Article Content
Abstract
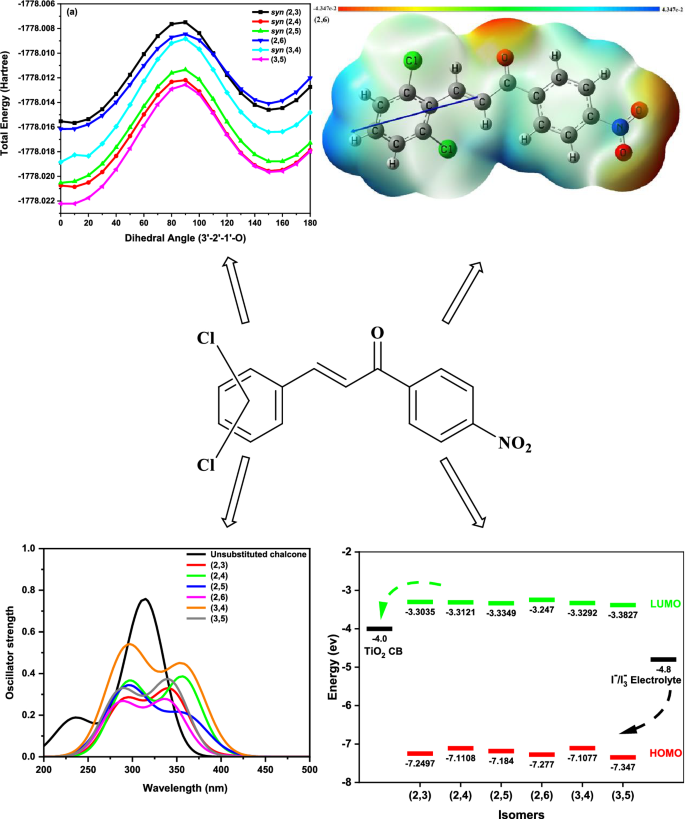
The density functional theory (DFT) with Becke’s three-parameter Lee–Yang–Parr (B3LYP) hybrid functional and the 6-311G(d,p) basis set was utilized to investigate the structural and conformational stability, the energies of the highest occupied molecular orbital (HOMO), and lowest unoccupied molecular orbital (LUMO), as well as related reactivity parameters, electrostatic potential, electronic transitions, nonlinear optical (NLO) properties, and dye-sensitized solar cell (DSSC) characteristics of six isomers of (E)-3-(i,j-dichlorophenyl)-1-(4′-nitrophenyl)prop-2-en-1-one, where i, j = 2–6 and i ≠ j. The s-cis conformers are more stable than the s-trans conformers. The syn conformers are more stable for five of the isomers than their anti-syn counterparts. The (3,5) isomer was the most stable among the isomers. All isomers demonstrated the ability to inject and recover electrons. The (2,5) isomer exhibited the highest exciton binding energy, while the (3,4) isomer showed the lowest dye regeneration driving force. The highest open-circuit voltage was observed for the (2,6) and (2,3) isomers. The (3,4) isomer had the highest light-harvesting efficiency, whereas the (2,5) isomer had the lowest. All chalcones exhibit higher first-order hyperpolarizability β values than urea, with the anti-(3,4) isomer having the highest β and the smallest (HOMO–LUMO) energy gap. TD-DFT (B3LYP/6-311G(d,p)) in the gas phase reveals that the chalcones display two UV bands. Band 1 arises from the electronic transition between the HOMO and LUMO. However, band 2 consists of electron excitation from HOMO and HOMO-2 to LUMO + 1. The chalcones investigated show promise as candidates for NLO and DSSC applications.
Graphical abstract
Explore related subjects
Discover the latest articles and news from researchers in related subjects, suggested using machine learning.
- Computational Chemistry
- Fullerenes
- Organic Chemistry
- Physical Chemistry
- Supramolecular Chemistry
- Theoretical Chemistry
Data availability
Data are provided in the manuscript and supplementary information files. No datasets were generated or analysed during the current study.
