Article Content
Introduction
Reconfigurable all-liquid systems have been used to control the structural and chemical characteristics of soft matter1. Most efforts in the field have harnessed solvent-water interactions to achieve polarity-based spatial compartmentalization2,3,4. More recent efforts have focused on aqueous two-phase systems (ATPSs), produced by the phase separation of mixed aqueous solutions containing incompatible polymers5,6,7,8,9. ATPSs offer a natural platform for all-aqueous biomimicry10,11, e.g., based on aqueous dextran (DEX) and polyethylene glycol (PEG) solutions12. However, due to the ultralow interfacial tension between the two aqueous phases, typical ATPSs lack structural stability13,14, resulting in irreversible collapse of the liquid phases. ATPSs stabilization has been achieved using polyelectrolytes or colloidal particles15,16,17; however, component formulation and stability are not yet optimized.
Structured liquids, generated by complexing two oppositely charged polyelectrolytes (PE) at the interface of two contiguous liquid phases18, afford a route to address this challenge19,20,21. This approach capitalizes on the inherent mobility and transport characteristics of each phase, ease of access to the interface to enable reaction or interaction between two immiscible components, and the spatial and dimensional confinement of the interface itself 22. The complexation of the components at the interface can lock in the shapes of the liquid phases while not impeding their response to environmental stimuli, such as light23, electric or magnetic fields20,24. Previous studies have demonstrated the ability of polyelectrolyte (PE) pairs, a polycation and a polyanion, to form complexes at the water/water interface, preventing coalescence and locking-in non-equilibrium shapes to the two aqueous phases25. By adjusting the flux of oppositely charged substances to the interface so as to optimize interfacial complexation, and accordingly, stabilization of the phases can be achieved. Charged nanoparticles (NPs) have also been used to produce PE/NP complexes that encapsulate the aqueous phases26,27. Up to now, interfacial complexes have enabled the generation of non-equilibrium all-aqueous structures, such as tubular shapes5,28, as well as complex multi-compartmentalized microcapsule structures29. Endowing such constructs with additional functions offer significant potential for cargo encapsulation and release30.
In principle, PE/PE systems form deformable membranes, while PE/NP complexes are thicker and more rigid31. A recent study on the complexation of positively charged diallyl dimethylammonium chloride and negatively charged rodlike cellulose nanocrystals (CNC) has shown that the thickness of the layer at the interface increased with time32, enhancing the mechanical properties of the interfacial complexes. These composite complexes can be used as biomimetic constructs33, such as artificial tissues from functional ATPSs. However, for the structured ATPSs from either PE/PE or PE/NP complexes, integrating multifunctional attributes into a single all-aqueous system has been little studied. We introduce all-aqueous constructs based on NP/NP complexes with oppositely charged biological nanoparticles; specifically, rodlike anionic CNCs and cationic chitin nanofibers (ChNFs)34,35. The nanocrystalline structure of CNC is designed to provide structural integrity for the interfacial assembly, whereas the disordered domains in ChNF facilitate the flexibility properties (i.e., enabling bending and twisting behaviors)36,37,38. The ChNF/CNC complexes possess both excellent deformability and rigidity, comparable to those of PE/PE and PE/NP systems, respectively. Moreover, their rod-on-rod porous assembly structure results in enhanced permeability, distinguishing them from conventional PE-based complexes.
In this work, ChNF/CNC complexes are formed at water-water interface to produce membranes with a tunable mechanical strength and permeability, and allow for the formation of all-aqueous microcapsules with the ability to compartmentalize. The ChNF/CNC complex layer can be thickened by liquid flow across the interface, for example, under an imbalanced osmotic stress. In concert with other features of the ChNF/CNC complexes, e.g., deformability and permeability, the resultant microcapsules show autonomous, switchable sub-surface mobility, tuned by the deformation of the meniscus at the surface. Such biological nanoparticle-structured ATPSs open possibilities for autonomous transport and exchange in artificial cells.
