Article Content
1 Introduction
The demand for advanced biomaterials in healthcare has intensified due to the exponential growth of the global population, increasing life expectancy, and the prevalence of degenerative diseases, congenital disabilities, and injuries from accidents and conflicts [1, 2]. These materials are crucial in medical applications, especially tissue and organ replacement. A thorough understanding of their properties is necessary to improve quality and performance. Titanium and titanium alloys are particularly significant for their use in osteosynthesis because of their excellent biocompatibility and mechanical properties, such as bone [3,4,5,6].
Extensive research has shown that surface roughness and porosity significantly influence the interaction between tissue and implants [7,8,9,10]. In particular, nanostructures like TiO2 nanotubes (TNTs) are beneficial due to their structural properties that mimic natural bone, enhancing osteoblast adhesion and proliferation [11,12,13]. Since their introduction in 1999, TNTs have been extensively explored, with anodic oxidation being an essential technique for their fabrication [14]. This process allows precise control over nanotube dimensions, such as length and diameter, which are crucial for optimizing biological responses and corrosion resistance of this material [15,16,17].
Anodic oxidation parameters, such as the applied potential, duration of the process, and fluoride ion concentration, are crucial in determining the morphology and properties of TNTs [18,19,20]. These parameters influence the structural properties of nanotubes and their functional characteristics, including surface wettability and protein adsorption, which are crucial for cell interactions. Therefore, a stable oxide film on the surface can facilitate regeneration in the presence of physiological fluids, thereby improving corrosion resistance.
Despite the advancements, challenges still exist in optimizing the process parameters for anodic oxidation in HF solutions to balance biomaterial surfaces’ biocompatibility and corrosion resistance. Previous studies have often focused solely on analyzing the effect of individual anodic oxidation process (AOP) parameters, highlighting the crucial need for a more comprehensive approach. This study aims to fill this gap by investigating the combined effects of AOP parameters (voltage, time, and HF concentration) on the cellular adhesion and corrosion resistance of nanostructured TiO2 surfaces.
This paper contributes to the existing knowledge on the optimization of TiO2 nanotubes formation by anodic oxidation in HF electrolytes; its key contributions are as follows:
- Identification of optimal anodization parameters: It has been determined that the combination of moderate anodization voltage and prolonged time in HF solutions allows the formation of TiO₂ nanotubes with appropriate diameters and lengths, which optimizes the corrosion resistance and biocompatibility of treated surfaces.
- Relationship between nanotube morphology and cell adhesion: The research reveals a significant correlation between TiO₂ nanotube size and cell adhesion capacity, highlighting that larger diameter nanotubes favor cell interaction, which is crucial for biomedical applications, such as orthopedic and dental implants.
- Improved understanding of the balance between biocompatibility and corrosion resistance: The study provides new insights into how anodization parameters influence corrosion resistance and biocompatibility, guiding the design of implant surfaces with optimized properties that balance these two critical aspects.
2 Materials and methods
This section details the materials and methods employed in the present study, outlining the experimental procedures and protocols followed to achieve the research objectives.
2.1 Materials
Ti6Al4V ELI (extra low interstitial) alloy was used in this study. This titanium alloy is widely employed in surgical implant applications due to its excellent strength-to-weight ratio, biocompatibility, and corrosion resistance. The properties of this alloy are defined by the ASTM F136 standard, which specifies the chemical composition (see Table 1) and mechanical properties required for medical-grade titanium alloy. In the present study, cylindrical disc-shaped samples of Ti6Al4V ELI were used, with dimensions of 12 mm in diameter and 2 mm in thickness.
All chemical reagents used in this study, including hydrofluoric acid (HF) and nitric acid (HNO₃), sodium chloride (NaCl), potassium chloride (KCl), sodium phosphate dibasic anhydrous (Na₂HPO₄), potassium dihydrogen phosphate (KH₂PO₄), calcium chloride (CaCl₂), magnesium sulfate heptahydrate (MgSO₄·7 H₂O), magnesium chloride hexahydrate (MgCl₂·6 H₂O), D-glucose and sodium bicarbonate (NaHCO₃) were purchased from Sigma-Aldrich and used without further purification.