References
-
Grätzel, M.: Dye-sensitized solar cells. J. Photochem. Photobiol. C Photochem. Rev. 4, 145–153 (2003)
-
Hagfeldt, A., Boschloo, G., Sun, L., Kloo, L., Pettersson, H.: Dye-sensitized solar cells. Chem. Rev. 110, 6595–6663 (2010)
-
Kumara, N.T.R.N., Lim, A., Lim, C.M., Petra, M.I., Ekanayake, P.: Recent progress and utilization of natural pigments in dye sensitized solar cells: a review. Renew. Sustain. Energy Rev. 78, 301–317 (2017). https://doi.org/10.1016/j.rser.2017.04.075
-
Nizar, S.N.A.M., Rosli, M.M., Samsuri, S.A.M., Razak, I.A., Arshad, S.: Studies in the influence of D-π-A pyrenyl chalcone containing methoxy substitution as dye-sensitizer in DSSC. IOP Conf. Ser. Earth Environ. Sci. (2023). https://doi.org/10.1088/1755-1315/1281/1/012028
-
Conradie, J.: Effective dyes for DSSCs–Important experimental and calculated parameters. Energy Nexus. 13, 100282 (2024). https://doi.org/10.1016/j.nexus.2024.100282
-
El Mzioui, S., Bouzzine, S.M., Bourass, M., Naciri Bennani, M., Hamidi, M.: A theoretical investigation of the optoelectronic performance of some new carbazole dyes. J. Comput. Electron. 18, 951–961 (2019). https://doi.org/10.1007/s10825-019-01339-x
-
Obasuyi, A.R., Glossman-Mitnik, D., Flores-Holguín, N.: Electron injection in anthocyanidin and betalain dyes for dye-sensitized solar cells: a DFT approach. J. Comput. Electron. 18, 396–406 (2019). https://doi.org/10.1007/s10825-019-01331-5
-
Fadili, D., Bouzzine, S.M., Hamidi, M.: Effects of adding cyanovinyl moiety on the photovoltaic DSSCs phosphonic acid based cells. J. Comput. Electron. 19, 1629–1644 (2020). https://doi.org/10.1007/s10825-020-01546-x
-
Rosli, M.M., Anizaim, A.H., Nizar, S.N.A.M., Razak, I.A., Arshad, S.: Designing ferrocenyl thiophene chalcones as light harvester candidates for dye-sensitized solar cells. J. Organomet. Chem. 1022, 123415 (2024). https://doi.org/10.1016/J.JORGANCHEM.2024.123415
-
Nizar, S.N.A.M., Rosli, M.M., Samsuri, S.A.M., Razak, I.A., Arshad, S.: Involvement of halogen and polyaromatic substituents in chalcone derivatives as dye sensitizers in solar cell applications. New J. Chem. 47, 5804–5815 (2023). https://doi.org/10.1039/D2NJ05937A
-
Nassar, M.F., Abdulmalek, E., Ismail, M.F., Ahmad, S.A.A.: Enhancing the performance of pyridyl carboxamide-N,N-dimethyl amino chalcone (PCC) as a dye sensitizer for dye-sensitized solar cells (DSSCs) through the incorporation of electron donor moieties. Int. J. Electrochem. Sci. 19, 100715 (2024). https://doi.org/10.1016/j.ijoes.2024.100715
-
Pina, V., da Costa Duarte, R., Vesga-Hernández, C., dos Santos Carvalho, R., Melo, D.G., Pedrozo-Penãfiel, M.J., Barreto, A.R.J., dos Santos, A.M., Dal-Bó, A.G., Aucélio, R.Q., Cremona, M., Limberger, J.: Carboxy-substituted D-π-A arylated chalcones: Synthesis, photophysical properties and preliminary evaluation as photosensitizers for DSSCs. Opt. Mater. (Amst). 149, 115039 (2024). https://doi.org/10.1016/J.OPTMAT.2024.115039
-
Mustafa, M.N., Hussain, F., Hussain, M., Hussain, R., Ayub, K., Muhammad, S., Khan, M.U., Ehsan, M., Adnan, M.: Elucidating the potential of nonlinear optical behavior of azo dyes for advanced laser-based technologies. Adv. Theory Simul. (2025). https://doi.org/10.1002/adts.202401202
