig. 4(l)] clearly capture the existence of a hetero-interface. Oxygen (green) and carbon (cyan) are predominantly detected on Cu and Ag, respectively, similar to the macroscopic maps shown in Figs. 4(d) and 4(e). Figures 4(m) and 4(n) show the SEM images around the hetero-interface and the elemental distributions along the magenta colored scanning line after CO2RR, respectively. The results for Cu, Ag, and C are similar to those in Fig. 4(l). On the other hand, interestingly, we see that the oxygen profile for post-reaction has a peak on the local Cu region neighboring the hetero-interface. Thus, the micropatterned catalyst enabled us to directly observe oxygen adsorption near the hetero-interface. Indeed, an increase in the number of remaining Cu–O bonds after the reaction has been reported by introducing Cu–Ag hetero-interfaces in Cu-based catalysts, resulting in the promotion of hydrocarbon and alcohol production.39,47 Our corresponding results strongly indicate that the number of remaining Cu–O bonds dominantly increased near the Cu–Ag hetero-interface. Therefore, the micropatterned catalyst is a useful platform for directly observing changes in local reaction sites before and after electrochemical reactions.
B. Quantification of catalytic surface structure
The scalability of the patterned catalysts allows us to easily design flat and large catalytic material surfaces that are normally difficult to prepare for nanoparticle-based composite catalysts. Taking advantage of these artificial structures, we evaluated the surface roughness of Ag and Cu before and after the reaction using AFM. Such structural information, alongside material composition, is crucial for understanding the mechanism of catalytic reactions.
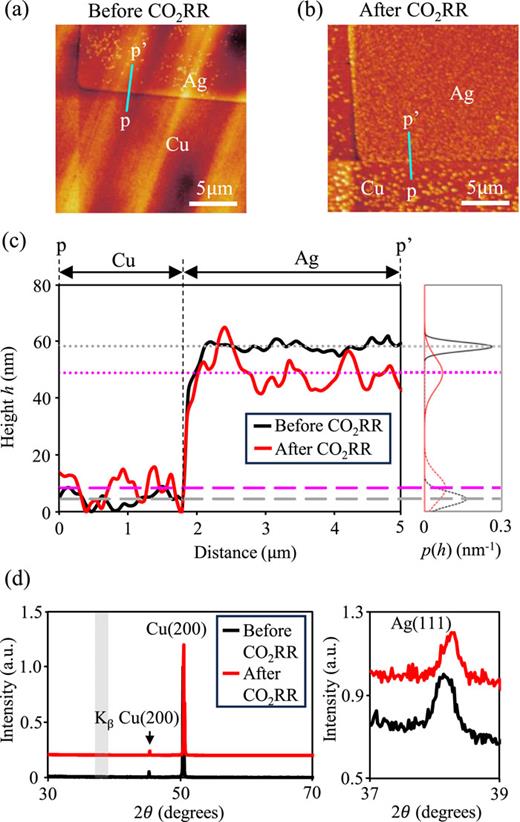
Figures 5(a) and 5(b) present AFM images of an Ag pattern and a Cu surface, taken before and after CO2RR, respectively. Our analysis revealed that the Ag square pattern remained intact, showing no significant signs of reaction or degradation. To further investigate the surface condition, we plotted the 1D-height profiles along the cyan lines from p to p′ in Figs. 5(a) and 5(b). The solid black (red) lines in Fig. 5(c) show the height (h) profile for pre- (post-)reaction. This AFM analysis indicated that CO2RR induced an increase in the surface roughness of Cu and Ag and a decrease in the height h of the Ag. Because of the flatness of the patterned catalyst, we quantitatively evaluated its surface structure. In particular, we calculated the averages and variances of the heights of the Cu and Ag surfaces before and after the CO2RR from the height profiles. The right panel in Fig. 5(c) shows the visualized surface information expressed as a probability density function p(h), i.e., . Here, μ and σ2 denote the average and variance of the height profiles, respectively. The dashed and dotted gray (magenta) lines in Fig. 5(c) show the average heights of Cu(=4.44 nm) [(=8.27 nm)] and Ag(=58.27 nm) [(=48.89 nm)] before (after) CO2RR. Compared with the profile before CO2RR, the Ag height from the Cu surface decreased by 24.5% after CO2RR. The variance σ2 calculated from the profile indicates the degree of surface roughness. The height variance on Ag (Cu) surface increases from σ2 = 2.28 nm2 (5.97 nm2) to σ2 = 32.1 nm2 (23.4 nm2) following CO2RR. This result contributes to providing a preferable particle size by considering the increase in surface roughness by CO2RR.
