Article Content
Abstract
Sodium oxybate extended-release suspension (LUMRYZ™; hereafter referred to as sodium oxybate ER) represents a promising novel option for the treatment of narcolepsy. It is the first once-nightly sodium oxybate formulation to be approved in the USA for the treatment of cataplexy or excessive daytime sleepiness (EDS) in adults with narcolepsy. Sodium oxybate ER has a pharmacokinetic profile that supports a single bedtime dose. In a pivotal phase 3 trial, once-nightly sodium oxybate ER dose-dependently increased sleep latency during the daytime, reduced cataplexy and daytime sleepiness, and improved disrupted nighttime sleep (DNS) compared with placebo in patients aged ≥ 16 years with narcolepsy. These improvements were statistically significant and clinically meaningful. Most patients who switched from twice-nightly immediate-release sodium oxybate (hereafter referred to as sodium oxybate IR) to once-nightly sodium oxybate ER in an open-label extension/switching study preferred the once-nightly formulation. Once-nightly sodium oxybate ER was generally well tolerated, including over the longer term, with a tolerability profile consistent with that of twice-nightly sodium oxybate IR.
Plain Language Summary
Narcolepsy, a disorder of sleep-wake instability, is a lifelong condition characterized primarily by overwhelming and persistent daytime sleepiness. Other symptoms include sudden muscle weakness associated with strong emotions (cataplexy), disturbed nighttime sleep, sleep paralysis, and vivid hallucinations when falling asleep or when awakening. The standard of care for patients with narcolepsy has been sodium oxybate, which requires twice-nightly dosing. Patients take their first dose at bedtime and must then awaken 2.5–4 h later to take their second dose. A less burdensome once-nightly formulation, sodium oxybate extended-release suspension (LUMRYZ™; hereafter referred to as sodium oxybate ER), has been approved for the treatment of cataplexy or excessive daytime sleepiness in patients with narcolepsy. In a clinical trial, patients treated with once-nightly sodium oxybate ER were able to stay awake longer during the day, felt less sleepy in the daytime, had fewer cataplexy attacks, and had less disruption to nighttime sleep than placebo recipients. Sodium oxybate ER was generally well tolerated. Nausea, dizziness, bed-wetting, headache, and vomiting were the most common treatment-related adverse events. Thus, once-nightly sodium oxybate ER represents a promising new option for the treatment of narcolepsy.
What is the rationale for using sodium oxybate extended-release (ER) in narcolepsy?
Narcolepsy is a chronic neurological condition associated with dysregulation of the sleep-wake cycle [1, 2]. Excessive daytime sleepiness (EDS) is the cardinal symptom of narcolepsy [1, 3]. Additional symptoms vary between patients and include cataplexy, disrupted nighttime sleep (DNS), sleep paralysis, and hallucinations (hypnopompic and hypnagogic) [1, 3, 4]. There are two main types of narcolepsy: narcolepsy type 1 (NT1), which involves cataplexy; and narcolepsy type 2 (NT2), which does not involve cataplexy [1,2,3,4]. Narcolepsy can be debilitating when not treated adequately [3].
The American Academy of Sleep Medicine (AASM) clinical practice guidelines for the treatment of narcolepsy strongly recommend the wake-promoting agents modafinil, pitolisant, solriamfetol, and sodium oxybate for the first-line treatment of narcolepsy [3, 5], with sodium oxybate widely recognized as a standard of care [4]. However, immediate-release formulations of sodium oxybate (hereafter referred to as sodium oxybate IR) require twice-nightly dosing and have the potential to further disrupt sleep through forced awakening during the night for the second dose, which must be administered 2.5–4 h after the first (bedtime) dose [1, 6]. Twice-nightly dosing is also associated with an increased risk of accidental dosing administration errors (e.g., early dosing) resulting in adverse events (AEs) and patient harm [1].
Sodium oxybate extended-release suspension (LUMRYZ™; hereafter referred to as sodium oxybate ER) is administered once-nightly, thereby avoiding the burden of a second dose in the middle of the night [6]. Sodium oxybate ER is approved in the USA for the treatment of cataplexy or EDS in adults with narcolepsy [7]. Due to the risks of CNS depression, and abuse and misuse (Table 1), sodium oxybate ER is only available through a restricted distribution program called LUMRYZ Risk Evaluation Mitigation Strategy under which the drug is prescribed and dispensed by specially certified healthcare providers and pharmacies, and is only available to patients enrolled in the program [7]. An overview of the US prescribing information for sodium oxybate ER is provided in Table 1.
What are the pharmacological properties of sodium oxybate ER?
