Article Content
Abstract
Ovarian folliculogenesis is a complex biological process critical for female fertility, intricately regulated by various signaling pathways, among which the Mitogen-Activated Protein Kinase (MAPK) signaling pathway plays a pivotal role. This review provides a comprehensive overview of the physiological functions of MAPK signaling in key stages of folliculogenesis, including primordial follicle formation and activation, dominant follicle selection, cumulus-oocyte complex (COC) expansion, ovulation, and luteinization. The orchestrating roles of MAPK on steroidogenesis and ovarian cell death are also delineated, highlighting its essential contributions to normal reproductive function. Furthermore, we explore the implications of dysregulated MAPK signaling in ovarian aging, primary ovarian insufficiency (POI), polycystic ovary syndrome (PCOS), and ovarian hyperstimulation syndrome (OHSS). By elucidating the multifaceted roles of MAPK signaling in ovarian biology, this review aims to enhance our understanding of folliculogenesis and its associated pathologies, paving the way for future research and therapeutic interventions targeting MAPK pathways in reproductive health.
Clinical trial number Not applicable.
Introduction
Follicular development is a complex biological process that occurs sequentially under the precise regulation of hormones, ovarian regulatory factors, and intercellular interactions [1]. During early embryonic development, primordial germ cells are established and migrate to the future gonadal region, proliferating to form nests of cells that ultimately develop into primordial follicles [2]. The migration and homing of these primordial germ cells within the genital ridge require the coordinated regulation of various factors, including oocyte-derived peptide growth factors, growth differentiation factor 9 (GDF-9), and bone morphogenetic proteins [3]. Most follicles remain dormant, with only a few being activated to join the pool of growing follicles. The transition from primordial follicles to primary follicles, and subsequently to secondary follicles, culminates in pre-ovulatory follicles. This progression involves oocyte growth, proliferation of granulosa cells (GCs), and the formation of theca cells (TCs) [4]. Follicular steroidogenesis depends on pituitary gonadotropins, including follicle-stimulating hormone (FSH) and luteinizing hormone (LH). During each reproductive cycle, only a subset of follicles develops into dominant follicles, which are more sensitive to LH and ultimately undergo ovulation [5]. Ovulation represents the final stage of follicular development, involving meiosis resumption in the oocyte, expansion of cumulus cells, rupture of the follicle, and the release of the cumulus-oocyte complex(COC) containing a fertilizable oocyte [6]. Post-ovulation, the remaining GCs and TCs undergo terminal differentiation to form the corpus luteum (CL), which secretes progesterone to sustain pregnancy [7]. Most follicles will eventually undergo atresia, a natural regression phenomenon during follicular development [8]. Concurrently, various cell death pathways permeate the process from primordial germ cell migration to follicular atresia, contributing to the establishment of normal ovarian reserve, selection of dominant follicles, and degeneration of the corpus luteum [9].
In addition to the widely studied intercellular interactions among ovarian cells, various signaling pathways coordinate the entire process from primordial germ cell migration to follicular atresia, as summarized by researchers. The PI3K/AKT/FOXO3 signaling pathway regulates the activation of primordial follicles through FOXO3 [10]. The nucleocytoplasmic shuttling of FOXO3 plays a critical role in follicle activation, with its phosphorylation status influencing its localization and transcriptional activity within the nucleus [11]. The WNT signaling pathway regulates various cellular processes, including proliferation, differentiation, and apoptosis, via both β-catenin-dependent and independent mechanisms. Wnt2 and Wnt4 are particularly vital in follicular development, as they regulate GCs proliferation through β-catenin [12]. The Notch signaling pathway not only participates in ovarian angiogenesis but also interacts with FOXO3, potentially modulating follicular development through its influence on FOXO3 [13]. In the ovary, Hedgehog signaling operates through communication between GCs and TCs, with excessive activation leading to ovulatory dysfunction and abnormal ovarian vascular development [14]. The evolutionarily conserved intracellular signaling cascade known as the MAPK (mitogen-activated protein kinase) pathway has increasingly come into focus, playing a significant role in both the physiological and pathological processes within the ovary. This review will delve into the intricate roles of the MAPK signaling pathway in ovarian function and dysfunction.