(a) and (b) AFM images before (a) and after (b) CO2RR. (c) One dimensional height profiles along the cyan line from p to p′ in (a) and (b), and probability density distributions p(h) of height h. The black (magenta)-broken and -dashed lines represent the average height of Cu and Ag surfaces before (after) CO2RR. (d) X-ray diffraction patterns of the micropatterned Cu–Ag catalyst before (black) and after (red) CO2RR. The left panel shows the spectra in a wide range of 2θ from 30° to 70°. The right panel indicates the enlarged spectra in the range of the gray shaded region in the left panel.
In addition to the surface roughness, the crystallinity of Cu and Ag was investigated. Figure 5(d) shows the XRD spectra of the patterned catalyst before (black line) and after (red line) CO2RR. In the left panel, the spectra having peaks corresponding to Cu(200) ensure that the Cu substrate maintains single crystallinity for both pre- and post-CO2RR. The right panel indicates the enlarged spectra around the peak of Ag(111), which is in the gray shaded region in the left panel. Compared with the two spectra, we see that the crystallinity does not deteriorate because of CO2RR regardless of the increase in the surface roughness. Note that a slight peak shift was guessed to occur due to flatness distortion induced by fixing the substrate during the electrochemical measurements because all the observed peaks [Cu(200), Kβ Cu(200), and Ag(111)] similarly shifted 0.1° to the lower angle side.
Therefore, the platform enables us to quantitatively analyze changes in the catalytic structure, such as surface roughness, for which it has been difficult to obtain sufficiently flat and large surface areas for direct observation using conventional composite catalysts.
C. Selectivity of reaction products by controlling the structural parameters in micropattern
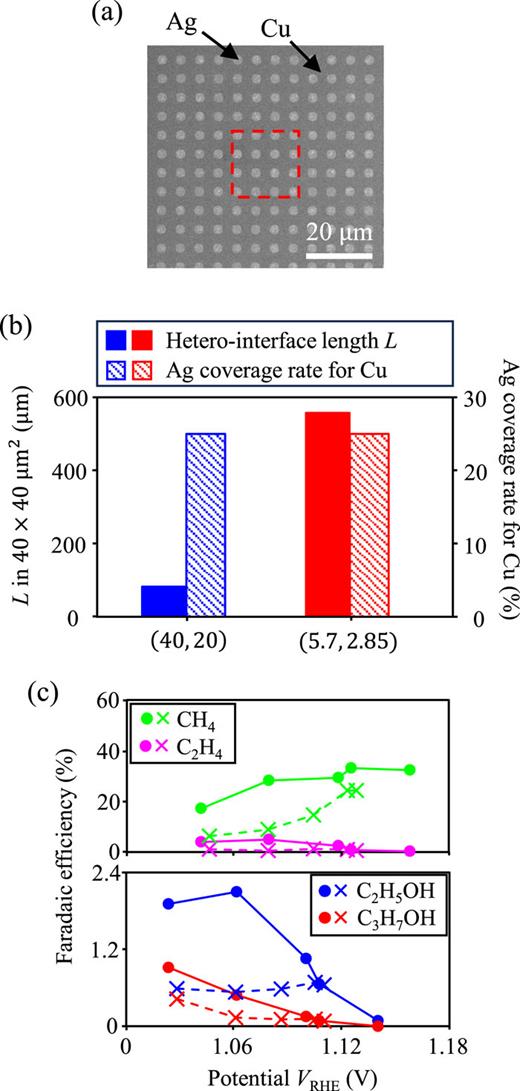
The results in the previous sections clearly show that the hetero-interface between Cu and Ag contributes to CO2RR. To further show the evidence of the catalytic function on the hetero-interface, we investigate the dependence of the length of hetero-interface on the ratio of reaction products. Microengineering technology has the potential to control reaction activity by accurately and arbitrarily designing structural parameters such as the hetero-interface. In this study, we prepared two micropatterned electrodes with (D, w) = (40 μm, 20 μm) and (5.7 μm, 2.875 μm) to compare their electrochemical CO2RR performance. Figure 6(a) shows the SEM image of the micropatterned electrode with (D, w) = (5.7 μm, 2.875 μm). The red squares in Fig. 6(a) show the size of one Ag square for the catalyst, where (D, w) = (40 μm, 20 μm). Figure 6(b) shows the Cu–Ag hetero-interface length L and the Ag coverage rate for Cu for each catalyst in an area of 40 × 40 μm2. The flexible designability of microengineering technology allowed us to tune L while maintaining a constant coverage rate for the two catalysts. Thus, we evaluated the effect of the Cu–Ag hetero-interface as a local reaction site for electrochemical reduction reaction products. Figure 6(c) shows the Faradaic efficiency of all multielectron reduction products, i.e., CH4 (green), C2H4 (magenta), C2H5OH (blue), and C3H7OH (red), as a function of VRHE. The circles and crosses represent the results for micropatterned Cu–Ag catalysts with (D, w) = (40 μm, 20 μm) and (5.7 μm, 2.875 μm), respectively. The micropatterned catalyst with a longer hetero-interface showed a higher Faradaic efficiency for all products than that with a shorter hetero-interface. Through the demonstration in terms of the ratio of reaction products, the reliability of the analysis of the hetero-interface is enhanced. In addition, referring to the literature,16,18,39,47 the obtained result is reasonable because the Cu–Ag hetero-interface satisfies the conditions as a favorable local reaction site for multistep reduction reaction from CO2 to multielectron reduction products through CO. First, CO is formed on the Ag surface from CO2. Then, around the hetero-interface, CO can quickly reach the Cu surface for further reduction, resulting in multielectron reduction products. Therefore, accurately controllable microengineering technology has great potential for designing local reaction sites for the selectivity of each desired product.