2.2 Sample preparation
Ti6Al4V Alloy cylindrical discs were ground with sandpaper of different grades, from 120 to 1200 grit. The ground surfaces were cleaned in an ultrasonic bath for 5 min in distilled water and 10 min in ethanol.
2.3 Anodic oxidation process
Hydrofluoric acid dissolved in distilled water was used as the electrolyte for the anodic oxidation process (AOP). Before the AOP, the existing oxide layer on the disc surface was removed by immersing the specimens in an acid mixture of 10 mL of HF, 45 mL of HNO3, and 45 mL of distilled water for 1 min. Ti6Al4V ELI discs were used as the working electrode (anode), and a platinum mesh was used as the counter electrode (cathode). The parameters used to obtain nanostructured titanium oxide surfaces are shown in Table 2. After the AOP, the samples were subjected to an extensive cleaning protocol to remove any potential residual HF and prevent toxicity. This was achieved through ultrasonic baths with distilled water for 15 min and ethanol for 15 min. Previous studies have demonstrated that proper neutralization steps and prolonged washing with distilled water can effectively minimize the impact of residual fluoride ions, ensuring biocompatibility [21, 22]. Therefore, the anodic samples were dried in an oven at 50 °C for 30 min.
The selection of the anodic oxidation parameters (applied potential, time, and HF concentration) is based on optimizing the formation of TiO2 nanostructures, which is crucial for enhancing biocompatibility and corrosion resistance properties in biomedical applications. Recent studies have shown that voltage significantly influences the morphology and size of TNTs. Higher voltages increase the nanotubes’ diameter and the oxide layer’s growth rate [20]. Hydrofluoric acid concentration affects the formation of nanotubes through the chemical dissolution process of TiO2. Higher concentrations of HF increase etching and nanotube formation [23, 24], and the time is also critical in determining the length of the nanotubes, with longer anodic oxidation times resulting in longer nanotubes and thicker oxide layers [19].
2.4 Criteria for sample selection
Following the AOP process, the morphological characteristics of each sample were evaluated using scanning electron microscopy (SEM). The primary focus of this study is on the formation and optimization of TiO₂ nanotubular surfaces due to their well-documented advantages in enhancing biocompatibility and corrosion resistance. Consequently, only samples that exhibited clear nanotubular morphology were selected for further electrochemical behavior analysis and biological response testing. Samples that displayed amorphous or porous morphologies were excluded from further testing as they fall outside the scope of this research.
2.5 Morphological characterization
The nanostructures were observed by scanning electron microscopy using a microscope FESEM Quanta FEG 650. The roughness of the titanium dioxide nanostructures growing under different conditions was characterized using an atomic force microscope AFM NX10 Park System using a tip NCHR in tapping mode. The scanning area was 50 × 50 μm.
2.6 Surface wettability
The surface wettability was measured using the sessile drop method with a Dataphysics OCA 15EC goniometer. A 3 µL droplet of Hank’s solution was carefully placed on the surface of the samples. The contact angle was measured three times per sample, and the average contact angle from these measurements was reported. The measurements were performed at room temperature, and all results were obtained from three independent trials to ensure accuracy and repeatability of the data.
2.7 Electrochemical behavior
The electrochemical characterization was performed using a Gamry 600 potentiostat, with Hank’s solution as the electrolyte. The electrolyte pH was adjusted to 7.4, and the working temperature was maintained at 37 ºC to simulate physiological conditions. Hank’s solution contained 8.0 g/L of NaCl, 0.4 g/L of KCl, 0.048 g/L of Na₂HPO₄, 0.06 g/L of KH₂PO₄, 0.14 g/L of CaCl₂, 0.2 g/L of MgSO₄·7 H₂O, 0.1 g/L of MgCl₂·6 H₂O, 1.0 g/L of D-glucose, and 0.35 g/L of NaHCO₃. A conventional three-electrode cell was used, where the Ti6Al4V ELI samples were the working electrode, a graphite high-purity rod was the counter electrode, and Ag/AgCl (3 M KCl) was a reference electrode. For each sample, the open circuit potential was recorded for 1 h. The potentiodynamic polarization behavior of the samples was performed by applying a scan from − 500 mV to + 2500 mV (versus open circuit potential) at a scanning rate of 1 mV/s. All electrochemical tests were performed in triplicate to ensure data reproducibility.