The MAPK signaling pathway is a highly conserved signal transduction cascade that is widely present from yeast to humans [15]. It plays a critical role in regulating various biological activities, including cell growth, differentiation, apoptosis, and reproduction [16, 17]. The upstream regulatory components of the MAPK pathway are diverse and complex, encompassing Rho family GTPases, the TNF receptor family, IL-1 receptors, and TLR family receptors. Key upstream activators include receptor tyrosine kinases (RTKs), G protein-coupled receptors (GPCRs), and ion channels, which activate Ras(rat sarcoma) kinases through adaptor proteins such as Grb2 and SOS, initiating a cascade of phosphorylation events that sequentially activate MAPKKKs, MAPKKs, and ultimately MAPKs [18].
MAPKKKs are Ser/Thr protein kinases, usually activated by phosphorylation or interaction with small GTP-binding proteins from the Ras or Rho family. The activation of MAPKKKs leads to the phosphorylation and activation of MAPKKs, which, in turn, activate MAPKs through dual phosphorylation of specific threonine and tyrosine residues on the MAPK [19,20,21]. The MAPK family primarily consists of three subfamilies: ERK (Extracellular Signal-Regulated Kinases), JNK (c-Jun N-terminal Kinases), and p38 protein kinases [20]. The activation of MAPKs requires dual phosphorylation on a specific tripeptide motif (Thr-X-Tyr) located within the kinase activation loop (T-loop) [22]. MAPKs can regulate various processes through both transcription-dependent and transcription-independent mechanisms. The integration of signals and spatiotemporal regulation are influenced by multiple factors, including the cellular localization of MAPKs and their substrates, interactions with scaffold proteins, and the dephosphorylation activity of MAPK phosphatases [23]. Activated MAPKs further phosphorylate downstream effectors, which may include membrane proteins, cytoplasmic proteins, and nuclear proteins [24]. Notably, MAPKs can directly phosphorylate a variety of transcription factors, such as AP-1, p53, c-Fos, and c-Jun, thereby modulating their activity and stability, which in turn affects the expression of downstream genes [25].
The activities of ERK1 and ERK2 are regulated by upstream kinases and phosphatases, which are particularly important in female fertility [26]. The ERK pathway is the most extensively studied MAPK pathway, involving the Raf-MEK-ERK cascade [27]. Members of the Raf family, such as Raf-1, B-Raf, and A-Raf, activate MAPK/ERK kinases 1 and 2 (MEK1/2), which act as dual-specificity protein kinases (DUSPs) to phosphorylate specific sites on ERK1/2 [28]. Cyclic AMP (cAMP) can activate B-Raf through Rap1 while inhibiting Raf-1, thereby influencing the activation of the MAPK pathway [28]. The ERK pathway promotes cell proliferation by activating transcription factors such as Elk-1 and c-Fos, and it also regulates the transition from the G1 to S phase by affecting the expression of Cyclin D1 and the activity of Cdk2 [29].
The JNK pathway is associated with various physiological processes and involves multiple kinases, such as MEKK, MLK, and ASK. These kinases phosphorylate and activate MAPKKs (such as MEK4/7), which subsequently phosphorylate and activate JNK, thus initiating or modulating intracellular signaling processes [30]. JNK binds to and phosphorylates c-Jun, enhancing its transcriptional activity [31]; c-Jun is a component of the AP-1 transcription complex that regulates the expression of numerous cytokine genes [32].
The p38 pathway responds to cellular stress and is involved in the transmission of apoptosis and survival signals, featuring at least four distinct p38 MAPK isoforms (p38α, β, γ, and δ) [33,34,35,36]. The p38 MAPK pathway can be activated by various cellular stress conditions, including UV irradiation, heat shock, and certain mitogens [17, 37, 38]. MAPKKs (MEK3/6) activate p38 MAPKs, forming a cascading response. Notably, the activation of p38α involves a non-traditional mechanism, where TAB1 (TGF-β activated kinase 1 binding protein 1) acts as an adaptor or scaffold protein in the activation process, rather than through the phosphorylation by MKKs [39].