Sodium oxybate is the sodium salt of gamma hydroxybutyrate (GHB), which is an endogenous metabolite of the inhibitory neurotransmitter gamma aminobutyric acid (GABA) [7]. The mechanism by which sodium oxybate ER produces a therapeutic effect on narcolepsy is unknown. The therapeutic effects of sodium oxybate ER on cataplexy and EDS are thought to be mediated via the activity of GABAB at noradrenergic, dopaminergic, and thalamocortical neurons [7].
The pharmacokinetic profile of sodium oxybate ER supports once-nightly dosing, according to several phase 1 studies in healthy volunteers aged 18–65 years [8]. In a pilot study, once-nightly sodium oxybate ER 4.5 g had a lower peak plasma concentration (Cmax), a lower plasma concentration 8 h after dosing (C8h), similar area under the concentration-time curve (AUC), and comparable interperson variability versus twice-nightly sodium oxybate IR 4.5 g [8]. In two relative bioavailability studies, once-nightly sodium oxybate ER 6 g had lower C8h than twice-nightly sodium oxybate IR 6 g but equivalent AUC [8, 9]. Cmax fell below the bioequivalence threshold in one study [8] but met the criteria for bioequivalence in the other study [9]. In a dose-proportionality study, the median time to Cmax (Tmax) was 1.7 h, 1.5 h, and 2 h following oral administration of sodium oxybate ER 4.5 g, 7.5 g, and 9 g, respectively [8]. There were dose-proportional increases in Cmax and slightly more than dose-proportional increases in AUC as the total daily dose of sodium oxybate ER was increased from 4.5 g to 9 g [8].
In a food-effect study, administration of sodium oxybate ER 6 g with a high-fat meal increased Tmax by 1 h, decreased Cmax by 33%, and decreased AUC by 14% versus the fasted state [8]. In vitro studies demonstrated that warm water (50°C) had no effect on the release of sodium oxybate from the ER suspension, while hot water (90°C) or alcohol (20% or 40%) induced ‘dose-dumping’ of sodium oxybate from the ER suspension [7]. GHB has an apparent volume of distribution of 190–384 mL/kg. At concentrations of 3–300 μg/mL, < 1% of GHB is bound to plasma proteins [7].
GHB is primarily metabolized to carbon dioxide and water via the tricarboxylic acid (Krebs) cycle, with β-oxidation identified as a secondary pathway [7]. Carbon dioxide, produced from GHB metabolism, is removed from the body by expiration. No active metabolites have been identified. Less than 5% of a dose is excreted in the urine as unchanged drug within 6–8 h of dosing [7]. The apparent terminal elimination half-life of GHB is 0.66 h [9]. At a concentration of 378 μg/mL (i.e., higher than that achieved with the maximum recommended dose), sodium oxybate is not an inhibitor of CYP1A2, CYP2C9, CYP2C19, CYP2D6, CYP2E1, or CYP3A [7].
What is the efficacy of sodium oxybate ER in narcolepsy?
Once-nightly sodium oxybate ER increases sleep latency during the daytime, reduces cataplexy and daytime sleepiness, and improves DNS in patients with narcolepsy, based on the results of the 13-week, randomized, double-blind, placebo-controlled, multicenter, phase 3 REST-ON trial [10, 11]. Eligible patients were aged ≥ 16 years with a diagnosis of NT1 or NT2 according to International Classification of Sleep Disorders-3 criteria, had an Epworth Sleepiness Scale (ESS) score of > 10, and had a mean sleep latency of < 11 min on the Maintenance of Wakefulness Test (MWT) following baseline polysomnography (PSG) [10]. The MWT is a validated objective measure of the ability to stay awake for a defined time [12], with higher sleep latency values indicating less daytime sleepiness. All patients were required to demonstrate current continuing presence of EDS (assessed by patient report) for the previous 3 months [10]. Patients with NT1 were also required to demonstrate current continuing presence of cataplexy (assessed by patient report) for the previous 3 months, defined as an average of eight cataplexy attacks per week. Exclusion criteria included a diagnosis of moderate and severe sleep apnea or other sleep disorder known to cause EDS, as determined by PSG and sleep history, including any PSG results indicating an apnea hypopnea index (AHI) ≥ 15, or AHI < 15/h requiring continuous positive airway pressure; and previous use of sodium oxybate (except use of sodium oxybate ≤ 4.5 g for ≤ 2 weeks and ≥ 1 year prior to study entry) [10].