2.8 Biological response
The human osteosarcoma cell line (HOS, ATCC CRL-1543) was selected for the biological tests due to its relevance in orthopedic material studies, as it simulates human osteoblast behavior in response to biomaterials [23]. Owing to its osteosarcomatous origin, this cell line is particularly suitable for evaluating biocompatibility and cell adhesion in orthopedic and dental implants, facilitating the assessment of cell-material interactions [24,25,26].
Before the tests, HOS cells were maintained in culture flasks using RPMI-1640 medium supplemented with 10% heat-inactivated FBS (hiFBS, Gibco, USA) and penicillin/streptomycin at a final concentration of 100 IU/mL and 100 µg/mL, respectively. Although DMEM or EMEM are commonly used for culturing HOS cells, RPMI-1640 was selected in this study based on previous experience in our laboratory and supporting literature [25, 26], showing no negative effects on cell viability, morphology, or adhesion under our experimental conditions. The cells were cultured following standard protocols as recommended by the manufacturer at 37 °C in a 5% CO2, 95% air mixture, and saturated humidity. For subculturing, the cells were detached from the flask surface using a solution of trypsin (0.25% w/v) – EDTA (0.53 mM) for 10 min at 37 °C. The 10-minute incubation time was selected based on prior experience with this cell line in our laboratory and is in line with ATCC guidelines; cell viability consistently remained above 90% as confirmed by eosin yellow staining. The cells were washed with culture medium, centrifuged, and resuspended in fresh medium.
For the adhesion assay, the methodology previously described in [27] was followed. Briefly, 2.5 × 10⁴ cells/mL in the culture medium were placed on each surface and incubated under the same conditions of temperature and atmosphere used during the initial culture. A non-anodized Ti6Al4V ELI sample was used as a control to compare cell adhesion on treated and untreated surfaces. The Ti6Al4V ELI control sample was prepared with a smooth, polished surface by initially polishing the material to remove any oxide layer and ensure a consistent baseline for comparison. No anodization treatment was applied to this sample, and it served as the non-modified control in the study. After 3 days of incubation, the cells were detached using 0.5 mL of trypsin-EDTA solution for 5 min and counted microscopically in a Neubauer chamber using the vital dye eosin yellow. All biological tests were performed in triplicate to ensure the statistical validity of the results.
One-way analysis of variance (ANOVA) was used to determine the differences between the groups in the adhesion assay experiments. The Tukey-Kramer test was then applied to determine statistical differences when comparing two groups. Statistical significance was considered at *p < 0.05 and **p < 0.01.
This methodology accurately evaluates HOS cell biocompatibility and adhesion capacity to the tested materials and allows for a direct and controlled comparison with untreated reference surfaces.
2.9 Statistical analysis
A Pearson correlation analysis was conducted to explore the relationships between the anodization parameters (voltage, time, and HF %v/v), the corrosion current density (Icorr), and cell adhesion (Cells/cm²), specifically on the selected samples that exhibited a nanotubular morphology. This analysis used Python to calculate the correlation coefficients, quantifying the strength and direction of the linear relationships between these variables. The purpose of this analysis was to identify key anodization conditions that optimize both corrosion resistance and biocompatibility. The correlation results provided insights into the influence of each parameter on the desired surface properties, helping to guide the optimization process.
3 Results and discussion
This section presents a detailed analysis of the morphological, electrochemical, and biological properties of TiO₂ nanostructures formed under various anodic oxidation parameters, emphasizing their relevance for optimizing biocompatibility and corrosion resistance in biomedical applications.
3.1 Morphology of the nanostructures
Figure 1 shows the morphologies of the nanostructured layers obtained under different anodic oxidation conditions. Samples M1, M5, and M8 surface display amorphous TiO2 structures attributed to specific anodizing conditions such as short anodizing times or high HF concentrations. The amorphous morphology observed in M1 is associated with the short anodizing time. In the case of M5, the amorphous TiO2 layers are due to the prolonged anodizing time, which favors the dissolution of structures formed in shorter periods. For M8, the combination of high applied potential, high HF concentration, and prolonged time resulted in amorphous TiO2 layers, mainly due to the dissolution of the TiO2 surface, as reported in previous studies [27].