In addition to the three traditional MAPKs, several atypical MAPKs have been identified in recent years, including ERK3/4, ERK7/8, and NLK (Nemo-like kinase) [40]. The activation mechanisms of these atypical MAPKs differ from those of conventional MAPKs; for instance, ERK3/4 and NLK lack the Thr-X-Tyr motif typically required for phosphorylation [40]. Targeted knockout of the ERK3 gene has been shown to result in intrauterine growth restriction and early neonatal mortality [41]. Moreover, ERK7 and ERK8 may play critical roles in regulating cell proliferation and responses to estrogen [42] and glucocorticoids [43]. In Caenorhabditis elegans, NLK activates the TCF/LEF transcription factor POP1 through TAK-1, phosphorylating it and inhibiting its activity, which promotes the establishment of the anterior-posterior axis in a Wnt-dependent manner [44]. However, the roles of atypical MAPKs in follicular development remain to be characterized.
Previous reviews have highlighted the multifaceted roles of the MAPK pathway in male reproductive function [27], emphasizing that the regulation of ERK1/2 activity is crucial for the production and secretion of pituitary gonadotropins [45]. Currently, our understanding of the role of MAPKs in follicular development is still incomplete. This review aims to contribute to future research designs, enabling better management of infertility associated with abnormalities in female follicular development.
MAPK in the physiology of folliculogenesis
MAPK in primordial follicle formation
Primordial follicle formation is a critical process in ovarian development, serving as the foundation for oocyte maturation and female fertility. It involves the assembly and activation of oocytes encased within GCs, transitioning from a quiescent state to a more active phase. This intricate process is governed by a range of signaling pathways, with the MAPK pathway emerging as a key regulator. Analysis using RNA-seq and ATAC-seq has revealed significant changes in the expression of genes associated with the MAPK signaling pathway during the early ovarian development of geese, particularly during the pre-hatching period when oocyte loss occurs. These alterations suggest that the MAPK signaling pathway may play a crucial role in regulating the formation and assembly of primordial follicles [46]. Furthermore, exposure to DEHP[di(2-ethylhexyl)phthalate] has been shown to result in changes in the miRNA expression profile in mouse ovaries. Some of the differentially expressed miRNAs may regulate ovarian development by targeting mRNAs involved in the MAPK signaling pathway. Gene Ontology (GO) enrichment analysis and pathway enrichment analysis revealed that the target genes of the differentially expressed miRNAs are implicated in the MAPK signaling pathway. This indicates that DEHP may influence the expression of miRNAs, thereby affecting the regulation of MAPK signaling-related genes and subsequently impacting the formation of primordial follicles [47].
Early studies have indicated that the MAPK pathway plays a crucial role in the formation of primordial follicles, primarily functioning as a downstream effector of the KIT(Kinase Insert Domain Receptor) signaling pathway [48]. When KIT ligand (KITL) is added to the culture medium containing mouse fetal or neonatal ovarian tissues, an increase in phosphorylated MAPK (p-MAPK) levels can be observed [48, 49]. Additionally, research has highlighted that KIT can regulate the formation of primordial follicles by activating the PI3K and MAPK3/1 signaling pathways [49]. Gene network analysis in neonatal rats has revealed that ERK1/2 is significantly expressed in several gene modules, correlating with other key genes involved in the assembly of primordial follicles, which suggests that ERK1/2 may regulate follicle assembly by influencing the expression of other genes. Enrichment analysis of gene modules further demonstrated that genes associated with the MAPK signaling pathway are enriched in specific modules, confirming the role of the MAPK signaling pathway in primordial follicle assembly [50]. In adult mice, FSH promotes the assembly of primordial follicles through the FSH receptor 3 (FSHR3), a process that involves the MAPK/ERK pathway [51, 52]. In a study conducted on chickens, FSH and stem cell factor (SCF) were found to enhance ovarian cell proliferation and inhibit apoptosis by activating the MAPK pathway, potentially affecting the assembly of primordial follicles through the regulation of cell adhesion molecule expression [53].