Results
Nanoparticle complexation at water-water interfaces
As a proof-of-concept, biomimetic underwater microcapsules were fabricated in ATPSs32 inspired by permeable human cell membranes and water-walking striders (Fig. 1). Water-water interfaces can be generated with aqueous solutions containing DEX and PEG and stabilized in situ by the self-assembled complexes formed by oppositely charged biological nanoparticles, ChNF and CNC (Supplementary Fig. 1), suspended in the polymer solutions (Fig. 1c). The interfacial complexation of ChNF and CNC is driven by electrostatic interactions at the interface between the phases. The electrostatic interactions between the cationic amine groups of ChNF and the anionic half sulfate ester groups of CNCs are enhanced by release of counterions and water (entropic gain, )39. Considering the ultralow interfacial tension at the water-water interface (limited enthalpic contributions) and the structural features of the oppositely charged nanoparticles, the spontaneous interfacial electrostatic interactions are significantly enhanced by the entropic gain. Fourier transform infrared spectroscopy (FTIR) results confirmed that no covalent bonds were formed during the interfacial complexation process, indicating that the assembly was driven purely by physical interactions (Supplementary Fig. 2). Meanwhile, a slight shift in the 1550 cm−1 (N-H bending) peak suggests electrostatic interactions between the amino groups (-NH2) of ChNF and the sulfate groups (-SO3H) of CNC, similar to those observed in ChNF/SA complexes40.
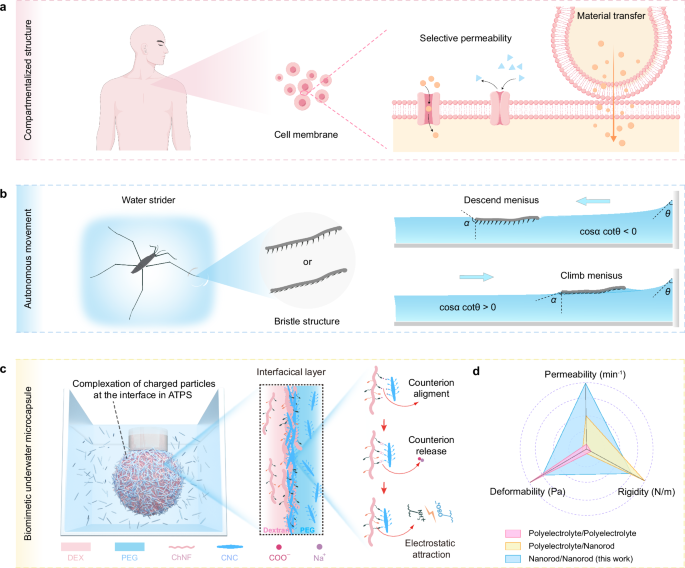
Sources of inspiration: (a) Schematic illustration of the structure of human cell membranes and their functions of selective permeability and intercellular material transport (by figdraw.com), and (b) Schematic illustration of water strider harness capillary forces by changing their bristle structure posture on the feet to climb or descend the meniscus between the surface of water and a solid object. The arrows represent the direction of motion of water strider. c Schematic illustration of a pendant droplet where the internal phase is encapsulated to form a biomimetic underwater microcapsule by self-assembled interfacial complexes. Interfacial complexes formed from entropy-driven electrostatic complexation of chitin nanofibers (ChNF) and rodlike cellulose nanocrystals (CNC) at the interface of aqueous dextran (DEX) and polyethylene glycol (PEG) solutions. d A radar chart illustrates tunable performance of nanoparticle/nanoparticle complexes (blue) in this work, compared with the counterparts assembled from polyelectrolyte/polyelectrolyte (pink) and polyelectrolyte/nanoparticle (yellow) systems.
Compared to interfacial complexes formed through polyelectrolyte/polyelectrolyte (PE/PE) and polyelectrolyte/nanoparticle (PE/NP) interactions (Fig. 1d), the ChNF/CNC complexes distinctively integrate the key features of all systems: rigidity, deformability, and permeability. This combination enables applications beyond the capabilities of traditional PE-based systems. While PE/PE systems have shown enhanced performance in autonomous underwater transport, controlled delivery, and microreactor applications, our nanoparticle/nanoparticle (NP/NP) system provides a broad and versatile platform for such functionalities. Nevertheless, we note that there is no intent to compare ChNF/CNC-based microcapsules to the same level of performance as biological cell membranes or the bristle structures on water strider legs. Instead, the latter serves as a natural inspiration to the development of all-aqueous, biomass-based interfacial assemblies that share similar fundamental properties using a NP/NP approach.
