Article Content
Introduction
Complementary medicine (CM) products are widely used in many countries, including Australia, where they are regulated medicines under a low-risk framework [1]. CM products are generally regarded as safe for direct purchase by consumers without clinical supervision or consumer medicine information beyond the product label, which typically does not include information about potential drug interactions. Despite being considered low risk, CM products can interact with other medicines and result in negative clinical outcomes not observed when either is used alone [2, 3]. In particular, herbal ingredients are complex, multi-constituent compounds with demonstrable pharmacological actions, posing risks for both pharmacokinetic and pharmacodynamic herb–drug interactions [2].
The real-world prevalence of CM–drug interactions is difficult to estimate because of chronic underreporting to pharmacovigilance systems [4] that in part reflects a lack of healthcare professional enquiry [5] and low consumer awareness of potential risks [6]. Consequently, indirect evidence from surveys measuring concurrent use of CM and other medicines are often used to estimate the risk of harm from CM–drug interactions. For instance, the reported prevalence of concurrent use of prescription drugs and herbal medicinal products in a systematic review of cross-sectional surveys of older adults ranged from 5.3 to 88.3% [7]. The wide variations reflect differences between study designs, CM definitions, and population groups.
Real-world data quantifying the prevalence of CM–drug interactions in the Australian community settings are lacking. A small cross-sectional study of 404 adult patients attending an emergency department reported the prevalence of potential CM–drug interactions between 1 February 2002 and 31 March 2003 [8]. Data from the 275 CM users indicated a total of 15 (3.3%) CM–drug combinations with clinical evidence of an interaction and an additional 97 (18.6%) combinations with a theoretical risk of an interaction [8].
The most recent indirect data about CM–drug interaction risk in Australia comes from two surveys conducted in 2017 [9, 10]. The first was a cross-sectional survey of 2351 adults [9]. Almost half (49.4%) reported using a CM product. Vitamin and mineral supplements (44.9%) were most common, followed by Chinese herbal medicines (5.1%) and Western herbal medicines (4.8%). Of those who used Chinese or Western herbal medicines, 46.2% and 40.2%, respectively, reported frequently taking them on the same day as taking prescription or over-the-counter medications [9]. The second survey found that CM use was even higher among adults aged ≥70 years, at 64.4%, of which 15% reported using a CM containing herbal ingredients [10]. Notably, polypharmacy was a significant predictor of use [10]. Although these data reveal that concurrent use of prescription medicines and CM is widespread, it remains unknown whether combinations that pose a clinically significant risk are being used. To address the knowledge gap in Australia, we sought to estimate the incidence of two herb–drug interactions: Hypericum perforatum (St John’s Wort) with hormonal contraceptives, and Camellia sinensis (green tea) with atorvastatin. We selected these combinations because of their widespread use and well-documented potential for clinically important consequences (as described below) that are not typically severe enough to warrant reporting as adverse drug reactions.
Nearly 45% of Australian women aged 18–39 years use hormonal contraceptives [11]. One of the most common side effects of the hormonal contraception is mood changes [12]. Hormonal contraceptive use is associated with an increased risk of a first diagnosis of depression and subsequent treatment with antidepressants [13]. Being female is a predictor of CM use [9], and Australians living with mental health problems are among the highest users of CMs [14]. One such product, Hypericum perforatum is widely promoted for supporting mood balance associated with mild to moderate depression and relieving symptoms of anxiety [15, 16]. Therefore, Hypericum perforatum products are likely to attract the attention of women taking hormonal contraception who are also experiencing mood disturbances and prefer natural products.
