Article Content
1 Introduction
Tetracycline is a commonly utilised antibiotic in veterinary and human medicine. With an annual production range reaching thousands of tons and usage of about 100–200 tons, it presently ranks as one of the most produced and used antibiotics globally [1]. This widespread use, coupled with wastewater release from pharmaceutical industries, has led to the indiscriminate release of TC into the environment, with high concentrations detected in surface water, sediments, the marine environment, and soil [2, 3]. Its ability to accumulate in the environment and access the food chain impacts the microbial environment, while also causing antibiotic resistance. Hence, its negative effect on the environment cannot be ignored [4,5,6,7]. Moreover, TC poses a significant threat to the quality of water sources due to its capacity to disturb the balance of microbes in the human intestine. These potential detrimental effects of TC contamination in the environment, therefore, raise serious concerns and present a possible global public health challenge [8,9,10,11].
TC, being a refractory organic pollutant, is often inefficiently removed by conventional wastewater treatment systems [12,13,14]. Alongside some transformation products, it has been detected at ng/L concentration in some municipal wastewater treatment plants [15]. Therefore, the development of more efficient wastewater treatment technologies for dealing with pollutants such as TC is of global importance. Photocatalysis has attracted significant attention in recent times due to its environmental friendliness, cost-effectiveness, and high pollutant degradation [16]. Consequently, the development of suitable photocatalysts with enhanced activity is highly desirable to achieve a notably improved photocatalytic process for wastewater treatment [17, 18]. A major advantage of photocatalysis is the ease with which it could be coupled with other processes [19]. Photocatalytic activation of persulfate is one of such processes that has gained significant attention because of its wider adaptability to different wastewater conditions [19,20,21].
Various semiconductor materials have been investigated as catalysts for the photocatalytic activation of persulfate, including TiO2, Bi2MoO6, and metal-organic frameworks [22,23,24]. Most of them suffered from limited visible-light absorption, rapid electron-hole recombination, metal leaching, difficult recovery, and narrow pH applicability, whereas zinc ferrite addresses these issues with its visible-light responsiveness, reduced recombination, low leaching, magnetic recyclability, and broad pH stability [25]. Recently, metal ferrites with spinel structures, such as cobalt ferrite (CoFe₂O₄), nickel ferrite (NiFe₂O₄), and manganese ferrite (MnFe₂O₄), have attracted significant interest as visible-light-active photocatalysts for environmental remediation. These ferrites have narrow band gaps, which allow for efficient use of visible light, and their intrinsic magnetic properties that permit facile separation and recycling from treated water [26]. For instance, CoFe₂O₄ has been extensively studied for the degradation of persistent organic pollutants and dyes, with its efficacy further augmented by doping and the formation of heterojunctions with materials such as g-C₃N₄. This leads to enhanced charge separation and photocatalytic efficiency. Similarly, NiFe₂O₄-based composites have significant efficacy in the photodegradation of antibiotics and industrial dyes, particularly when integrated with other semiconductors such as ZnO and Co₃O₄, which enhance light absorption and mitigate electron-hole recombination [27]. MnFe₂O₄, when combined with carbon-based materials, has exceptional efficacy in pollutant degradation and hydrogen evolution under visible light, attributed to its extensive surface area and advantageous band structure [28]. These advancements collectively underscore the adaptability and potential of spinel ferrites as effective photocatalysts for the remediation of various pollutants, such as pharmaceuticals, pesticides, and dyes, while also providing benefits in catalyst recovery and operating stability [29]. Zinc ferrite (ZnFe2O4) is distinguished by its narrow band gap that can be excited by UV and visible light without any modification [30, 31]. However, due to the high recombination rate of the photogenerated charge carriers, its efficiency is significantly limited [32]. By combining the photocatalytic process with persulfate activation, the activity of ZnFe2O4 can be enhanced, with PS acting as an electron acceptor, with a consequent enhancement in the charge carrier separation. Additionally, the electron can result in the decomposition of PS, leading to the generation of SO4•– and O2•– radicals [33], which also contribute to the photodegradation process.