Pendant drop tensiometry was used to investigate the interfacial complexation, where a 10 wt% DEX solution (pH = 3, = 1.0471) was slowly introduced into 10 wt% PEG solution (pH = 3, = 1.0208) (Supplementary Table 1). Complexation and pendant droplet stabilization were found for the nanoparticle complexes (ChNF in DEX and CNC in PEG) (Fig. 2a). No complexation was evidenced in the absence of the nanoparticles (Supplementary Movie 1), where the DEX droplet fell due to the ultralow interfacial tension. At higher pH values, the amine groups on the surface of ChNFs are deprotonated (Fig. 2b)41, giving rise to weaker complexes with CNCs and failing to stabilize the interface (illustration of Fig. 2b). This sensitivity to pH changes is expected to be affected by the non-uniform distribution of amine groups on the surface of ChNFs, due to the random deacetylation process used in their preparation42.
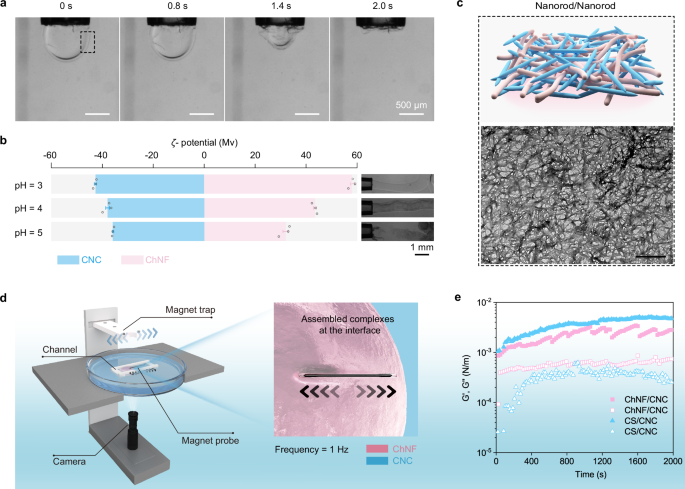
a Evolution of pendant dextran-in-polyethylene glycol (DEX-in-PEG) droplet stabilized by ChNF/CNC complexes (black dashed box) following a time sequence when drawing the internal phase at 2 μL/s. The ChNF and CNC concentrations are 1 wt% in the respective DEX (10 wt%) and PEG (10 wt%) aqueous solutions. b ζ-potential of ChNF (pink) and CNC (blue) at pH value of 3-5. The illustration demonstrated the feasibility of complexation of ChNF and CNC at the water-water interface at pH value of 3-5 of the DEX phases. The 10 wt% DEX phases (1 wt% ChNF) was injected into 10 wt% PEG phases (1 wt% CNC) at an extrusion speed of 2 μL/s. c Transmission electron microscope (TEM) image of the ChNF/CNC complexes obtained from the interface at the black dotted box mark in a). The scale bar is 600 nm. d Experimental set-up used to access the interfacial shear rheology of the complexes. e Storage (G’, closed symbol) and loss (G”, open symbol) moduli of the complexes assembled from ChNF/CNC (pink square) and chitosan (CS)/CNC (blue triangle). Data in b are presented as mean ± standard deviation from n = 3 independent samples.
We further investigated the microstructure of ChNF/CNC complexes by transmission electron microscopy (TEM), scanning electron microscopy (SEM), and atomic force microscopy (AFM). These data showed an interconnected, multi-layered network with voids, consisting of both rigid CNC clusters and flexible ChNF networks, contributing to a balance of mechanical strength and adaptability (Fig. 2c and Supplementary Fig. 3). The permeability of ChNF/CNC interfacial complexes was evaluated using fluorescein isothiocyanate, FITC-labeled PEG (Mw = 10,000 Da). Microcapsules formed by the ChNF/CNC complexes allowed rapid diffusion of PEG within ~5 min, attributed to the porous rod-on-rod structure of the interface (Supplementary Fig. 4). In comparison, chitosan (CS)/sodium alginate (SA) microcapsules exhibited much slower PEG diffusion, requiring around 60 min for similar exchange (Supplementary Fig. 5). The slower transport in CS/SA capsules reflects the presence of a denser polymeric network, which restricts molecular permeation5,32,43. However, the ChNF/CNC complex layer was effective in blocking particles (50 nm polystyrene spheres, Supplementary Fig. 6), implying size exclusion effects due to the characteristic cells in the network formed at the interface.