Numerous in vitro, in vivo, and clinical studies have evaluated the antidepressant activity and clinical effects of Hypericum perforatum [15]. The two major bioactive constituents of Hypericum perforatum are hyperforin and hypericin. Pharmacokinetic studies support that hyperforin is the main constituent implicated in herb–drug interactions [17]. Hyperforin is a potent activator of the pregnane X receptor and therefore induces cytochrome P450 enzymes, notably CYP3A4 and the drug transporter p-glycoprotein, which enhances the metabolism and clearance of hormonal contraceptives [17]. Well-established evidence shows that concomitant intake of Hypericum perforatum 600–900 mg containing 26.7–33.3 mg hyperforin daily with hormonal contraceptives containing ethinylestradiol and norethisterone or desogestrel increases the incidence of breakthrough bleeding [18,19,20]. Further, a significant increase in the oral clearance of norethindrone, a reduction in the half-life of ethinylestradiol, and a 13–15% reduction in dose exposure to hormonal contraceptives has been reported with this combination [18, 19]. A review of 11 cases reported in the UK over a 4-year period (1996–2000) supported that this interaction does occur in the community and resulted in intermenstrual bleeding (n = 6) and unintended pregnancy (n = 5) [15].
Atorvastatin is the most commonly used prescription medicine in Australia, with 80.72 defined daily doses per 1000 population per day [21]. The use of CM is also highly prevalent among Australians living with chronic disease and associated risk factors [9]. In a 2013 survey of 2915 adults in Australia with a diagnosis of cardiovascular disease and/or diabetes mellitus, 40.3% reported using at least one CM, 96.3% of whom were also taking at least one prescription medicine [22].
Camellia sinensis is a plant native to eastern Asia traditionally used for its cardioprotective and weight-reducing properties [23]. As such, people with cardiovascular disease and associated risk factors who are taking medications such as atorvastatin are likely to use Camellia sinensis concomitantly. The green tea catechins have been reported to influence the pharmacokinetics of several drugs [24]. Camellia sinensis extract interacts with atorvastatin by inhibiting organic anion transporting polypeptides and p-glycoprotein, which play a role in the absorption of atorvastatin [25]. A double-blinded randomised cross-over study (n = 12) demonstrated that the concomitant consumption of dry Camellia sinensis extract 300–600 mg and atorvastatin 40 mg daily could significantly decrease atorvastatin plasma concentrations by 24–25% and the atorvastatin area under the plasma concentration–time curve by 22–24% [26]. Conversely, a separate clinical study (n = 24) reported that concurrent use of Camellia sinensis extract 400 mg daily for 10 days significantly increased the bioavailability of atorvastatin by up to 22.7% [27]. Collectively, the clinical implications of these results may seem conflicting, but there is a clear risk of interactions between Camellia sinensis extracts and atorvastatin that warrants a degree of caution and monitoring if used concurrently. In time, evidence may emerge to support a broader concern that concurrent use of Camellia sinensis with any statins could result in an interaction associated with reductions in statin concentrations. However, the evidence to date does not support this assumption. Pharmacokinetic studies have found reductions in rosuvastatin and atorvastatin but not fluvastatin and simvastatin to be associated with Camellia sinensis catechins [24].
Despite the widespread use of these two herbal products and drugs, the true incidence of cases involving hormonal contraceptives and Hypericum perforatum or atorvastatin with Camellia sinensis remains unknown. The aim of this study was to estimate the number of Australians who are potentially exposed to these avoidable CM–drug interactions.
Methods
A basket analysis approach (i.e., investigating combinations of health products purchased together in a single consumer transaction) was used to analyse pharmacy transaction data [28]. The number of instances where a concurrent purchase of a hormonal contraceptive and a product containing Hypericum perforatum, or atorvastatin and a product containing Camellia sinensis were calculated and then extrapolated to estimate the number of people in Australia at risk of either of the CM–drug interactions profiled for the purpose of this study.
Two separate analyses were conducted for the hormonal contraceptives: one for oral hormonal formulations and another for all hormonal contraceptive formulations. This was because the clinical evidence of unwanted interactions between oral contraceptives and Hypericum perforatum is well established, whereas the evidence is only theoretical for non-oral formulations [29].