-
Boyd, R.W.: Nonlinear Optics (3rd edn). (2019)
-
Agrawal, G.P.: Nonlinear Fiber Optics (6th edn). Academic Press (2019)
-
Runowski, M., Woźny, P., Martín, I.R., Soler-Carracedo, K., Zheng, T., Hemmerich, H., Rivera-López, F., Moszczyński, J., Kulpiński, P., Feldmann, S.: Multimodal optically nonlinear nanoparticles exhibiting simultaneous higher harmonics generation and upconversion luminescence for anticounterfeiting and 8-bit optical coding. Adv. Funct. Mater. 34, 1–10 (2024). https://doi.org/10.1002/adfm.202307791
-
Marder, S.R.: Organic nonlinear optical materials: where we have been and where we are going. Chem. Commun. (2006). https://doi.org/10.1039/b512646k
-
Hadji, D., Bensafi, T.: Deeper insights on the nonlinear optical properties of O-acylated pyrazoles. J. Electron. Mater. 53, 1868–1883 (2024). https://doi.org/10.1007/s11664-024-10954-9
-
Nourai, N.E.H., Sebih, F., Hadji, D., Allal, F.Z., Dib, S., Kambouche, N., Rolland, V., Bellahouel-Benzine, S.: Nonlinear optical and antimicrobial activity of N-acyl glycine derivatives. J. Mol. Liq. 398, 124260 (2024). https://doi.org/10.1016/j.molliq.2024.124260
-
Hadji, D., Baroudi, B., Bensafi, T.: Nonlinear optical properties of azo sulfonamide derivatives. J. Mol. Model. (2024). https://doi.org/10.1007/s00894-024-05915-2
-
Hadji, D., Haddad, B., Brandán, S.A., Panja, S.K., Paolone, A., Drai, M., Villemin, D., Bresson, S., Rahmouni, M.: Synthesis, NMR, Raman, thermal and nonlinear optical properties of dicationic ionic liquids from experimental and theoretical studies. J. Mol. Struct. 1220, 128713 (2020). https://doi.org/10.1016/j.molstruc.2020.128713
-
Hadji, D.: Phosphates branching effect on the structure, linear and NLO properties of linear phosphazenes. Mater. Chem. Phys. 262, 124280 (2021). https://doi.org/10.1016/j.matchemphys.2021.124280
-
Gheribi, R., Hadji, D., Ghallab, R., Medjani, M., Benslimane, M., Trifa, C., Dénès, G., Merazig, H.: Synthesis, spectroscopic characterization, crystal structure, Hirshfeld surface analysis, linear and NLO properties of new hybrid compound based on tin fluoride oxalate and organic amine molecule (C12N2H9)2[SnF2(C2O4)2]2H2O. J. Mol. Struct. 1248, 131392 (2022). https://doi.org/10.1016/j.molstruc.2021.131392
-
Bensafi, T., Hadji, D., Yahiaoui, A., Argoub, K., Hachemaoui, A., Kenane, A., Baroudi, B., Toubal, K., Djafri, A., Benkouider, A.M.: Synthesis, characterization and DFT calculations of linear and NLO properties of novel (Z)-5-benzylidene-3-N(4-methylphenyl)-2-thioxothiazolidin-4-one. J. Sulfur Chem. 42, 645–663 (2021). https://doi.org/10.1080/17415993.2021.1951729
-
Hadji, D., Benmohammed, A., Mouchaal, Y., Djafri, A.: Synthesis and characterization of novel thiosemicarbazide for nonlinear optical applications: combined experimental and theoretical study. Rev. Roum. Chim. 68, 463–471 (2023). https://doi.org/10.33224/rrch.2023.68.9.07
-
Zyss, J., Ledoux, I.: Nonlinear optics in multipolar media: theory and experiments. Chem. Rev. 94, 77–105 (1994). https://doi.org/10.1021/cr00025a003
-