Following stratification by narcolepsy type (NT1 or NT2), patients were randomized to receive oral sodium oxybate ER (n = 111) or placebo (n = 111) once nightly [10]. The dose of sodium oxybate ER was titrated as follows: 4.5 g for 1 week, followed by 6 g during weeks 2–3, 7.5 g during weeks 4–8, and 9 g during weeks 9–13. Patients were permitted to receive stable doses of concomitant stimulants during the trial. At baseline, patients in the sodium oxybate ER and placebo groups had similar mean sleep latency on the MWT (5.0 and 4.7 min), mean Clinical Global Impression of Severity (5.1 and 5.1), mean number of weekly cataplexy attacks (18.9 and 19.8), and mean ESS scores (16.6 and 17.5). Most patients (≈ 75%) had NTI [10].
Sodium oxybate ER demonstrated statistically significant and clinically meaningful improvement relative to placebo for all three co-primary endpoints [change from baseline in mean sleep latency on the MWT, in investigator-assessed rating of ‘much improved’ or ‘very much improved’ on the Clinical Global Impression of Improvement (CGI-I), and in the mean number of weekly cataplexy attacks] (Table 2) [10]. These improvements were seen with all evaluated doses of sodium oxybate ER (i.e., 6 g, 7.5 g, and 9 g). The change from baseline in mean ESS score (secondary endpoint) also significantly favored sodium oxybate ER over placebo (Table 2).
In terms of secondary objectives, sodium oxybate ER improved both objective and subjective assessments of DNS [11]. Compared with placebo, all three doses of sodium oxybate ER significantly (p < 0.001) reduced the total number of transitions to wake or N1 (light sleep) from N1, N2, N3 (deep sleep) and rapid eye movement (REM) sleep. Sodium oxybate ER also significantly (p < 0.05) reduced the number of nocturnal arousals (defined as the number of transient arousals on nocturnal PSG according to the AASM Scoring Guidelines). Patient-reported sleep quality and refreshing nature of sleep [both assessed on a visual analog scale from 1 (‘did not sleep’/‘not refreshed’) to 100 (‘slept very well’/‘refreshed’)] were significantly (p < 0.001) improved with all three doses of sodium oxybate ER relative to placebo [11].
Post hoc sensitivity analyses of the co-primary endpoints conducted with various methods to handle missing data provided support for the efficacy of sodium oxybate ER [13]. Other post hoc analyses confirmed the efficacy of sodium oxybate ER regardless of narcolepsy subtype [14] and concomitant stimulant use [15]. In further post hoc analyses, sodium oxybate ER was associated with:
- Significant (p < 0.05 vs placebo) reductions in time spent in N1 and REM sleep and increased time spent in N3 sleep, with a significant (p < 0.05 vs placebo) increase in delta power for non-REM sleep [11];
- Improvements in sleep latency during the daytime (as measured by the MWT), with significantly (p < 0.05) more sodium oxybate ER than placebo recipients experiencing increased mean sleep latency ranging from ≥ 5 min to ≥ 20 min; these improvements were seen as early as week 3 with the 6 g dose [16];
- Significant (p < 0.05 vs placebo) improvements in daytime sleepiness, sleep quality, and refreshing nature of sleep during the first 2 weeks of treatment [17];
- Reductions in cataplexy, with significantly (p < 0.05) more sodium oxybate ER than placebo recipients experiencing ≥ 25%, ≥ 50%, and ≥ 75% reductions in the number of cataplexy attacks per week, and significantly (p < 0.05) more sodium oxybate ER than placebo recipients experiencing 100% reduction in the number of cataplexy attacks per week when receiving the 7.5 g or 9 g doses [18].
Open-label extension/switch study
Once-nightly sodium oxybate ER can be titrated to a therapeutic dose and is preferred by patients over twice-nightly sodium oxybate IR, according to results from RESTORE, an open-label, multicenter, phase 3 extension/switch study designed to assess long-term safety and tolerability [19, 20]. The trial comprised a titration period (1–2 months), a stable dosing period (up to 2 years) and a follow-up period (1 week) [20]. Patients aged ≥ 16 years with a confirmed diagnosis of NT1 or NT2 were enrolled into one of the following groups: those who completed the REST-ON trial with no current sodium oxybate use (group A); those switching from a stable dose (≥ 1 months) of twice-nightly sodium oxybate IR (group B); and oxybate-naïve patients (group C) [19, 20]. Once-nightly sodium oxybate ER was initiated at a dose of 4.5 g for patients in groups A and C [19], while patients in group B were switched to an initial sodium oxybate ER dose equivalent/closest to their previous total nightly dose of sodium oxybate IR [20]. The dose of sodium oxybate ER could be adjusted in weekly increments of 1.5 g (maximum 9 g/night) according to efficacy and tolerability [20].