Data sources
The analysis was conducted using IQVIA Consumer Health (North Sydney, NSW, Australia) pharmacy sales data during the period April 2023 to March 2024. We obtained the pharmacy transaction data from 4168 of the 6215 Australian community pharmacies. These included pharmacy banner groups representing traditional, discounter, and variety pharmacy business models. The basket data covered 67% of pharmacies located across Australia, representing approximately 75% of national sales for prescription and over-the-counter medicines, including some products containing the ingredients Hypericum perforatum or Camellia Senesis.
Market definitions for medicinal products of interest
Oral contraceptive transactions included in the analysis contained:
- ethinylestradiol and levonorgestrel,
- ethinylestradiol and drospirenone,
- levonorgestrel,
- ethinylestradiol and norethisterone,
- drospirenone
- ethinylestradiol and drospirenone,
- estetrol and drospirenone,
- estradiol and dienogest,
- estradiol and nomegestrol acetate,
- ethinylestradiol and desogestrel,
- ethinylestradiol and gestodene,
- norethisterone,
- norethisterone and mestranol.
Drugs used as emergency oral contraceptives, such as levonorgestrel 1.5 mg tablets and ulipristal 30 mg tablets, were excluded. Transactions for vaginal rings containing ethinylestradiol/etonogestrel, intrauterine devices containing levonorgestrel, depot injections containing medroxyprogesterone, and subdermal implants containing etonogestrel were also included in the analysis for all hormonal contraception.
IQVIA identified products available for self-selection that contained Hypericum perforatum as a single ingredient or in a combination as the most commonly sold products (n = 29). We then searched the Therapeutic Goods Administration (TGA) Australian Register of Therapeutic Goods to confirm that these products contained Hypericum perforatum and to identify any other commonly sold products that were missed [30]. Five products were excluded, and two additional products were added, leaving a total of 26 products included in the analysis (14 single-ingredient and 12 combination products). Data on the hyperforin content for each of the products were recorded by reviewing the available product information, including labelling and product webpages.
Transactions containing atorvastatin available in all brands and dose form (and combination products amlodipine/atorvastatin and atorvastatin/ezetimibe) were included in the analysis (n = 71).
IQVIA identified 33 products containing Camellia sinensis as the most commonly sold products. We then searched the TGA Australian Register of Therapeutic Goods to confirm that the products contained Camellia sinensis [30]. A total of 236 products containing Camellia sinensis were registered with the TGA; however, for pragmatic reasons, these were not included in the analysis as they did not represent a substantial percentage of the total sales data and were therefore unlikely to significantly change the estimates. In total, 33 products were selected for the analysis: two single-ingredient products, 11 combination products, and 20 concentrated food extracts. The single and combination green tea products included in this basket analysis contained a maximum daily dose > 300 mg because of the known effects of this dose on atorvastatin plasma concentrations [26].
Analysis
We used IQVIA’s Drug Distribution Data sales data, which captures Australian pharmacies’ sales data, to estimate the total number of patients who had purchased a CM or prescription medicine of interest between April 2023 to March 2024. As illustrated in Tables 1, 2, and 3, since hormonal contraceptives and statins are mostly used continuously over a 12-month period, the annual patient count for hormonal contraceptives and atorvastatin were estimated by the quantity and package size of drug purchased: for hormonal contraceptives and atorvastatin, the total number of transactions in a year was divided by how many months each medication purchase lasts. For example, if the transaction for an oral contraceptive contained a 3-month supply, we divided the total annual unit sales for that product by four to estimate unique individual prescriptions. In contrast, we assumed that each purchase of a CM reflected a 1-month supply.
We analysed transaction data to identify instances when the hormonal contraception or atorvastatin prescription medicine and the CM were purchased together (i.e., occurrence frequency). We then multiplied the estimated proportion (%) of the prescription medicine transactions where there was a risk of the CM–drug interaction of interest by the estimated total annual patient count for that prescription medicine. These are reported as the total number of individuals who were at risk of experiencing the CM–drug interaction (i.e., ‘at risk’ population).