Agilandeshwari, R., Meenatchi, V., Meenakshisundaram, S.P.: Synthesis, growth, structure and characterization of chalcone crystal: a novel organic NLO material. J. Mol. Struct. 1118, 356–366 (2016). https://doi.org/10.1016/j.molstruc.2016.02.099
-
Patil, P.S., Maidur, S.R., Jahagirdar, J.R., Chia, T.S., Quah, C.K., Shkir, M.: Crystal structure, spectroscopic analyses, linear and third-order nonlinear optical properties of anthracene-based chalcone derivative for visible laser protection. Appl. Phys. B Lasers Opt. 125, 1–13 (2019). https://doi.org/10.1007/s00340-019-7275-z
-
Shruthi, C., Ravindrachary, V., Guruswamy, B., Prasad, D.J., Goveas, J., Kumara, K., Lokanath, N.K.: Molecular structure, Hirshfeld surface and density functional theoretical analysis of a NLO active chalcone derivative single crystal—a quantum chemical approach. J. Mol. Struct. 1228, 129739 (2021). https://doi.org/10.1016/j.molstruc.2020.129739
-
Arshad, M.N., Al-Dies, A.A.M., Asiri, A.M., Khalid, M., Birinji, A.S., Al-Amry, K.A., Braga, A.A.C.: Synthesis, crystal structures, spectroscopic and nonlinear optical properties of chalcone derivatives: a combined experimental and theoretical study. J. Mol. Struct. 1141, 142–156 (2017). https://doi.org/10.1016/j.molstruc.2017.03.090
-
Shkir, M., AlFaify, S., Arora, M., Ganesh, V., Abbas, H., Yahia, I.S.: A first principles study of key electronic, optical, second and third order nonlinear optical properties of 3-(4-chlorophenyl)-1-(pyridin-3-yl) prop-2-en-1-one: a novel D- π -A type chalcone derivative. J. Comput. Electron. 17, 9–20 (2018). https://doi.org/10.1007/s10825-017-1050-3
-
Nehru, J., Subramani, S., Rosli, M.M., Zainuri, D.A., Subramanian, U.M., Marappan, V., Savaridasson, J.K., Kasthuri, B., Arshad, S., Alsaee, S.K., Venkatachalam, R., Madhukar, H.: Shining light on chalcone compounds: A comprehensive exploration through optical and thermal studies. Opt. Mater. (Amst). 149, 115069 (2024). https://doi.org/10.1016/j.optmat.2024.115069
-
Nizar, S.N.A.M., Ab Rahman, S.N.F., Zaini, M.F., Anizaim, A.H., Abdul Razak, I., Arshad, S.: The photovoltaic performance of sensitizers for organic solar cells containing fluorinated chalcones with different halogen substituents. Crystals 11, 1–18 (2021)
-
Marcovicz, C., Camargo, G. dos A., Scharr, B., Sens, L., Levandowski, M.N., Rozada, T. de C., Castellen, P., Inaba, J., de Oliveira, R.N., Miné, J.C., Corrêa, S. de A.P., Allegretti, S.M., Fiorin, B.C.: Schistosomicidal evaluation of synthesized bromo and nitro chalcone derivatives. J. Mol. Struct. 1258, 132647 (2022). https://doi.org/10.1016/j.molstruc.2022.132647
-
Shainyan, B.A., Sigalov, M.V.: Hydrogen bonding-assisted transformations of cyclic chalcones: E/Z-isomerization, self-association and unusual tautomerism. Russ. Chem. Rev. 91, RCR5035 (2022). https://doi.org/10.1070/rcr5035
-
Nithya, R., Santhanamoorthi, N., Kolandaivel, P., Senthilkumar, K.: Structural and spectral properties of 4-bromo-1-naphthyl chalcones: a quantum chemical study. J. Phys. Chem. A 115, 6594–6602 (2011). https://doi.org/10.1021/jp1098393
-