This study reports the degradation of TC through the photocatalytic activation of persulfate using ZnFe2O4 as the catalyst. The hybrid process demonstrated a high synergy factor, enhancing the activity of ZnFe2O4. Radical scavenging study further confirmed the generation of O2•– radicals alongside others, which had minimal influence on the degradation process. The versatility of the process for other pharmaceuticals as well as dyes was explored, and it exhibited significant activity for NSAIDs (diclofenac) and methylene blue. The study highlights the versatility of the photocatalytic activation of persulfate and the potential benefits in wastewater treatment plants. The novelty of this study lies in the synthesis of a highly efficient, visible-light-active, and magnetically recoverable ZnFe₂O₄/persulfate photocatalytic system, with proven versatility for multiple pollutants, offering a sustainable solution aligned with SDG goals.
2 Methodology
Zinc(II) chloride dihydrate (ZnCl2•2H2O), iron(III) chloride hexahydrate (FeCl3•6H2O), potassium persulphate, and polyvinyl alcohol were procured from Merck and used without further purification.
2.1 Characterization of zinc ferrite (ZnFe2O4) nanoparticles
The nanoparticles’ structural characteristics and crystalline phases were examined using a Bruker D8 Advance X-ray diffractometer (Karlsruhe, Germany), which features a proportional counter and operates with Cu-Kα radiation (λ = 1.5405 Å) filtered via nickel. The scanning was performed at a rate of 0.0018° per minute. The surface morphology was examined with a Quanta FEG 250 environmental scanning electron microscopy (SEM) at an accelerating voltage of 30 kV. The internal morphology and particle size were analysed using a TECNAI G2 transmission electron microscope (TEM, ACI). Optical absorption characteristics were assessed using an Agilent Cary 30 UV–visible spectrophotometer.
2.2 Synthesis of zinc ferrite (ZnFe2O4) nanoparticles
A wet chemical method was utilised to synthesise zinc ferrite (ZnFe2O4) nanoparticles. ZnCl2•2H2O and FeCl3•6H2O were each dissolved in 100 mL of distilled water in a molar ratio of 2:1. Subsequently, 4.00 g of PVA was added as a surfactant and agitated at 80 °C for 2 h. The resultant solution was evaporated at 100 °C overnight, and the acquired brown residue was calcinated at 800 °C for 4 h. The prepared photocatalyst was analysed by X-ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM), and UV-vis spectroscopy.
2.3 Evaluation of the photocatalytic activity on Tetracycline
The catalyst (50 mg) was incorporated in an aqueous solution of tetracycline (20 mg/L) and agitated for 30 min in the dark to achieve adsorption/desorption equilibrium. The mixture was thereafter lit with a visible bulb. During the reaction, small aliquots of the solution were collected at 20-minute intervals, filtered to isolate the catalyst from the drug solution, and absorbance was measured at 360 nm using a UV-visible spectrophotometer. This reaction was repeated using different concentrations (1mM, 2mM, 3mM, and 5mM) of persulphate salt.
3 Results and discussion
3.1 Structural and morphological studies
The crystalline structure and phase composition of the material were examined using XRD. As shown in Fig. 1(a), the XRD pattern was consistent with the spinel phase of ZnFe2O4 (JCPDS No.22-1012) [34]. The pattern showed that a highly crystalline ZnFe2O4 was obtained, and the absence of impurity peaks such as ZnO and Fe2O3 confirmed the formation of pure-phase ZnFe2O4 nanoparticles. ZnFe2O4 is known to exhibit mixed spinel structures with partial cation inversion, which enhances its visible-light photocatalytic activity and magnetic recoverability. This is consistent with recent studies on Co-substituted and rare-earth-doped spinel ferrites, which demonstrated that controlled cation substitution enhances lattice parameters, magnetic properties, and charge carrier dynamics, thereby advancing the multifunctionality and efficiency of ferrite-based photocatalysts for environmental and technological applications [35, 36]. Additionally, scanning electron microscopy (SEM) was used to study the microstructure and morphology of the material, as shown in Fig. 1b. The nanoparticles showed highly agglomerated clusters made up of irregularly shaped particles. This shows that the obtained materials are polycrystalline. Furthermore, the material showed high surface roughness and the presence of some large, defined grains, suggesting the possibility of grain growth during the synthesis. The TEM image (Fig. 1c) confirmed that the obtained nanoparticles were mostly irregular and aggregated. The average particle size of the nanoparticles estimated using the Image J software was 127 ± 31 nm.