c-JUN may participate in the transcriptional regulation during the follicle assembly process [50]. Recently, single-cell level studies have identified two novel genes, ANXA7 (annexin A7) and GTF2F1 (general transcription factor IIF subunit 1), that promote primordial follicle formation [54]; notably, GTF2F1 has been found to interact with JNK in yeast [55]. JNK is specifically localized in oocytes, and its activity increases as germ cell cysts undergo degeneration. The use of the JNK-specific inhibitor SP600125 or knockdown of JNK expression via Lenti-JNK-shRNAs significantly inhibits both the breakdown of germ cell cysts and the formation of primordial follicles [56]. Inhibition of JNK signaling results in abnormal accumulation of E-cadherin between oocytes, suggesting that JNK signaling may regulate the degradation of cysts by modulating E-cadherin expression or function [56]. Furthermore, treatment with Bisphenol S(BPS) leads to abnormal rupture of primordial germ cell cysts and influences subsequent ovarian differentiation while diminishing oocyte quality. The application of JNK inhibitors such as SP600125 can partially counteract the effects of BPS on the rupture of primordial germ cell cysts and the assembly of primordial follicles, further underscoring the important role of JNK signaling in follicle formation [57].
Currently, the role of p38 in the formation of primordial follicles remains underexplored. The activation of the FGF23-FGFR1-p38 MAPK signaling pathway is crucial for maintaining oocyte survival during primordial follicle formation in mice and preventing premature apoptosis [58]. Therefore, further experimental investigations are needed to elucidate the specific functions of the p38 MAPK branch in the formation of primordial follicles and oocyte survival.
Primordial follicle activation
The role of the MAPK signaling pathway in the activation of primordial follicles is primarily demonstrated through its interaction with the oncoprotein ErbB2. In rat models, c-erbB2 participates in the regulation of the initiation of primordial follicle growth via the MAPK signaling pathway. Transfection of ovarian tissue with c-erbB2 siRNA results in a significant downregulation of both c-erbB2 mRNA and ErbB2 protein levels, accompanied by a marked decrease in the expression of MAPK proteins [59]. Importantly, the changes in p-ERK1/2 protein levels in rat ovaries exhibit a significant positive correlation with c-erbB2 mRNA expression, indicating a close relationship between the activation state of ERK1/2 and c-erbB2 expression levels [60]. Epidermal growth factor (EGF) has been shown to promote the development of primordial follicles into secondary follicles through the activation of the MAPK and protein kinase C pathways [61,62,63]. A plausible mechanism involves the binding of EGF to its receptor EGFR, which may lead to the dimerization of EGFR and ErbB2, promoting the phosphorylation of ErbB2 and activating receptor protein tyrosine kinases (RPTK). The activated RPTK subsequently interacts with proteins containing SH2 domains via its phosphorylated tyrosine residues, activating Ras, which in turn activates Raf, ultimately leading to the activation of ERK1/2. The activated ERK1/2 modulates the activity of transcription factors, thereby influencing gene expression and promoting the initiation of primordial follicle growth [60]. Treatment with PD98059 significantly inhibits the transition of primordial follicles to primary follicles in rat ovarian tissue [62]. In a mouse model where the PTEN(phosphatase and tensin homolog deleted on chromosome ten) inhibitor bpV(HOpic) facilitates primordial follicle activation, U0126 was found to inhibit the phosphorylation of ERK1/2 and follicle activation, further confirming the role of ERK1/2 in follicle activation [64]. Additionally, pentachloronitrobenzene has been shown to enhance the expression of steroidogenic acute regulatory protein(StAR) and progesterone production through the activation of the ERK1/2 signaling pathway in rat ovaries, accelerating the transition of primordial follicles to growing follicles [65]. Furthermore, in chicken ovaries, FSH has been reported to activate the MAPK pathway to promote primordial follicle activation, which likely involves multiple factors within the MAPK pathway, such as RAS, RAF, MEK, and ERK [53]. Leukemia inhibitory factor(LIF) and basic fibroblast growth factor (bFGF) significantly enhance the expression of ERK and p-ERK in chicken ovaries, indicating the involvement of the ERK signaling pathway in bFGF-mediated primordial follicle activation [66, 67].