FE-SEM images of titanium dioxide nanostructures formed on Ti6AL4V under varying anodization conditions. Images correspond to samples (a) M1a, (b) M2p, (c) M3 p, (d) M4t, (e) M5 a, (f) M6 t, (g) M7 t, and (h) M8. aamorphus TiO2 layer, ppore and tnanotubuluar
Conversely, samples M2 and M3 exhibited nanoporous structures, consistent with the literature [28]. This morphology results from the anodic oxidation conditions where the kinetics of anodic layer formation and dissolution are nearly simultaneous, preventing the formation of well-defined nanotubes. Sample M2 shows early-stage nanotubular formation, which is not fully developed due to the slow anodizing rate. In M3, a smoother surface with nanopores was observed, attributed to the high HF concentration used during AOP.
The morphological analysis of discs M4, M6, and M7 reveals the formation of tubular nanostructures. These results align with [29], who observed similar morphologies under comparable anodic oxidation conditions. The average diameters for these samples were 33 nm, 73 nm, and 92 nm for M4, M6, and M7, respectively, primarily associated with the applied voltages and time. According to [30], the nanotube size is directly related to the applied voltage of 30 V for M7. The difference between M4 and M6 is due to the anodizing time of 1 and 3 h, respectively.
3.2 Surface properties
Surface roughness characterization using AFM reveals significant differences among the samples studied. The average roughness (Ra) values and their standard deviations are presented in Table 3.
The anodized surfaces (M4, M6, and M7) exhibit higher roughness than the untreated Ti6Al4V substrate. This increase in roughness is directly related to the formation of nanotubular structures during the anodization process, as shown in Fig. 2. Previous studies [31] and [32] have demonstrated that nanometric roughness values ranging from 100 to 300 nm are optimal for promoting the adsorption of extracellular matrix proteins, such as fibronectin and vitronectin, which mediate initial cell adhesion processes.
Among the samples, M7 exhibits the highest roughness (217.00 ± 18.45 nm), suggesting a superior capacity to favor protein adsorption and calcium phosphate nucleation. This phenomenon is crucial for apatite formation in physiological environments, as increased roughness provides a larger surface area for interaction with biomolecules and ions such as Ca²⁺ and PO₄³⁻ [33].
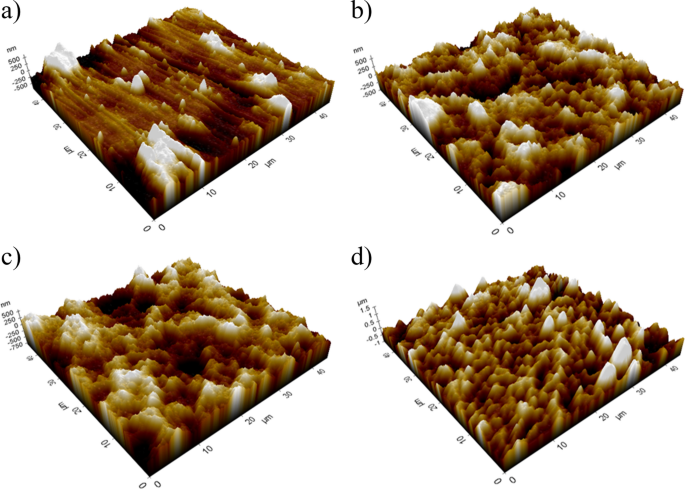
3D AFM images of TiO2 nanotubular surfaces formed under optimized anodic oxidation parameters: (a) Ti6Al4V, (b) M4, (c) M6, (d) M7
Contact angles for all samples are shown in Table 3. The contact angle is a key parameter for evaluating the wettability of a surface, which directly affects its ability to adsorb proteins and promote cell adhesion. Surfaces with contact angles below 90° (hydrophilic) tend to adsorb a greater quantity of proteins in conformations that enhance their biological functionality, whereas hydrophobic surfaces (θ > 90°) often limit this interaction [9, 34].