The rates of CM–drug interaction occurrence were considered in the context of the Australian Bureau of Statistics estimated resident population in Australia (n = 26,821,557) as of 30 September 2023 [31].
Results
Oral hormonal contraceptives and Hypericum perforatum product transactions
Upon analysing transactions related to oral contraceptives between April 2023 and March 2024, 0.6% of these transactions included a CM containing Hypericum perforatum. During this period, an estimated 650,207 Australian females had used oral contraceptives (Table 1). This equated to approximately 3901 individuals using oral contraceptives who were potentially at risk of this herb–drug interaction (Fig. 1).
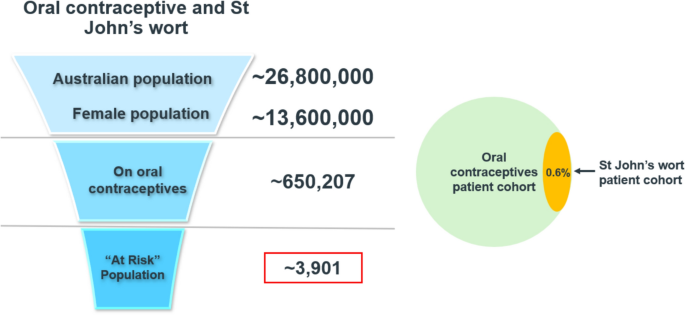
Estimated occurrence frequency of oral contraceptive transactions containing St John’s wort and ‘at risk’ population
Of the 26 Hypericum perforatum products included in this basket analysis, only four products provided information regarding the hyperforin content. Of these, the hyperforin daily dose ranged from 15 to 27 mg per dose. Of the remaining 22 products that did not report hyperforin content, 17 non-liquid formulation products reported the dry herbal extract content with a daily dose ranging from 167 to 1200 mg.
Hormonal contraceptives and Hypericum perforatum product transactions
We conducted a separate analysis to identify the concurrent purchase of hormonal contraceptives in any formulation (including oral) and a CM containing Hypericum perforatum. We found that 2.8% of hormonal contraceptive transactions between April 2023 and March 2024 also included a CM containing Hypericum perforatum. During this period, an estimated 719,949 Australian females filled prescriptions for any form of hormonal contraceptive drugs (Table 2). This means that approximately 20,159 individuals using any form of hormonal contraceptive were potentially at risk of this CM–drug interaction (Fig. 2).
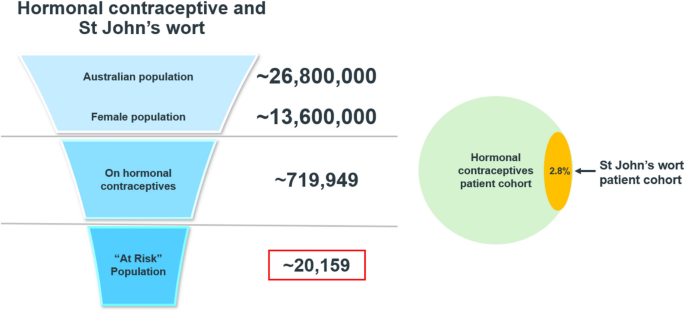
Estimated occurrence frequency of hormonal contraceptive transactions containing St John’s wort and ‘at risk’ population
Atorvastatin and Camellia sinensis transactions
Analysis of transactions related to atorvastatin between April 2023 and March 2024 indicated that 1.8% of these transactions also included products containing Camellia sinensis. During this period, an estimated 1,241,598 Australians were taking atorvastatin (Table 3). This equates to approximately 22,408 Australians potentially at risk of this CM–drug interaction (Fig. 3).