Xue, Y., Gong, X.: The conformational, electronic and spectral properties of chalcones: a density functional theory study. J. Mol. Struct. Theochem. 901, 226–231 (2009). https://doi.org/10.1016/j.theochem.2009.01.034
-
Hameed, S.A.: Electronic structure of some chalcone derivatives. I. Ground state geometric parameters and charge density distributions, AM1-MO treatment. JKAU Sci. 18, 13–25 (2006)
-
Carvalho, P.S., Custodio, J.M.F., Vaz, W.F., Cirilo, C.C., Cidade, A.F., Aquino, G.L.B., Campos, D.M.B., Cravo, P., Coelho, C.J., Oliveira, S.S., Camargo, A.J., Napolitano, H.B.: Conformation analysis of a novel fluorinated chalcone. J. Mol. Model. (2017). https://doi.org/10.1007/s00894-017-3245-8
-
Zainuri, D.A., Abdullah, M., Arshad, S., Aziz, M.S.A., Krishnan, G., Bakhtiar, H., Razak, I.A.: Crystal structure, spectroscopic and third-order nonlinear optical susceptibility of linear fused ring dichloro-substituent chalcone isomers. Opt. Mater. (Amst.) 86, 32–45 (2018). https://doi.org/10.1016/j.optmat.2018.09.032
-
Gandhimathi, R., Vinitha, G., Dhanasekaran, R.: Effect of substituent position on the properties of chalcone isomer single crystals. J. Cryst. Process Technol. 3, 148–155 (2013)
-
Ashburn, B.O.: Computational analysis of a series of chlorinated chalcone derivatives. Comput. Chem. 07, 106–120 (2019). https://doi.org/10.4236/cc.2019.74008
-
Hussein, H.A., Fadhil, G.F.: Theoretical investigation of para amino-dichloro chalcone isomers, part I: A DFT structure—stability study. J. Phys. Org. Chem. 33, 1–15 (2020). https://doi.org/10.1002/poc.4073
-
Yousif, A.A., Fadhil, G.F.: DFT of para methoxy dichlorochalcone isomers. Investigation of structure, conformation, FMO, charge, and NLO properties. Chem. Data Collect. 31, 100618 (2021). https://doi.org/10.1016/j.cdc.2020.100618
-
Hadji, D., Champagne, B.: First principles investigation of the polarizability and first hyperpolarizability of anhydride derivatives. Chem. Africa. (2019). https://doi.org/10.1007/s42250-019-00060-3
-
Hadji, D., Rahmouni, A., Hammoutène, D., Zekri, O.: First theoretical study of linear and nonlinear optical properties of diphenyl ferrocenyl butene derivatives. J. Mol. Liq. 286, 110939 (2019). https://doi.org/10.1016/j.molliq.2019.110939
-
Hadji, D., Rahmouni, A.: Molecular structure, linear and nonlinear optical properties of some cyclic phosphazenes: a theoretical investigation. J. Mol. Struct. 1106, 343–351 (2016). https://doi.org/10.1016/j.molstruc.2015.10.033
-
Baroudi, B., Argoub, K., Hadji, D., Benkouider, A.M., Toubal, K., Yahiaoui, A.: Synthesis and DFT calculations of linear and nonlinear optical responses of novel 2-thioxo-3-N, (4-methylphenyl ) thiazolidine-4 one. J. Sulfur. Chem. (2020). https://doi.org/10.1080/17415993.2020.1736073
-
Cherif, F.Y., Hadji, D., Benhalima, N.: Molecular structure, linear, and nonlinear optical properties of piperazine-1,4- diium bis 2,4,6-trinitrophenolate: a theoretical investigation. Phys. Chem. Res. 11, 33–48 (2023). https://doi.org/10.22036/pcr.2022.330752.2035
-