When comparing samples M4 and M6, it is observed that an increase in anodization time results in a decrease in surface roughness and a concurrent increase in the contact angle. This behavior can be attributed to the evolution of the nanotubular morphology during prolonged anodization, where the structures become more homogeneous and vertically aligned, reducing the microscale surface irregularities. Furthermore, the alignment of nanotubes promotes a Cassie-Baxter wetting regime, where air pockets trapped between the nanotubes reduce the liquid-solid contact area, thereby increasing the apparent contact angle [27].
When comparing samples M4 and M7, it is observed that an increase in anodization potential leads to a significant increase in surface roughness and a decrease in the contact angle. This behavior can be explained by the intensified field-assisted oxidation and dissolution processes at higher voltages, which form larger-diameter nanotubes and a more textured surface morphology. Furthermore, the enhanced development of open-ended nanostructures promotes water infiltration into the nanotubes, favoring a more hydrophilic response [27].
Sample M7, with a contact angle of 78.90 ± 1.90° (hydrophilic), is particularly well-suited for biomedical applications, as this property facilitates the adsorption of key proteins involved in cell adhesion and osteoblast recruitment. Furthermore, the hydrophilic nature of M7 also favors the precipitation of calcium phosphate, a critical process for apatite formation and bone integration in implants [35, 36].
In contrast, sample M6, with a contact angle of 130.90 ± 2.60° (hydrophobic), may offer advantages in applications where bacterial biofilm formation needs to be minimized. However, the lower initial interaction with physiological fluids could reduce its ability to promote apatite formation.
The combination of high roughness (Ra = 217.00 ± 18.45 nm) and hydrophilic behavior (θ = 78.90 ± 1.90°) in sample M7 highlights its potential as an optimal bioactive surface. These properties indicate a greater capacity to absorb extracellular matrix proteins, promote cell adhesion, and facilitate calcium phosphate nucleation. This balance between roughness and wettability is essential for biomedical implants designed for orthopedic and dental applications [37, 38].
3.3 Electrochemical behaviour
Figure 3 shows typical potentiodynamic polarization curves for smooth Ti6Al4V and TiO2 nanostructured surfaces. All specimens exhibited a passive zone when the applied potential reached 1 V vs. open circuit potential (OCP), indicating no pitting corrosion [39]. The samples M4 and M7 exhibited the highest current densities in the passive zone, likely due to the anodic layers’ instability, allowing the electrolyte to attack the alloy’s base material easily.
Corrosion parameters, calculated from the polarization curves using the Tafel extrapolation method, are shown in Table 4. Specimens M4 and M6 presented the lowest corrosion rates in this study, especially sample M6, which has the lowest passivation current density and, therefore, is more stable. These results are attributed to small-diameter nanotubes forming on the surfaces after AOP at 20 V with 0.5% v/v HF for 1 and 3 h, respectively. AOP was expected to improve the alloy’s corrosive behavior; however, the results indicate otherwise, which can be explained by considering the report by [39], attributing this behavior to the dissolution of the β phase, exposing the material’s surface to electrolyte attack. Additionally, inappropriate HF concentrations produced fragile and unstable TiO2 nanostructures, reducing the films’ protective qualities.
When comparing samples M4 and M7, it is observed that an increase in anodization potential leads to a significant increase in surface roughness and a decrease in the contact angle. This behavior can be explained by the intensified field-assisted oxidation and dissolution processes at higher voltages, which form larger-diameter nanotubes and a more textured surface morphology. Furthermore, the enhanced development of open-ended nanostructures promotes water infiltration into the nanotubes, favoring a more hydrophilic response.