At data cutoff of 1 July 2022 (n = 184), 86% of all non-switch participants (i.e., patients in groups A and C) had increased their dose of sodium oxybate ER from 4.5 g (median of three dose adjustments during initial titration) and 47% of patients were stable at 7.5 g [19].
Among the switch participants (n = 130), 59% maintained their initial dose of sodium oxybate ER, 36% increased their dose and 4% decreased their dose. Most patients (83%) who switched to sodium oxybate ER 9 g maintained this dose [19]. The majority of patients (94%) reported a preference for once-nightly sodium oxybate ER over twice-nightly sodium oxybate IR, with 88% of patients finding a second nightly dose inconvenient [20]. After switching, 63% of patients rated their narcolepsy as much better or somewhat better. Most patients were very satisfied (79%) or somewhat satisfied (16%) with once-nightly sodium oxybate ER compared with their previous narcolepsy treatments. Many patients reported improvements in their ability to get through the day without falling asleep (63%), sleep through the night (91%), get through the day without falling asleep (63%), socialize with friends/family (60%), accomplish more at work/school (59%) and perform daily activities (37%) [20].
What is the safety and tolerability of sodium oxybate ER?
Once-nightly sodium oxybate ER is generally well tolerated in patients with narcolepsy, with a safety profile consistent with that established for sodium oxybate IR [10]. A number of warnings and precautions pertain to the use of sodium oxybate ER (Table 1) [7]. There are black box warnings for both CNS depression, and abuse and misuse; other warnings and precautions include respiratory depression and sleep-disordered breathing, depression and suicidality, other behavioral or psychiatric adverse reactions, and parasomnias. Where specified, monitoring is recommended, with appropriate interventions required in some cases (Table 1) [7]. Discussion in this section focuses on data relevant to the use of sodium oxybate ER in patients with narcolepsy in the phase 3 REST-ON trial [10] and the open-label/switch study, RESTORE [21].
The safety population of REST-ON included all patients who had received ≥ 1 dose of study drug (107 sodium oxybate ER recipients and 105 placebo recipients) [10]. Treatment-related AEs (TRAEs) occurred in 57% of sodium oxybate ER recipients and 16% of placebo recipients. The most common (incidence ≥ 2%) TRAEs reported during the 6 g, 7.5 g, and 9 g dosing periods are presented in Fig. 1. ADRs led to treatment discontinuation in 16% of sodium oxybate ER recipients and 2% of placebo recipients. The most common (incidence ≥ 2%) TRAEs resulting in discontinuation of sodium oxybate ER included dizziness (5% of patients), nausea (3%), anxiety (3%), and headache (2%) [10].
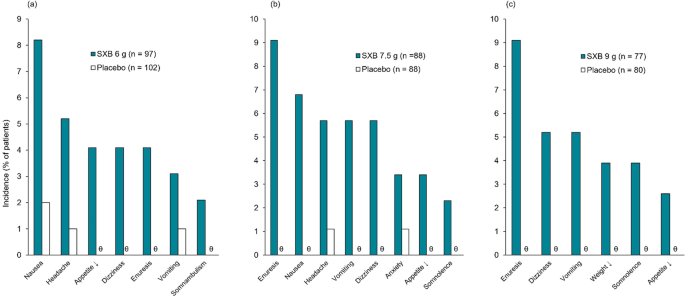
Most common treatment-related adverse events (occurring at ≥ 2% incidence and at a greater frequency with sodium oxybate extended-release than with placebo) in patients with narcolepsy during (a) weeks 2–3, (b) weeks 4–8, and (c) weeks 9–13 of the phase 3 REST-ON trial [10]. SXB sodium oxybate extended-release, ↓ decreased, ϑ indicates 0% incidence
The overall incidence of treatment-emergent AEs (TEAEs) was 78% with sodium oxybate ER and 47% with placebo [10]. Most TEAEs were of mild or moderate severity. Rates of known sodium oxybate AEs (i.e., nausea, vomiting, somnolence, dizziness, enuresis) were low across all dosing periods. Serious TEAEs occurred in 5% of sodium oxybate ER recipients and 2% of placebo recipients [10].
In a post hoc analysis, the mean change from baseline in body weight at week 13 was – 1.29 kg in patients who received sodium oxybate ER and 0.19 kg in those who received placebo [22]. The proportion of patients experiencing ≥ 5% weight loss was 18% with sodium oxybate ER versus 4% with placebo. The mean change from baseline in body mass index at week 13 was – 0.51 kg/m2 in sodium oxybate ER recipients and 0.08 kg/m2 in placebo recipients [22].