Gaussian 09, R.D. 0., M. J. Frisch, G. W. Trucks, H. B. Schlegel, G.E.S., M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B.M., G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H.P.H., A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M.H., M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T.N., Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery, J., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E.B., K. N. Kudin, V. N. Staroverov, T. Keith, R. Kobayashi, J.N., K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J.T., M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J.B.C., V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R.E.S., O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J.W.O., R. L. Martin, K. Morokuma, V. G. Zakrzewski, G.A.V., P. Salvador, J. J. Dannenberg, S. Dapprich, A.D.D., O. Farkas, J. B. Foresman, J. V. Ortiz, J.C., and D. J. Fox, Gaussian, Inc., Wallingford CT, 2013.: Gaussian 09, (2013)
-
Dennington, R., Keith, T.A., Millam, J.M.: GaussView 6.0. 16, (2016)
-
Origin(Pro), Version 2019b. OriginLab Corporation, Northampton, MA, USA
-
ChemDraw Professional, Version 23.1.1.3, revvity signals software, inc
-
Merouane, A., Mostefai, A., Hadji, D., Rahmouni, A., Bouchekara, M.: Theoretical insights into the static chemical reactivity and NLO properties of some conjugated carbonyl compounds : case. Monatshefte für Chemie – Chem. Mon. (2020). https://doi.org/10.1007/s00706-020-02653-y
-
Abu-awwad, F., Politzer, P.: Variation of parameters in becke-3 hybrid exchange-correlation functional variation of parameters in becke-3 hybrid exchange-correlation functional. J. Comput. Chem. 21, 227–238 (2000). https://doi.org/10.1002/(SICI)1096-987X(200002)21
-
Hantosh, L.A., Sami, S.A., Fadhil, G.F.: Structure-stability and energy storage capacity of para acetyl-dichloro chalcone and chromen isomers : a density functional theory investigation. Orient. J. Chem. 40, 1774–1785 (2024)
-
Politzer, P., Murray, J.S.: Molecular electrostatic potentials: Significance and applications. Chem. React. Confin. Syst. Theory Model Appl. (2021). https://doi.org/10.1002/9781119683353.ch7
-
de Paula, R.L.G., Carvalho, F.B., D’Oliveira, G.D.C., Duarte, V.S., Santin, L.G., Pérez, C.N., Oliveira, S.S., Napolitano, H.B.: Synthesis, crystal structure and molecular modeling of a novel chalcone-quinolone hybrid. J. Mol. Struct. 1217, 128355 (2020). https://doi.org/10.1016/j.molstruc.2020.128355
-
Sirleto, L., Righini, G.C.: An introduction to nonlinear integrated photonics devices: nonlinear effects and materials. Micromachines 14, 1–25 (2023). https://doi.org/10.3390/mi14030604
-
Naik, V.S., Patil, P.S., Gummagol, N.B., Wong, Q.A., Quah, C.K., Jayanna, H.S.: Crystal structure, linear and nonlinear optical properties of three thiophenyl chalcone derivatives: a combined experimental and computational study. Opt. Mater. (Amst.) 110, 110462 (2020). https://doi.org/10.1016/j.optmat.2020.110462
-
Chen, R., Xu, K., Li, Q., Ma, J., Wang, T., Zhang, Z., Mu, X., Xuan, F., Cao, L., Teng, B.: Synthesis and characterization of a new chalcone-based nonlinear optical crystal: BBC. J. Mol. Struct. 1293, 136320 (2023). https://doi.org/10.1016/j.molstruc.2023.136320
-
Aneesa, V.M., Safna Hussan, K.P., Lekshmi, S., Babu, T.D., Muraleedharan, K.: Analysis of non-linear optical properties of phytochemical photosensitizers in cancer photodynamic therapy by quantum computational. Results Chem. 8, 101580 (2024). https://doi.org/10.1016/j.rechem.2024.101580