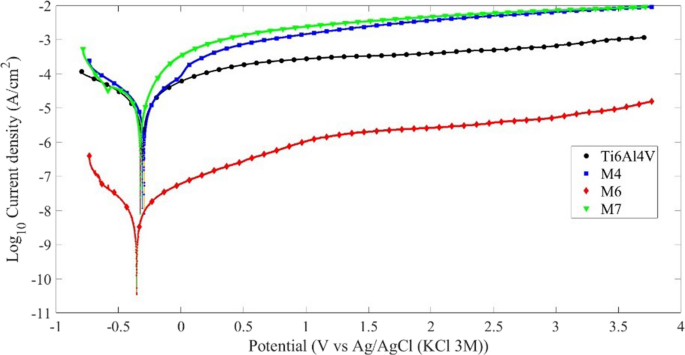
Potentiodynamic polarization curves of TiO2 nanostructured surfaces (M4, M6, M7) and untreated Ti6Al4V alloy recorded in Hank’s solution (pH 7.43, 37 ºC)
3.4 Biological assays
The results of the in vitro adhesion of HOS cells to titanium dioxide nanostructures, obtained under different anodic oxidation parameters, are shown in Fig. 4. The cell adhesion counts for samples M6 and M7 showed a higher number of cells adhered to the surface than the Ti6Al4V control. Average values of 8.2 × 10⁴, 1.5 × 10⁵, and 6.3 × 10⁴ cells/cm² were found for M6, M7, and Ti6Al4V, respectively.
This significant increase in cell growth on M7 compared to Ti6Al4V was statistically significant (P < 0.05), suggesting that the nanotubular morphology and a tube diameter of approximately 92 nm promote cell growth. No significant differences were found for M6 and M4 compared to the control. On the other hand, a considerable decrease was observed for sample M4 (2.9 × 10⁴ cells/cm²).
This finding is consistent with previous studies suggesting that surface topography and the physical properties of nanostructures, such as the size and arrangement of nanotubes, play a decisive role in cell adhesion and proliferation. The nanotubular morphology observed in sample M7 could increase the available surface area for cell interaction, improving initial adhesion and creating a more favorable environment for cell growth. Additionally, it has been proposed that the diameter of the nanotubes plays a crucial role in cell orientation and intracellular signaling, which could explain the significant increase observed in this sample.
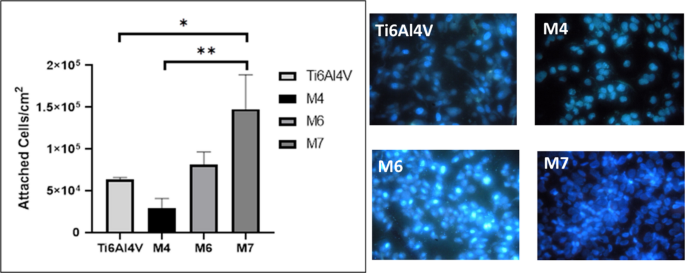
HOS cell adhesion results on TiO2 nanostructured surfaces (M4, M6, M7) compared to untreated Ti6Al4V alloy
The diameter of the TiO₂ nanotubes significantly influences cell adhesion and growth. Nanotubes with smaller diameters (typically in the range of 20–100 nm) are known to improve cell adhesion by providing a higher surface area for protein adsorption and by promoting the formation of focal adhesions, which are essential for cellular attachment and spreading. Moreover, smaller diameters create a more favorable environment for cell alignment and orientation. Conversely, larger nanotube diameters can hinder cell spreading and reduce cellular interaction with the surface, as the distance between nanotube walls becomes too large to support optimal cell attachment. Therefore, an optimal nanotube diameter is critical for achieving improved cellular responses, including enhanced cell growth and differentiation.
Similar results have been reported in previous studies, such as the work by [40], which demonstrated that the dimension of titanium nanotubes can dictate cell fate, promoting specific cellular behaviors like proliferation or differentiation. Furthermore, studies like [41]confirm that surfaces with TiO2 nanotubes enhance cell adhesion and proliferation, supporting these findings.
3.5 Correlation analysis
A Pearson correlation analysis was conducted to further understand the relationship between the anodization parameters and the surface properties of TNTs. This analysis focused on the relationships between the anodization voltage, anodization time, corrosion current density, and cell adhesion. The results are summarized in Table 5.