To investigate the deformability of the ChNF/CNC interfacial complex, we measured the internal pressure of microcapsules during deformation using a microsyringe pump system equipped with a vacuum pressure gauge (Supplementary Fig. 7a–c). As a reference, the deformability of CS/CNC interfacial complexes under the same matrix conditions was also measured (Supplementary Fig. 7d). PE/PE complexes are typically considered highly deformable due to their soft polymeric nature5,32. The results indicate that the CS/CNC interfacial complexes required higher negative pressure to achieve the same deformation as the ChNF/CNC complexes, supporting the hypothesis that ChNF/CNC complexes exhibit greater deformability compared to CS/CNC complexes (Supplementary Fig. 8). More importantly, we found that the mechanical properties of ChNF/CNC interfacial complexes can be tuned by adjusting the surface charge density of ChNF. Specifically, the low-charge-density chitin nanofiber (ChNF-L)/CNC complexes deformed more easily under lower negative pressure, which can be attributed to the lower relative content of rigid CNC crystals within the interfacial complexes. However, the high-charge-density chitin nanofiber (ChNF-H)/CNC interfacial complexes exhibited enhanced rigidity (Supplementary Fig. 8). In subsequent experiments investigating the thickening behavior of the interfacial complexes, under identical osmotic pressure gradients (), ChNF-L/CNC complexes exhibited wrinkling behavior similar to PE/PE interfacial complexes, indicating higher deformability (Supplementary Fig. 17). These findings demonstrate that we can precisely regulate interfacial complexes mechanics by modifying the surface charge density of ChNF, enabling their adaptation to various applications requiring different mechanical performance.
In-situ interfacial shear rheology was performed by using a magnetic probe to quantitatively assess the mechanical properties of the interfacial layer (Fig. 2d and Supplementary Movie 2). Supplementary Fig. 9, as a control, shows a good correlation between the probe and the trap position during the test, validating the reliability of the method. The results shown in Fig. 2e and Supplementary Fig. 10 correspond to the time and frequency evolution of the storage (G’) and loss (G”) moduli for the interfacial complexes (ChNF/CNC and CS/CNC) at the water-water interface. The CS/SA interfacial complexes were not studied due to the rapid and dense association between CS and SA. Both the ChNF/CNC and CS/CNC complexes had a high G’, e.g., solid-like behavior. Notably, although ChNF/CNC complexes had a slightly lower G’, their loss factor value was significantly higher than that of the CS/CNC complexes. This indicates that the ChNF/CNC system formed a tough, yet flexible interfacial layer. To further validate these findings, we conducted additional interfacial shear rheology measurements using a double-walled Couette flow-cell setup, obtaining consistent results (Supplementary Fig. 11). The agreement between these independent methods shows that both interfacial complexes possess mechanical rigidity, with CS/CNC forming a stiffer interfacial structure, while ChNF/CNC complexes exhibit a balance of toughness and flexibility. The observed moduli differences can be explained by the interaction and arrangement of rod-like, rigid CNCs in the complexes44,45. In the CS/CNC complexes, the CS chains facilitated strong binding and orderly arrangement with CNCs imparting the rigidity features. The high aspect ratio (Supplementary Fig. 1) and random distribution of surface charges of ChNF resulted in a less ordered complex, contributing to the formation of a flexible interfacial film. SEM and AFM further support the coexistence of randomly arranged nanofibers and ordered nanocrystalline clusters within the ChNF/CNC complex layer (Supplementary Fig. 3). Overall, complexation of ChNFs and CNCs at the interface stabilized the shapes of the ATPSs and the permeable complexes with rigidity and deformability, while maintaining the structure when the external environment changed, e.g., osmotic stress.
Osmotic stress balance between the two aqueous phases
Osmotic stress can modulate the direction of water flow across the water-water interface in ATPSs (Fig. 3a), a simple route to adjust the properties of all-aqueous constructs32. As such, water transport occurs between the interior of the pendant droplet and the external phase, under the influence of osmotic pressure gradients. A microcapsule and a pendant drop encapsulated with the ChNF/CNC complexes are subjected to three different regimes of osmotic stress, , between the internal (DEX) and external (PEG) phases (Fig. 3 and Supplementary Table 1), namely , , and . The osmotic pressure of each phase was adjusted by the concentrations of DEX and PEG, while keeping the nanoparticle loading the same (1.0 wt% in each phase). Initially, ChNF-in-DEX and CNC-in-PEG met at the interface and formed an interfacial layer by electrostatic interactions. No apparent wrinkling was observed on the droplet surface (Fig. 3b). With time, the interfacial complexes equilibrated, affecting both convective flow and permeability (Fig. 3b).