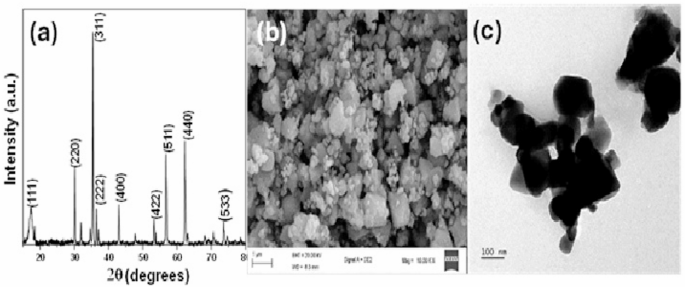
a XRD pattern, b SEM image, and (c) TEM images of ZnFe2O4 nanoparticles
3.2 Absorption spectroscopy
The absorption spectrum of ZnFe2O4 (Fig. 2(a)) shows broad absorption in the UV and visible regions. The first absorption around 200–350 nm is mainly due to charge transfer transitions from the 2p orbitals of oxygen to the metal cations, while d-d transitions of Fe3+ in the octahedral coordination of the spinel structure account for the absorption around 350–500 nm [37]. The extended photo-absorption of the ZnFe2O4 nanoparticle could be ascribed to the presence of sub-energy levels induced by defects in the agglomerated nanoparticles [38]. The broad absorption in the ultraviolet and visible spectra indicates the synthesised material’s potential for photocatalytic activity in both sections of the electromagnetic spectrum. The band gap energy of the material was estimated using the Tauc plot (Fig. 2b). The direct band gap was estimated to be 1.82 eV, which agrees with other reports in the literature [39]. Furthermore, the electronic band alignment of the ZnFe2O4 catalyst was determined using the empirical equations given in Eqs. 1 and 2 [40]:
X represents the absolute electronegativity of the semiconductor, Ee denotes the energy of a free electron on the hydrogen scale (about 4.5 eV), and Eg signifies the band gap energy of the material. From the Muliken electronegativities of the constituent elements of ZnFe2O4, the value of X was estimated to be 6.05 eV. From these values, the Ecb and Evb of the ZnFe2O4 were estimated to be 0.64 eV and 2.46 eV, respectively, which are in close agreement with values reported in previous studies [41].
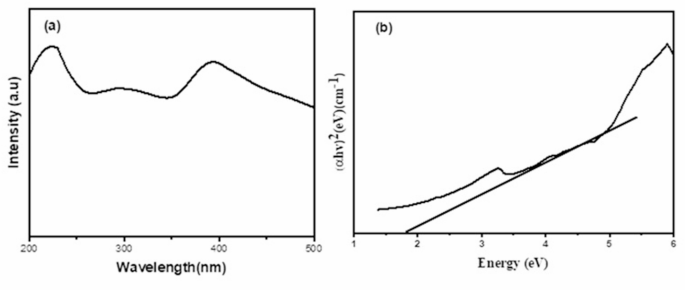
a UV-vis spectrum and (b) Tauc plot for ZnFe2O4
3.3 FTIR analysis
Figure 3 shows the FTIR spectrum of zinc ferrite (ZnFe₂O₄), which reveals the characteristic vibrational bands that confirm the formation and structure of the spinel ferrite phase. The peaks near 3000 cm⁻¹, correspond to the stretching vibrations of aliphatic C–H bonds, likely originating from residual reactant or surfactants used during synthesis. A distinct band around 2350 cm⁻¹ may be ascribed to C‒O, which may arise from the organic residues. The Zn‒O‒Zn strain appears as several peaks that merge into a broad band found around wave numbers of 1420–1655 cm-1, consistent with earlier reports [42]. The main absorption band of ZnFe2O4 is identified in the wavenumber range 500–900 cm−1, corresponding to the stretching vibrations of the metal oxygen bond (Zn-O-Fe). The prominent vibrations below 700 cm⁻¹, particularly at approximately 550 cm⁻¹ and 450 cm−1, are assigned to the stretching vibrations of Zn-O and Fe-O bonds in the tetrahedral and octahedral sites of the spinel structure, consistent with literature [43]. These low-frequency bands are definitive signatures of the spinel ferrite structure and confirm the successful synthesis of ZnFe₂O₄. Recent studies also corroborate these identified peaks, emphasizing that the presence of Fe–O and Zn–O bands in the 400–700 cm⁻¹ region is a reliable indicator of spinel ferrite formation [44,45,46].