Once-nightly sodium oxybate ER is generally well tolerated over the longer term, according to an interim analysis of RESTORE (n = 180) [21]. The overall incidence of AEs was 58% and most were mild or moderate in severity. The incidence of TRAEs was 42%, and 3% of patients discontinued sodium oxybate ER due to TRAEs. The most common (incidence ≥ 3%) TRAEs were nausea (12% of patients), somnolence (7%), headache (5%), enuresis (5%), dizziness (4%), somnambulism (4%), tremor (4%), and vomiting (3%). No new safety signals were observed [21].
What is the current clinical position of sodium oxybate ER in narcolepsy?
As the first FDA-approved once-nightly sodium oxybate formulation, sodium oxybate ER represents a promising novel option for the treatment of cataplexy or EDS in patients with narcolepsy. It addresses an unmet need by avoiding the burden of a second, middle-of-the-night dose. With a pharmacokinetic profile that supports once-nightly dosing [8], sodium oxybate ER increases sleep latency during the daytime, reduces cataplexy and daytime sleepiness, and improves DNS in patients with narcolepsy [10, 11].
In the pivotal phase 3 REST-ON trial, once-nightly sodium oxybate ER demonstrated statistically significant and clinically meaningful improvements in all three co-primary endpoints versus placebo [10]. One of these endpoints, sleep latency during the daytime, was assessed on the MWT [10]. According to AASM clinical practice guidelines, the clinical significance threshold for this endpoint is ≥ 2 min (and applies to the comparison of post-treatment effects between study drug and placebo as well as a pre-/post-treatment difference) [23]. All three evaluated doses of sodium oxybate ER achieved this threshold [10].
EDS can have serious detrimental effects on quality of life, functioning, and productivity [24]. Sodium oxybate ER was associated with improvement on the ESS, which may correspond to improvement in quality of life; however, it should be noted that the trial did not directly assess quality of life [10]. Sodium oxybate ER also improved DNS, as evidenced by clinically relevant improvements in both objective measures (sleep continuity and sleep architecture) and subjective measures (patient-reported sleep quality and refreshing nature of sleep), providing additional support for the efficacy of sodium oxybate ER in patients with narcolepsy [11]. Given the beneficial effects of sodium oxybate on both ESS and DNS, future trials assessing quality of life would be valuable.
Sodium oxybate ER is generally well tolerated [10], including over the longer term [21]. The overall safety profile of once-nightly sodium oxybate ER is consistent with that of twice-nightly sodium oxybate IR [10]. Both formulations are classified as Schedule III controlled substances, carry black box warnings for CNS depression, and abuse and misuse (Table 1), and are available only through restricted distribution programs. The most common TRAEs observed with sodium oxybate ER in the phase 3 trial included nausea, dizziness, enuresis, headache, and vomiting (Fig. 1). Albeit in the absence of comparative studies, evidence suggests that the incidence of TRAEs may be lower with once-nightly sodium oxybate ER than with twice-nightly sodium oxybate IR. Possible reasons for this potential difference include the lower overall Cmax, having a single Cmax instead of two, and the slower titration scheme used in REST-ON [10]. However, results from such indirect comparisons should be interpreted with caution.
In the absence of head-to-head trials directly comparing once-nightly sodium oxybate ER with twice-nightly sodium oxybate IR, patient preference is likely to dictate which formulation is used in real-world clinical practice [10]. Once- versus twice-nightly dosing is generally less burdensome for patients [25]. The RESTORE study demonstrated that patients can be successfully switched from twice-nightly sodium oxybate IR to once-nightly sodium oxybate ER, with most patients preferring the once-nightly formulation [20]. Discrete choice experiments designed to capture preferences for specific attributes of sodium oxybate treatment have been conducted in patients with narcolepsy [26] and with clinicians who treat patients with narcolepsy [6]. These studies found that dosing frequency was the most important attribute for overall product choice, reducing anxiety/stress, likelihood of taking the medication exactly as directed (patients), and improving patient quality of life (clinicians), with the once-nightly profile of sodium oxybate ER preferred over that of twice-nightly sodium oxybate IR [6, 26]. However, it should be noted that ‘customized’ asymmetric and/or atypical dosing of twice-nightly sodium oxybate IR has been shown to improve compliance, efficacy, and tolerability compared with symmetric dosing [25]. In future studies, data from patients about their preferences for the choice of sodium oxybate should be assessed.
Change history
07 November 2024
A Correction to this paper has been published: https://doi.org/10.1007/s40267-024-01119-z