(a) Top-view SEM image of the periodic Ag pattern with (D, w) = (5.7 μm, 2.85 μm) on the Cu(100) substrate. The dashed red square represents the size of one Ag pattern of (D, w) = (40 μm, 20 μm), as shown in Figs. 2 and 4. (b) Total hetero-boundary length L in the area on 40 × 40 μm2 square (solid bars) and Ag coverage rate for Cu (shaded bars). The blue and red colored bars represent the patterned catalysts with (D, w) = (40 μm, 20 μm) and (5.7 μm, 2.85 μm), respectively. (c) Faradaic efficiencies of the pattered catalysts with (D, w) = (40 μm, 20 μm) (cross symbols) and (5.7 μm, 2.85 μm) (circle symbols). The green, magenta, blue, and red plots indicate Faradic efficiencies for CH4, C2H4, C2H5OH, and C3H7OH, respectively.
IV. CONCLUSION
In this work, we analyzed changes in the hetero-interface and surface state before and after electrochemical CO2RR using a micropatterned Cu–Ag catalyst. The micropattern successfully functioned as a CO2-reduction catalyst, producing hydrocarbons and alcohols. The finely controllable scalability and position of the pattern enabled direct and straightforward observation of changes in elemental distributions around the Cu–Ag hetero-interfaces through CO2RR. After CO2RR, oxygen adsorption on the Cu surface increased locally near the Cu–Ag hetero-interface. This finding strongly supports the theory that the hetero-interface facilitates oxygen supply and enhances local electrochemical reaction sites for producing hydrocarbons and alcohols from CO2. In addition, we observed the surface roughness of the Cu–Ag catalyst, which is conventionally difficult to quantitatively analyze using composite catalysts due to the size and non-flatness of nanoparticles. The flatness and scalability of the micropatterned catalyst enabled the evaluation of average heights and variances of the Cu and Ag surfaces. CO2RR induced a 24.5% decrease in the height of the Ag patterns and an increase in the variance of the Ag (Cu) surface roughness by 14.1 (3.9) times. Quantifying such structural information is critical for understanding catalyst degradation mechanisms. Finally, as a demonstration of the future potential of our platform, we investigated the selectivity of reaction products by controlling the structural parameters. The Faradaic efficiencies of hydrocarbons and alcohols increased with the length of the Cu–Ag hetero-interface, without altering the Ag coverage of Cu. This flexible designability through microengineering is valuable for identifying and observing local reaction sites, enhancing specific electrochemical reactions, and optimizing the spatial design and composition of catalytic materials. We hope that this work will contribute to the development of an effective analytical tool and framework for understanding catalytic performance, paving the way for advancements in catalyst design and application.

![(a)–(j) Top-views of SEM images [(a) and (b)] and elemental mappings for Cu [(b) and (g)], Ag [(c) and (h)], C [(d) and (i)], and O [(e) and (j)] before and after CO2RR. (k)–(n) Top-views of SEM images around hetero-interface between Ag and Cu [(k) and (m)], and one-dimensional elemental distributions along the magenta lines [(l) and (n)] before and after CO2RR.](https://aipp.silverchair-cdn.com/aipp/content_public/journal/adv/15/7/10.1063_5.0271594/1/m_075111_1_5.0271594.figures.online.f4.jpeg?Expires=1754966183&Signature=DxEHps~0CgO4yiHwxT6fttcNIn6hQ7n0CmustujwetWW5hmTWpgeYPj8BYq-MQR0mH~i8XpfHfcRGsq68bwjwmB76vsmCX8JyjICcIXs4Ks5UfHlXpICHjAfiegfIE0z3EkjFc~uyYAV7Eku89-bKxkAdcIN3DJlx2QwyUMWdv7ZzvR-xdAAlcBQDuj0arURtg0ssL4VNy3r8crnLhpJqQ1ItDZnWTYLN~HyfBH4YY9PKULBzlcxwMtY8LM2q6ZLjoOrnY9Kqee~Bxz70coR5LKH3RmyqPv3VKNcQTJqj4fEVMdFddPyqjJ77WT3YV2IZWzXJXPtp3KR0Lbb2RLB7Q__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)