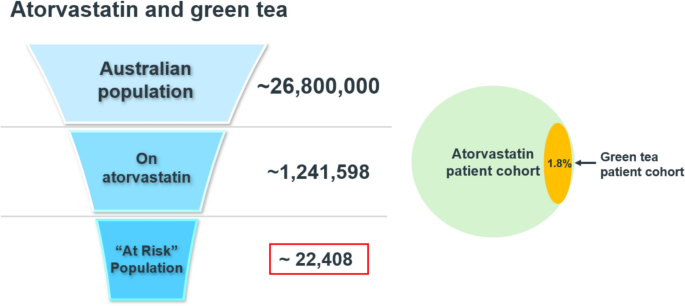
Estimated occurrence frequency of atorvastatin transactions containing green tea and ‘at risk’ population
Discussion
This is the first study to estimate the prevalence of two potential clinically significant CM–drug interactions among the Australian population using a basket analysis. We report the estimated total number of individuals who may have been exposed to an avoidable CM–drug interaction involving a hormonal contraceptive and Hypericum perforatum, or atorvastatin and Camellia sinensis. The results revealed that, between April 2023 and March 2024, a total of 42,567 Australians were at risk of experiencing one of these two herb–drug interactions of interest.
The proportions of oral contraceptive and any hormonal contraceptive transactions that included a CM product containing Hypericum perforatum were 0.6% and 2.8%, respectively. Although concurrent purchases do not confirm that the same individual concurrently used them both, it is a concern. A small cross-sectional survey of adults attending an emergency department in Australia confirmed that females do use these products concurrently, as one of the 15 CM–drug interactions that were identified involved the combination of Hypericum perforatum and an oral contraceptive [8].
There is well-established evidence from clinical trials showing that the concomitant intake of Hypericum perforatum 600–900 mg, containing hyperforin 26.7–33.3 mg, daily with hormonal contraceptives increases the incidence of breakthrough bleeding [18,19,20]. Of the 26 Hypericum perforatum-containing products included in this analysis, only four reported the hyperforin content on the product label, with daily doses ranging from 15 to 27 mg, which is slightly lower than the daily dose range of 26.7–33.3 mg examined in the clinical pharmacokinetic studies [18,19,20]. Whether the dose range of those four products included in the analysis of this study poses a clinically significant risk is unknown, but safety cannot be assumed.
For the other 17 products, the reported the daily dose of Hypericum perforatum dry herbal extract was 167–1200 mg, of which 13 had a daily dose ≥600 mg but no specification of hyperforin content. The remaining four products were either liquid extraction (n = 2) or did not document the extract content. These doses were not comparable to the doses supported by clinical evidence. The Australian TGA requires herbal products to be prepared in accordance with accepted pharmacopeia standards and dosages in accordance with internationally accepted monographs such as the European Scientific Cooperative on Phytotherapy [32] and the European Medicines Agency [33]. The monograph for Hypericum perforatum preparations states that products should contain a dose range of 600–1800 mg of Hypericum perforatum extract, per day, standardized to 3–6% hyperforin and/or 0.12–0.28% hypericin and not to exceed 600 mg of extract per single dose [34]. Since the 17 products did not report standardized hyperforin content on their labels, we used the TGA guidelines and the European Scientific Cooperative on Phytotherapy monograph to extrapolate and estimate that the Hypericum perforatum content (167–1200 mg) would contain 3–6% (36–72 mg) of hyperforin, likely posing a significant interaction risk.
The estimated number of 3901 females identified in this study who were at risk of an oral contraceptive–Hypericum perforatum interaction, along with the additional 16,258 at risk of an interaction with a non-oral contraceptive, has important implications, the most obvious being an unexpected pregnancy. The resultant bio-psycho-social and economic impact to individuals and the healthcare system are profound [35]. The direct and in-direct costs from an unplanned pregnancy arising from this herb–drug interaction include costs associated with termination of pregnancy, live birth, miscarriage, stillbirth, government subsidies for childcare, adoption, income support, parental leave, individuals’ out-of-pocket non-healthcare expenses, childcare costs, lost wages, and employer absenteeism cost [36].