ERK signaling also plays a crucial role in the activation of primordial follicles through the mTOR-KIT pathway. ERK1/2 promotes primordial follicle activation by activating mTORC1 [68]. In murine models, the activation of mTORC1 triggers the recruitment of primordial follicles, with ERK1/2 regulating the activity of mTORC1 during this process [69]. Vasoactive Intestinal Peptide (VIP) treatment has been shown to increase the phosphorylation levels of mTOR and its downstream protein Ribosomal protein S6 (RPS6) in rat ovaries, which aligns with the activation of ERK1/2 [70]. Furthermore, mTORC1 activation leads to the activation of S6K1 and rpS6, resulting in increased expression of KITL (KIT Ligand), thereby facilitating the activation of primordial follicles [64, 69]. Upon binding of KITL to its receptor KIT, the PI3K-Akt signaling pathway is activated in oocytes, promoting the nuclear export of Foxo3a and thus enhancing the activation of primordial follicles [71]. The use of U0126 leads to a decrease in Akt phosphorylation levels and a reduction in Foxo3 nuclear export, indicating that ERK1/2 activity is essential for the activation of the PI3K-Akt signaling pathway [64]. Treatment with U0126 in neonatal mouse ovaries resulted in a significant reduction in the number of activated follicles. U0126 treatment also decreased the phosphorylation levels of Tsc2, S6K1, and rpS6, as well as the expression of KITL, suggesting that ERK1/2 promotes primordial follicle activation via the mTORC1-KITL signaling pathway [64]. Additionally, in Esr2-/- rat ovaries, there was an observed increase in ERK pathway activation, which correlates with enhanced primordial follicle activation [72]. The loss of ESR2 leads to the upregulation of upstream factors such as KITLG, KIT, and IGF1, which can activate RTK, further stimulating the AKT and mTOR pathways [72]. Comparative analysis of embryonic ovarian samples from 17.5 dpc and 15.5 dpc mice revealed a significant upregulation of estrogen receptors and p-ERK1/2 expression, further suggesting that estrogen and the ERK1/2 signaling pathway may be involved in the formation and activation of primordial follicles [73].
In sheep ovaries, the use of JNK inhibitors, such as SP600125 and JNK inhibitor VIII, significantly impedes the activation of primordial follicles. During the process of follicle activation, Foxo3a is translocated from the oocyte nucleus to the cytoplasm, where it is subsequently degraded following phosphorylation [74]. Inhibition of the JNK pathway can prevent the translocation of Foxo3a from the nucleus, resulting in elevated levels of Foxo3a remaining in the oocyte nucleus, which in turn inhibits the activation of primordial follicles [75]. This suggests that JNK signaling plays a critical role in facilitating the activation of primordial follicles by promoting the nuclear export and degradation of Foxo3a, thereby allowing the process of follicle activation to proceed (Fig. 1).
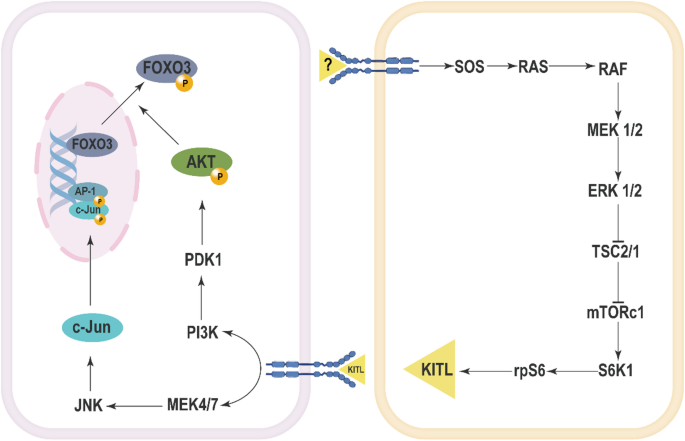
The Mechanisms of activation of primordial follicles involving MAPK pathway
Recent studies have highlighted the significant roles of MAPK pathway-related non-coding RNAs (ncRNAs) in the activation of primordial follicles. One research team employed a sub-pathway-based approach to analyze the relationship between long non-coding RNAs (lncRNAs) and biological processes during primordial follicle activation. They identified lncRNAs associated with the MAPK signaling pathway, such as FGD5-AS1, which influences primordial follicle activation by regulating genes within the MAPK pathway, including CRKL, HSPA8, MAPK8, MAX, and NFATC3 [76]. Additionally, miR-144-3p plays a crucial role in regulating the p38 MAPK pathway by targeting MAP3K9, significantly impacting both primordial follicle activation and GCs apoptosis [77]. Furthermore, circRNA-miRNA-mRNA network analyses have revealed that circ-009346 and circ-017054 are associated with the MAPK signaling pathway and may participate in primordial follicle activation by influencing this pathway [73]. These findings underscore the intricate regulatory roles of non-coding RNAs in the MAPK signaling pathway and their potential implications in the activation of primordial follicles.