FTIR Spectra of ZnFe2O4
3.4 Thermal analysis
The thermal behavior of the synthesized ZnFe₂O₄ nanoparticles was evaluated using thermogravimetric analysis (TGA) and differential scanning calorimetry (DSC) under a nitrogen atmosphere. The thermograms, presented in Fig. 4, reveal a two-stage thermal decomposition process. The sample remained thermally stable up to approximately 90 °C, beyond which a slight weight loss was observed, likely due to the evaporation of physically adsorbed moisture. A more pronounced weight loss of ~ 2% occurred between 400 °C and 850 °C, which can be attributed to the decomposition of residual organic species and possible crystallization enhancement or phase transitions within the ZnFe₂O₄/α-Fe₂O₃ system [47,48,49]. The DSC curve supports this observation, displaying a broad exothermic peak in the same temperature range, indicative of crystallization and structural transformation. These findings confirm that ZnFe₂O₄ achieves full thermal stability and crystallinity at temperatures above 850 °C.
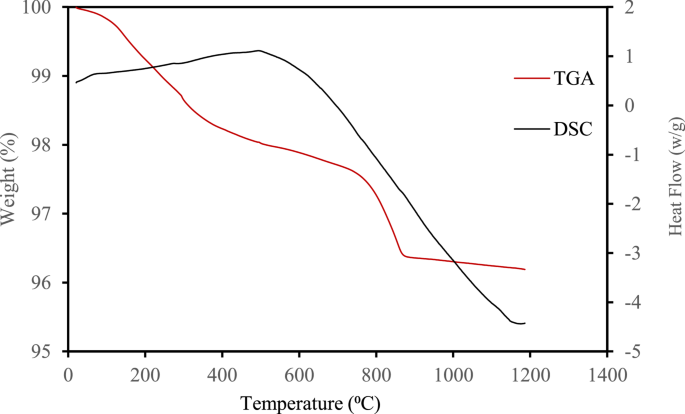
Overlaid TGA/DSC graph of ZnFe2O4
3.5 Catalytic activity studies
The photocatalytic activation of the persulfate activity of ZnFe2O4 was evaluated using tetracycline as the target pollutant. As shown in Fig. 5(a), in the presence of only visible light, the degradation of TC was negligible, showing the low efficiency of photolysis for the degradation of TC. Also, with only PS, the degradation of TC was also negligible. However, when ZnFe2O4 was employed in the activation of PS in the absence of light, the degradation of TC reached 66.9%, showing the significant activation of PS by the ZnFe2O4 catalyst. Furthermore, upon the activation of persulfate by ZnFe2O4 was done in the presence of light, the degradation of TC reached 97.3%, showing a significant synergy between the persulfate activation by ZnFe2O4 and the photocatalytic activity of ZnFe2O4. The effect of PS concentration on the degradation process was further estimated, and it was observed that at a concentration above 1 mM, the degradation process decreased slightly. This observation could be ascribed to the scavenging of other radical species such as and by excess persulfate anions as shown in Eq. (3). Also, at high concentrations, sulfate radicals could undergo self-quenching as shown in Eq. (4), resulting in diminished pollutant degradation [50].
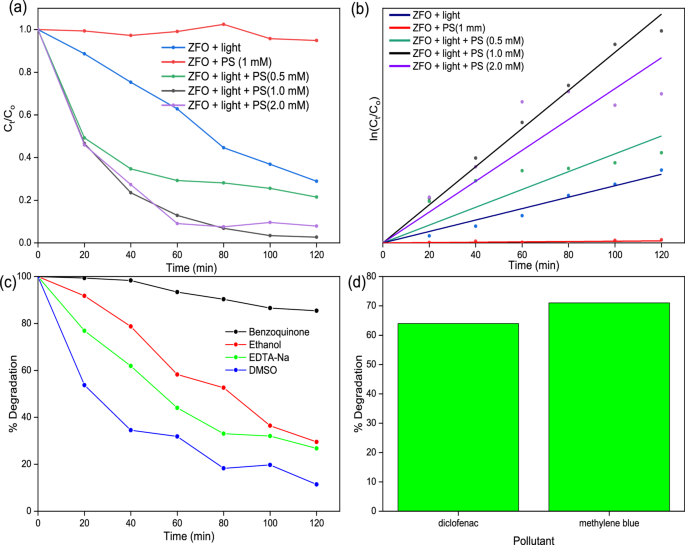
a Degradation profile of TC, b Pseudo first order kinetics plot for TC degradation, c radical scavenging experiment for TC degradation, and (d) Degradation efficiency for diclofenac and methylene blue
The kinetics of the degradation process were fitted using the pseudo-first-order kinetics, as shown in Fig. 5b. The rate constant (k) for the catalytic activation of PS was 2.89 × 10−4 min−1, while the value of k for the photocatalytic degradation of TC in the presence of ZnFe2O4 alone was 9.25 × 10−3 min−1. For the photocatalytic activation of PS in the presence of ZnFe2O4, the value of k was 3.25 × 10−2 min−1. To further establish the wide applicability of the photocatalytic activation of persulfate by ZnFe2O4, the system was explored for the degradation of diclofenac and methylene blue dye. As shown in Fig. 5c, the process showed appreciable activity against both pollutants, achieving a degradation efficiency of 64% and 71% for diclofenac and methylene blue, respectively.