In Australia, all CMs that contain Hypericum perforatum are required to include the statement “St John’s Wort affects the way many prescriptions medicines work—including oral contraceptives. Consult your doctor.” on the product label. Yet, our study identified a large number of concurrent purchases of Hypericum perforatum products and hormonal contraceptives in Australian community pharmacies. The findings strongly suggest that the use of label statements and associated public awareness campaigns are insufficient to mitigate risk. The involvement of pharmacists and staff during CM selection and transactions may potentially reduce the risk of a clinically significant interaction. However, 22 of the 26 St John’s wort products did not list the hyperforin content, which in turn leads to ambiguity for pharmacists and other healthcare professionals when assessing the plausibility and risk of an interaction.
The clinical implications for the estimated 22,408 Australians who were potentially at risk of atorvastatin–green tea interactions are perhaps less profound, as elevated lipids are only one of many risk factors for atherosclerosis vascular diseases and, in ideal circumstances, clinicians would monitor levels and adjust statin doses accordingly. Notwithstanding, if this interaction lowers atorvastatin levels in the individual and therefore its effectiveness, in the longer term there is still an increased risk of cardiovascular events, including myocardial infraction and stroke [37]. Further, perceived ineffectiveness may lead to unnecessary cessation. Conversely, if this interaction increases systematic exposure to atorvastatin, there is the risk of side effects, including myopathy, potentially leading to further instances of unnecessary cessation [38]. Therefore, irrespective of any uncertainties about the direction of potential interactions, patients who concomitantly use atorvastatin and green tea should use precaution, including more frequent monitoring of lipid profiles and adverse effects.
The findings of this study indicate an urgent need for larger-scale studies that seek to understand how Australians are choosing and using CM with other medicines and what specific combinations are being used. The data presented here support the position of researchers in the USA who suggested that there is a need for interdisciplinary strategies, including co-designed consumer-informed health technologies, to support informed decision-making about the concurrent use of CM with other medicines [39, 40]. Shorter-term measures include recommending that pharmacists and pharmacy staff refer to herb–drug interaction checkers when consumers are co-purchasing drugs and herbs to inform counselling consumers about appropriate and safe use.
Limitations Strengths of this study include the use of a population-based dataset that represented around 75% of prescription drugs sold via Australian community pharmacies, and the inclusion of sales data from two-thirds of community pharmacies, representing a wide variety of settings in Australia. However, this study did not capture sales data from retail outlets other than pharmacies, where these two herbal products may have been purchased, such as supermarkets, health food stores, and online health supplement stores. Thus, the total number of people at risk may be slightly higher. The biggest limitation of a basket analysis is that it cannot be assumed that every combination purchased led to the two products being used concurrently. We also assumed that each individual used the prescription medicine for the whole year of the study, as no information was available about patient adherence, new initiations, discontinuations, and duplicate therapy. This will have reduced the denominators in the analyses and therefore increased the estimated proportion of patients at risk by an unknown amount. However, this overestimation is counterbalanced by the prescription sales data in Australia not captured by this study. Other limitations included the omission of information about the clinical indications for using the products. Also, we did not consider confounding factors such as individual pharmacy demographics, and pharmacist and consumer characteristics, including health literacy and decision making about medicines use. Notwithstanding these limitations, the findings confirm that a substantial number of people in Australia were at risk of an avoidable CM–drug interaction.
Conclusion
Herb–drug interactions pose a burden of preventable harm that could be avoided if consumers and healthcare professionals were aware of the risks. Interdisciplinary strategies, including digital health technology, that are co-designed with patient and consumer input are indicated to ensure decisions are informed when CMs including those containing herbal ingredients are being used. This is of particular importance when CMs are used concurrently with prescription and over-the-counter medications.