Although the role of MAPK in primordial follicle activation has been initially characterized (Fig. 1), several shortcomings remain in the current studies. Many investigations are primarily conducted in animal models, which may not fully replicate human ovarian physiology. Future research should focus on integrative approaches, including human ovarian tissue studies and advanced molecular techniques, to unravel the complex regulatory networks involved in primordial follicle activation.
MAPK in steroidogenesis
Current research presents a range of divergent findings regarding the role of the MAPK signaling pathway in steroid hormone production (Table 1) [78,79,80,81,82,83,84,85,86,87]. Activation of the MAPK pathway influences the expression of cAMP response element-binding protein (CREB), Steroidogenic Factor-1 (SF-1), and DAX-1, subsequently affecting the expression of StAR and steroidogenesis [88, 89].
Adversely, studies have reported that the activation of ERK1/2 can inhibit estradiol synthesis and/or promote progesterone synthesis [90,91,92], which may arise from variations in receptor-effector coupling due to differences in cell types, cell line origins, characteristics, and developmental stages. The ERK1/2 signaling pathway has been shown to suppress CYP19A1 expression and estradiol secretion in GCs across various mammalian species. Activated ERK1/2 regulates steroidogenesis in bovine by inhibiting estradiol production while promoting progesterone synthesis, mediated by the downregulation of FOXL2 and upregulation of SOX9 [93]. Inhibition of the MAPK pathway within GCs has been associated with increased secretion of 17β-estradiol and decreased secretion of progesterone [90, 92, 94,95,96,97]. Specific ERK phosphorylation inhibitors have been shown to upregulate StAR protein expression in human and rat GCs, a regulation that can be blocked by specific PKA inhibitors [91]. In undifferentiated mouse GCs stimulated by FSH, activation of ERK1/2 resulted in increased expression of aromatase, StAR, and CYP11A1 [98]. In porcine COCs treated with U0126, an increase in 17β-estradiol secretion and a reduction in progesterone secretion were observed. This treatment also upregulated Cyp19A1 gene expression while downregulating 3β-HSD gene expression in porcine COCs[96]. Furthermore, activation of ERK1/2 is critical for the induction of StAR mRNA expression and progesterone production in human cumulus GCs by rosiglitazone, with U0126 preventing the rosiglitazone-induced increases in both StAR mRNA expression and progesterone production [97].
The JNK pathway plays a significant role in HB-EGF-induced estrogen secretion and apoptosis in KGN cells, primarily mediated through the cAMP-PKA-JNK/ERK-Ca2+-FOXO1 pathway [99]. Conversely, other studies indicate that activation of the JNK pathway can impair GCs function by inhibiting steroid hormone synthesis [100].Additionally, in bovine GCs, TNF-α has been shown to inhibit steroidogenesis through the JNK pathway [101].
Studies have shown that FSH can rapidly activate p38 MAPK, and this activation is dependent on protein kinase A (PKA). In rat GCs, treatment with the p38 MAPK inhibitor SB203580 effectively inhibits FSH-induced estradiol production but does not significantly affect progesterone production or cAMP levels [102].