-
Mishra, A.K., Tewari, S.P.: Density functional theory calculations of spectral, NLO, reactivity, NBO properties and docking study of Vincosamide-N-Oxide active against lung cancer cell lines H1299. SN Appl. Sci. 2, 1–13 (2020). https://doi.org/10.1007/s42452-020-2842-9
-
Chidan Kumar, C.S., Quah, C.K., Balachandran, V., Fun, H.K., Asiri, A.M., Chandraju, S., Karabacak, M.: Synthesis, single crystal structure, spectroscopic characterization and molecular properties of (2E)-3-(2,6-dichlorophenyl)-1-(3,4-dimethoxyphenyl)prop-2-en-1-one. J. Mol. Struct. (2016). https://doi.org/10.1016/j.molstruc.2016.02.089
-
Shinde, S.S., Sreenath, M.C., Chitrambalam, S., Joe, H., Sekar, N.: Non-linear optical properties of disperse blue 354 and disperse blue183 by DFT and Z-Scan technique. Polycycl. Aromat. Compd. (2019). https://doi.org/10.1080/10406638.2019.1686404
-
Chaitanya, K., Ju, X.H., Heron, B.M., Gabbutt, C.D.: Vibrational spectra and static vibrational contribution to first hyperpolarizability of naphthopyrans – a combined experimental and DFT study. Vib. Spectrosc. 69, 65–83 (2013). https://doi.org/10.1016/j.vibspec.2013.09.010
-
Niu, R., Wang, Y., Wu, X., Chen, S., Zhang, X., Song, Y.: D-π-A-type pyrene derivatives with different push-pull properties: broadband absorption response and transient dynamic analysis. J. Phys. Chem. C 124, 5345–5352 (2020). https://doi.org/10.1021/acs.jpcc.9b11667
-
Teo, K.Y., Tiong, M.H., Wee, H.Y., Jasin, N., Liu, Z.Q., Shiu, M.Y., Tang, J.Y., Tsai, J.K., Rahamathullah, R., Khairul, W.M., Tay, M.G.: The influence of the push-pull effect and a π-conjugated system in conversion efficiency of bis-chalcone compounds in a dye sensitized solar cell. J. Mol. Struct. 1143, 42–48 (2017). https://doi.org/10.1016/j.molstruc.2017.04.059
-
Abbo, H.S., Hung Lai, C., Titinchi, S.J.J.: Substituent and solvent effects on UV-visible absorption spectra of chalcones derivatives: experimental and computational studies. Spectrochim. Acta. Part A Mol. Biomol. Spectrosc. 303, 123180 (2023). https://doi.org/10.1016/j.saa.2023.123180
-
Szmant, H.H., Basso, A.J.: The absorption spectra of substituted chalcones. J. Am. Chem. Soc. 74, 4397–4400 (1952). https://doi.org/10.1021/ja01137a047
-
Xue, Y., Mou, J., Liu, Y., Gong, X., Yang, Y., An, L.: An ab initio simulation of the UV/Visible spectra of substituted chalcones. Cent. Eur. J. Chem. 8, 928–936 (2010). https://doi.org/10.1002/qua
-
Lipkowitz, K.B., Boyd, D.B., Larter, R., Cundari, T.R.: Reviews in Computational Chemistry Volume 20. In: Wiley, pp. 484 (2004)
-
Miar, M., Shiroudi, A., Pourshamsian, K., Oliaey, A.R., Hatamjafari, F.: Theoretical investigations on the HOMO–LUMO gap and global reactivity descriptor studies, natural bond orbital, and nucleus-independent chemical shifts analyses of 3-phenylbenzo[d]thiazole-2(3H)-imine and its para-substituted derivatives: Solvent and subs. J. Chem. Res. 45, 147–158 (2021). https://doi.org/10.1177/1747519820932091
-