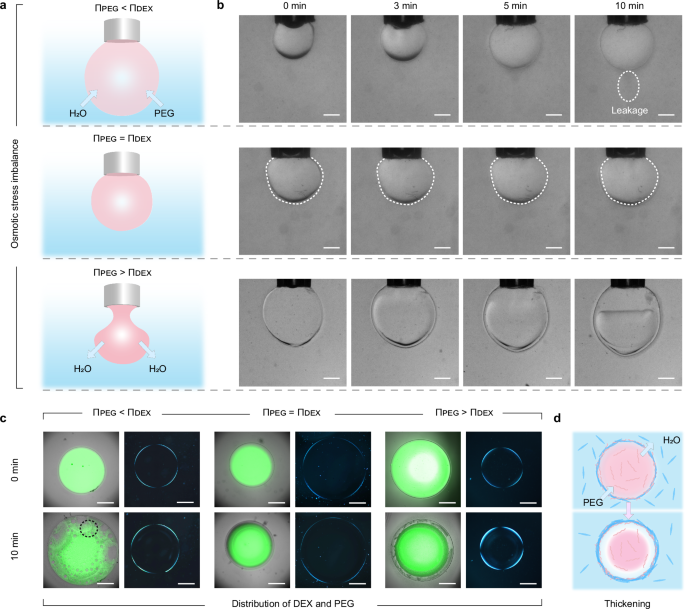
a Schematic illustration of the shape variation and (b) time evolution of the morphology of the pendant dextran-in-polyethylene glycol (DEX-in-PEG) droplet encapsulated with chitin nanofiber/cellulose nanocrystal (ChNF/CNC) complexes under osmotic stress () and corresponding exchange across the interface. The osmotic stress difference is adjusted by the concentration of DEX and PEG: , , and (with the ChNF and CNC loading in each phase fixed at 1 wt%). The dashed lines in the middle row of (b) highlights the contour of the droplet. c Fluorescent (left) and polarized optical (right) microscope images of the microcapsule under given osmotic stresses. The dashed black circle when indicates the formation of small PEG-in-DEX-in-PEG droplets. In the fluorescent tests, 0.001 wt% fluorescein isothiocyanate (FITC) -labeled DEX (green) was pre-mixed with the DEX phase. d Thickening of the ChNF/CNC complex layer where water and PEG flow outward and inward, respectively, facilitating assembly of CNC with ChNF originally dispersed in the DEX phase. All arrows represent the direction of materials diffusion. The scale bar is 400 μm.
At , a pendant DEX droplet gradually expanded due to continuous influx of water from the PEG solution (Fig. 3a, b). Fluorescence and optical microscopy images (Fig. 3c and Supplementary Fig. 12 in the case of a microcapsule) clearly showed the formation of small PEG-in-DEX emulsion droplets inside the larger droplet stabilized by the ChNF/CNC complex layer. The mechanical resistance of the latter accommodated slow deformation due to the water flow. However, the encapsulated components in the droplet gradually leaked out, e.g., following failure of the ChNF/CNC complex layer (see dashed circle in Fig. 3b, upper panel). Similar to the PE/PE system, a CS/SA complex layer expanded (flexibility), with no droplet breakage (Supplementary Fig. 13), while the CS/CNC complex layer burst soon after fluid influx (Supplementary Fig. 14). Consequently, the ChNF/CNC system bears characteristics of the other two complexes.