MAPK in dominant follicle selection
Follicular deviation and the selection of a dominant follicle are critical processes in ovarian physiology, significantly influencing female reproductive outcomes. Follicular bias refers to the preferential development of certain follicles over others, while dominant follicle selection determines which follicle will mature and ovulate. These mechanisms are essential for optimizing oocyte quality and ensuring successful fertilization. Recent studies have highlighted the pivotal role of various signaling pathways in follicular development, with the MAPK pathway emerging as a key regulator. The MAPK pathway mediates crucial cellular processes, including proliferation, differentiation, and apoptosis, thereby influencing follicular dynamics and hormonal responses.
The MAPK signaling pathway, particularly ERK1/2, plays a crucial regulatory role in dominant follicles of cattle, influencing steroid hormone biosynthesis and follicular development [103]. In the early stages of follicular waves in cattle, the levels of phosphorylated MAPK3/1 are significantly elevated in the GCs of follicles that are destined to become dominant [104]. Consistently, phosphorylated Akt and Erk1/2 have also been detected in samples from dominant follicles in sheep, whereas these phosphorylated forms were absent in subordinate follicle samples [105]. On the second day of the follicular wave, the GCs of the future dominant follicle (DF), identified as the largest follicle on that day, exhibited significantly higher levels of p-MAPK3/1 compared to the second largest follicle, suggesting that MAPK3/1 signaling is involved in the formation of the dominant follicle prior to the follicular deviation [106]. After the occurrence of follicular deviation, there were no significant differences in the abundance of p-MAPK3/1 between the GCs of the DF and subordinate follicles (SF), implying that MAPK3/1 may not be essential for the sustained growth of the DF [106]. Furthermore, p-MAPK3/1 levels in the SF did not change significantly by day four, despite undergoing apoptosis, indicating that MAPK3/1 may not participate in the apoptotic process of SF [106]. The TEK signaling pathway, through the activation of the Ras/ERK/MYC signaling cascade, promotes various cellular processes including growth, differentiation, migration, adhesion, proliferation, and survival in dominant follicles [107, 108]. In dominant follicles, the overexpression of ANGPT1 and TEK enhances the activation of the Ras-ERK signaling pathway, which is closely associated with cellular growth and proliferation [107]. Grb14, identified as a negative regulator of the MAPK pathway, is highly expressed in SFs, potentially inhibiting MAPK pathway activation, while its low expression in dominant follicles correlates with enhanced MAPK activity, thus facilitating follicle growth and deviation in cattle [109]. Additionally, TRIB2 expression in dominant follicles is associated with the processes of follicular growth and maturation [110, 111]. TRIB2 is abundantly expressed in the GCs of dominant follicles and is linked to the activation of the MAPK pathway. Inhibition of TRIB2 results in reduced phosphorylation levels of ERK1/2 and p38MAPK, alongside increased expression of the cell proliferation marker PCNA, indicating that TRIB2 may negatively regulate GCs proliferation through the MAPK pathway [110]. In summary, the MAPK pathway and its associated factors play a significant role in the processes of follicular deviation or selection, particularly during the early stages of dominant follicle formation. However, once follicular deviation has occurred, the role of MAPK3/1 in maintaining the growth of the DF or the regression of the SF appears to be less significant [106]. In studies utilizing an FSH-induced co-dominant follicle model to assess the effects of FSH on the GCs of the second largest follicles in cattle, despite higher mRNA levels of MAPK1/3 in SF, the abundance of phosphorylated MAPK3/1 proteins in the GCs of control group SF was found to be lower compared to the FSH-treated co-DF2 [112].