To understand the mechanism of action of the catalyst in the degradation of TC, radical scavenging experiments were carried out to estimate the influence of radical scavengers on the efficiency of the degradation process. Benzoquinone, ethanol, DMSO and EDTA-Na were employed as , , , and scavengers, respectively. As shown in Fig. 5c, benzoquinone had the most significant inhibitory effect on the degradation of TC by the photocatalytic persulfate activation process. This shows that the O2•– radical was the most dominant radical species in the degradation of TC, while the other radicals only played lesser degradation roles in the photocatalytic process. Based on the band alignment of ZnFe2O4, the generation of the O2•– cannot be accounted for by the photogenerated e−, since the potential of the conduction band of ZnFe2O4 is more positive than the redox potential of O2/O2•– (0.16 eV) [51]. However, the photogenerated h+ has a higher potential than the (2.40 eV), which makes the generation of possible. Therefore, the most probable source of the could be through secondary reactions involving other radical species and as shown in Eqs. 5 and 6 [52].
These reactions are most probable since the radical scavenging experiments showed their presence in the degradation process and thus must have been converted into for the degradation of tetracycline. Figure 6 shows a schematic representation of the proposed mechanism for the degradation of TC in the photocatalytic activation of persulfate by ZnFe2O4 nanoparticles.
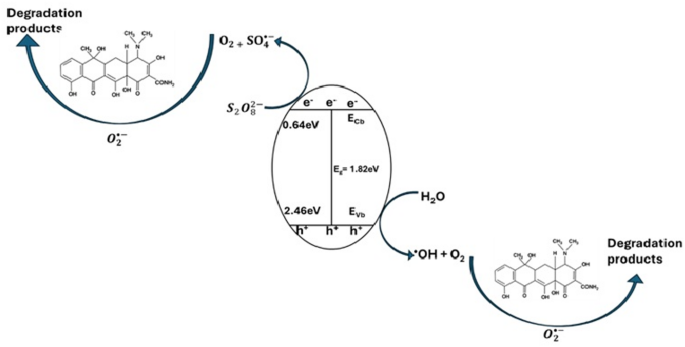
Proposed mechanism of TC degradation by the photocatalytic activation of persulfate by ZnFe2O4 nanoparticles
4 Conclusion
In this study, zinc ferrite (ZnFe₂O₄) nanoparticles were successfully synthesized via a co-precipitation method and demonstrated promising photocatalytic activity for the degradation of tetracycline (TC) in aqueous media. The incorporation of persulfate (PS) significantly enhanced the photocatalytic efficiency of ZnFe₂O₄ under visibe light irradiation, achieving up to 97.3% degradation efficiency. Radical scavenging experiments confirmed the predominant role of superoxide radicals in the degradation mechanism, while kinetic analysis revealed a substantial increase in the rate constant when persulfate activation was combined with photocatalysis. The nanomaterial also showed notable degradation capability for other emerging contaminants such as diclofenac and methylene blue, underscoring its versatility for broader environmental applications. Overall, the synergistic effect between ZnFe₂O₄ and persulfate offers a promising strategy for the remediation of antibiotic and pharmaceutical pollutants in water, supporting the advancement of sustainable wastewater treatment technologies aligned with global environmental goals.