The Pearson correlation analysis revealed a strong positive correlation between anodization voltage and corrosion current density (r = 0.997) and cell adhesion (r = 0.885), indicating that higher voltages lead to increased corrosion rates and enhanced biocompatibility. Conversely, anodization time showed a moderate negative correlation with corrosion current density (r = −0.600), suggesting that longer anodization times contribute to reduced corrosion, likely due to the formation of thicker oxide layers. However, the impact of anodization time on cell adhesion was minimal, as evidenced by the weak positive correlation (r = 0.200). Interestingly, a high positive correlation between corrosion current density and cell adhesion (r = 0.870) suggests a trade-off in which conditions that favor biocompatibility might also increase susceptibility to corrosion. These findings highlight the need for careful optimization of anodization parameters to balance corrosion resistance and biocompatibility in TiO₂ nanotube surfaces.
The Pearson correlation analysis highlighted the critical relationships between anodization parameters and surface properties, revealing a strong influence of voltage on corrosion current density and cell adhesion. This result is further supported by our experimental observations, where M4, M6, and M7 exhibited well-formed nanotubes with diameters ranging from 33 nm to 92 nm, consistent with the correlation between applied voltage and nanotube size, as reported by [30]. The larger nanotube diameters observed in M7, for instance, align with previous studies suggesting that nanotube diameters around 100 nm are optimal for cell adhesion and differentiation [35, 42, 43].
Additionally, the variability in sample hydrophobicity observed across the different samples correlates with surface topology and chemistry, which significantly influence wettability, as supported by [35, 36]. In our study, the increased hydrophilicity of larger nanotube surfaces likely results from the greater surface area, facilitating protein adsorption and enhancing cellular interaction.
The potentiodynamic polarization curves provided further insights, indicating a passive zone in all specimens, which suggests adequate corrosion resistance under physiological conditions. However, M1, M3, M5, and M7 showed higher current densities in the passive zone, potentially due to the instability of the anodic layers formed under specific conditions, a phenomenon reported in studies by [32, 37, 38]. This instability may be linked to the dissolution of the β phase in the Ti6Al4V alloy, particularly in high HF concentrations, which compromises corrosion protection [39, 42].
Interestingly, while M7, with its nanotubes approximately 92 nm in diameter, showed the best results in terms of cell adhesion, no significant differences were observed when compared to the control group. This finding suggests that factors beyond nanotube size, such as surface chemistry and roughness, are also crucial for optimizing cell adhesion. These results reinforce the notion that a careful balance of anodization parameters is essential for achieving desirable nanotube dimensions and enhancing biocompatibility and corrosion resistance.
4 Conclusions
This study provides insights into how anodization parameters affect the properties of TiO₂ nanotubular surfaces on Ti6Al4V alloy. The results show that applied voltage and anodization time determine nanotube diameter, impacting corrosion resistance and cell adhesion.
The Pearson correlation analysis shows a strong positive correlation between anodization voltage and corrosion current density and cell adhesion. This indicates that higher voltages enhance both corrosion susceptibility and biocompatibility. This dual effect underscores the necessity for optimizing anodization parameters to balance these critical surface properties.
Among the samples tested, M6, which was anodized at 20 V for 3 h, exhibited the most favorable combination of low corrosion current density and adequate cell adhesion. This suggests that lower voltages and extended anodization times produce more stable oxide layers, enhancing corrosion resistance without significantly compromising cell adhesion.
Additionally, the study highlighted the role of surface wettability, where larger nanotube diameters, as seen in M7, increased hydrophilicity, likely due to greater surface area facilitating protein adsorption and cellular interaction. However, despite the high cell adhesion observed in M7, the potential for increased corrosion emphasizes the need for a careful balance between enhancing biocompatibility and maintaining corrosion resistance.
Finally, this study’s findings suggest that the anodization process parameters must be finely tuned to optimize the trade-offs between corrosion resistance and biocompatibility. Future research should further refine these parameters and explore the synergistic effects of surface chemistry and topography to develop more robust and biocompatible titanium-based implants.
These advancements support the goals of improving healthcare technologies and sustainable innovation, in line with the United Nations Sustainable Development Goals, particularly SDG 3 (Good Health and Well-being) and SDG 9 (Industry, Innovation and Infrastructure).