Masnabadi, N., Thalji, M.R., Alhasan, H.S., Mahmoodi, Z., Soldatov, A.V., Ali, G.A.M.: Structural, electronic, reactivity, and conformational features of 2,5,5-Trimethyl-1,3,2-diheterophosphinane-2-sulfide, and its derivatives: DFT, MEP, and NBO calculations. Molecules 27, 4011 (2022)
-
Lavanya, M., Mahalakshmi, C.M.: DFT calculations on molecular structure, homo lumo study reactivity descriptors of triazine derivative. Int. Res. J. Educ. Technol. 5, 49–52 (2023)
-
Asiri, A.M., Ersanlı, C.C., Şahin, O., Arshad, M.N., Hameed, S.A.: Molecular structure, spectroscopic and quantum chemical studies of 1’,3’,3’-trimethylspiro[benzo[f]chromene-3,2’-indoline. J. Mol. Struct. (2016). https://doi.org/10.1016/j.molstruc.2016.01.086
-
Hussein, H.A., Fadhil, G.F.: Theoretical investigation of para amino-dichloro chalcone isomers. Part II: a DFT structure-stability study of the FMO and NLO properties. ACS Omega 8, 4937–4953 (2023). https://doi.org/10.1021/acsomega.2c07148
-
Hantosh, L.A.: Structure-stability, energy storage capacity, frontier molecular orbitals, and dye sensitized solar energy study of the para acetyl- dichloro chalcone and chromene isomers : a DFT investigation (2025)
-
Odey, J.O., Louis, H., Agwupuye, J.A., Moshood, Y.L., Bisong, E.A., Brown, O.I.: Experimental and theoretical studies of the electrochemical properties of mono azo dyes derived from 2-nitroso-1- naphthol, 1-nitroso-2-naphthol, and C.I disperse yellow 56 commercial dye in dye-sensitized solar cell. J. Mol. Struct. 1241, 130615 (2021). https://doi.org/10.1016/j.molstruc.2021.130615
-
Fei, E.T.L., Biswas, J., Datta, B., Kumar, D.: Computational studies of diindole-based molecules for organic bulk heterojunction solar devices using DFT and TD-DFT calculations. Struct. Chem. 32, 1973–1984 (2021). https://doi.org/10.1007/s11224-021-01777-z
-
Babu, N.S.: DFT and TD-DFT studies of new triphenylamine-based (D-A-D) donor materials for high-efficiency organic solar cells. Mater. Adv. (2022). https://doi.org/10.1039/d2ma00048b
-
Benmohammed, A., Hadji, D., Mouchaal, Y., Djafri, A.: Synthesis and characterization of novel chalcone with good nonlinear optical properties. Chem. Africa. (2024). https://doi.org/10.1007/s42250-024-01143-6
Acknowledgements
Hazhi Hasan Hussein would like to thank the University of Duhok for the PhD scholarship that enabled him to complete this article.
Funding
This research has received no funding.
Ethics declarations
Competing interest
The authors declare no competing interests.
Ethical approval
This research is not applicable to any human or animal studies.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (DOCX 7255 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Reprints and permissions
About this article
Cite this article
Hussein, H.H., Fadhil, G.F. Investigation of structural and conformational stability, electronic transition, NLO, FMO, and DSSC parameters of trans-dichloro-nitro chalcone isomers: a DFT insight. J Comput Electron 24, 140 (2025). https://doi.org/10.1007/s10825-025-02378-3
- Received
- Accepted
- Publishe d
- DOI https://doi.org/10.1007/s10825-025-02378-3
Keywords
- Chalcone isomer stability
- DFT
- ESP
- Electronic transition
- NLO
- DSSC