The droplet was stable when (see contour in Fig. 3b, middle panel). Although a slight degree of phase separation occurred, no leakage or thickening of the complex layer was observed under this condition (Fig. 3c and Supplementary Fig. 15), suggesting long-term stability. For , the droplet retained its original shape with a layered hierarchical structure forming on the outer edge of the interface (Fig. 3a, b), which was further confirmed by fluorescence and optical microscopy observations of the microcapsules (Fig. 3c and Supplementary Fig. 16). This was caused by a thickening of the ChNF/CNC complex layer, similar to destabilized emulsions or plasmolysis in plant cells. Water transport occurred from the DEX phase to the PEG phase due to the osmotic stress imbalance (Fig. 3d), providing a driving force for ChNF diffusion that led to its detachment. Meanwhile, as the volume of DEX phase was gradually reduced, PEG penetrated the droplet, forcing CNC to diffuse to the interface. As a result, more ChNF and CNC engaged in electrostatic interactions, increasing the thickness of the complex layer, while maintaining a high permeability. Under this osmotic pressure gradient ( ), the interfacial membrane thickness increased over time, initially undergoing a rapid thickening before gradually stabilizing (Supplementary Fig. 17). Notably, the final interfacial complexes thickness increased with higher ChNF charge density, which can be attributed to stronger electrostatic attraction between ChNF-H and CNC, leading to enhanced CNC accumulation at the interface. This also demonstrates that the presence of free nanoparticles, not involved in the interfacial assembly, is inevitable, which does not affect the preparation, performance, and application of microcapsules. From these results, it is evident that the ChNF/CNC complexes can be adjusted to tailor mass exchange (Fig. 1d).
The assembly of CNC and ChNF at the microcapsule surface contributed to the rigidity of the complex layer to balance osmotic stress. For all osmotic gradients, extinction was observed parallel to and perpendicular to the polarization direction using polarized optical microscopy for the ChNF/CNC complexes, which indicates that the ChNFs and CNCs were oriented parallel to the interface (Fig. 3c). However, owing to the morphology of ChNF and CNC, a different ordering behavior may occur than that found for the polyelectrolyte/CNC interfacial complexes (Supplementary Fig. 14c). The assembly of ChNF and CNC at the interface was further explored by changing their distribution in ATPSs (Supplementary Fig. 18). A droplet was stabilized when suspending ChNF (1 wt%) in PEG and CNC (1 wt%) in DEX (Supplementary Fig. 18a), where osmotic stress transferred water from the internal DEX phase to the external PEG phase, gradually reducing the droplet volume. However, thickening of the complexes did not occur, and the CNC/ChNF complex layer wrinkled (Supplementary Fig. 18b). Extinction along the crossed polarization was not observed (Supplementary Fig. 18c), suggesting a strong partitioning of CNC to the DEX phase46. The density of the DEX phase increased with a reduction in the water content and, due to the permeability and poor mechanical strength of the ChNF/CNC complex layer, eventually led to a phase separation at the bottom of the droplet (Supplementary Fig. 18a).
Selective transfer across ChNF/CNC complex layer
Ionic fluorescent probes were added to different phases across the complex layer47 (Fig. 4a). Negatively charged fluorescein (sodium salt, FSS) in the PEG phase immediately diffused into a microcapsule (DEX phase) and interacted with the ChNF/CNC complexes by electrostatic interactions between FSS and ChNF (Supplementary Fig. 19). Similarly, positively charged Nile blue A (NBA) in the DEX phase was excluded from the microcapsule and the thickened ChNF/CNC complex layer (Supplementary Fig. 20), due to the negatively charged CNC in the external PEG phase and the complexes. The permeability of the ChNF/CNC complex layer and the asymmetric diffusion of oppositely charged molecules suggest possible applications for separation, purification, and compartmentalized serial reaction systems. Separation of the mixed ionic dyes took place when dissolving FSS and NBA in either PEG or DEX solutions, soon after forming the microcapsule; no effect was noted for the initial location of the mixture in the PEG (Fig. 4b) and DEX (Supplementary Fig. 21) phases. At equilibrium, the NBA was preferentially located in the PEG phase, while FSS was present in the DEX phase. This was demonstrated by the time-dependent FSS and NBA fluorescence intensity in the given phases and at a given position (dashed lines in Fig. 4b) (Fig. 4c). FSS diffused across the boundaries when a microcapsule containing FSS was placed next to two unloaded microcapsules (Fig. 4d). This transfer was preferential to the neighboring microcapsules but not to the surrounding PEG solution, which was confirmed by time-dependent fluorescence intensity measurements of FSS at given positions (dashed line in the Fig. 4d) (Fig. 4e). Furthermore, no bridging between the microcapsules was observed upon contact (Supplementary Movie 3), implying that the FSS transferred across the non-coalescent complex layers. Moreover, thickening of the complexes continuously occurred, in contrast to the observation for PE/NP complexes32.