During the follicular selection process in cattle, MAPKKK5 (Ask1) may play a critical role in regulating the survival and death of GCs, thereby influencing which follicles are designated as dominant. The activity of the IGF-1 receptor is likely crucial for the maintenance of dominant follicles, as it has the capacity to inhibit apoptosis signals mediated by MAPKKK5 [113]. In yaks, MAPKAP1 exhibits the lowest expression levels in follicles with diameters ranging from 4 to 6 mm, suggesting that MAPKAP1 may be associated with follicular development, oocyte maturation, and the selection of dominant follicles [114]. Moreover, MAPK8IP1 mRNA is expressed in the GCs of dominant follicles and its expression levels increase following hCG treatment in ovulatory follicles. MAPK8IP1 is known to be associated with the JNK signaling pathway and is involved in regulating processes such as apoptosis, cell motility, adhesion, and morphology. In the context of dominant follicles, MAPK8IP1 may participate in preventing apoptosis or modulating cellular activity through its effects on cytoskeletal components [115].
MAPK in COC expansion
The expansion of the COCs, which is a critical process that occurs during the final stages of oocyte maturation and facilitates successful fertilization and subsequent embryonic development, involves the proliferation and differentiation of cumulus cells that secrete hyaluronic acid (HA) and other extracellular matrix components to form a gelatinous matrix surrounding the oocyte. This matrix, which not only protects the oocyte but also enhances its developmental competence, promotes communication between cumulus cells and the oocyte primarily through gap junctions and paracrine signaling. The process, which is tightly regulated by hormonal cues—most notably luteinizing hormone (LH)—triggering the activation of signaling pathways, including the MAPK pathway (Fig. 2), thereby leading to cumulus cell expansion. As this coordinated expansion occurs, it results in the detachment of the COC from the ovarian follicle, ultimately preparing it for ovulation.
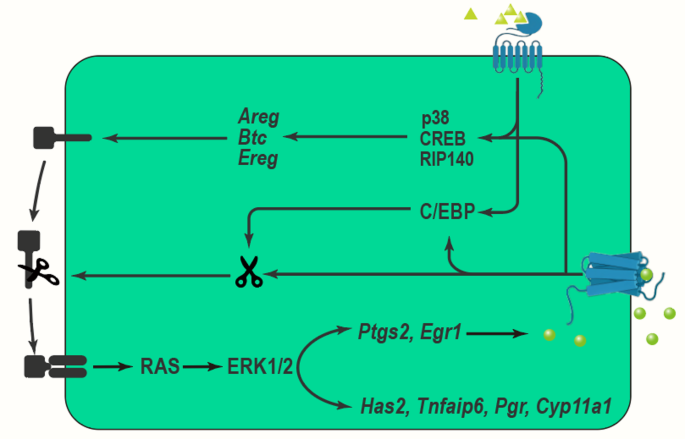
The Mechanisms of COC expansion involving MAPK pathway
LH exerts its effects by binding to its G-coupled receptors, subsequently activating adenylate cyclase and increasing cAMP production. This cascade results in the activation of protein kinase A (PKA) and downstream ERK1/2 signaling pathways [116,117,118], which play crucial roles in the regulation of EGF-like factor expression [119,120,121]. LH-induced activation of ERK1/2 leads GCs and cumulus cells to release members of the epidermal growth factor (EGF) family, such as amphiregulin (AREG), epiregulin (EREG), and betacellulin (BTC) [122]. These EGF-like factors then activate the epidermal growth factor receptor (EGFR), which further triggers downstream signaling pathways, including ERK1/2, in both GCs and cumulus cells. This signaling is essential for promoting oocyte meiotic maturation and cumulus expansion [119, 120, 123,124,125]. Research indicates that while a transient exposure to LH is sufficient to induce oocyte maturation and cumulus expansion, the sustained activity of EGFR is critical for maintaining the prolonged phosphorylation state of ERK1/2 [126, 127].
RIP140 has been identified as a crucial transcriptional coactivator in the LH signaling pathway. It enhances the transcription of the Areg gene by forming complexes with CREB and c-Jun family members. RIP140’s activity at the CRE element on the Areg gene promoter is responsive to forskolin, a compound that activates adenylate cyclase and mimics LH signaling, suggesting its involvement in the activation of the MAPK pathway [128]. EGF-induced phosphorylation of CREB is completely blocked by the ERK1/2 inhibitor U0126, indicating that EGF activates CREB through the ERK1/2 pathway to facilitate cumulus expansion [129]. During the in vitro maturation of porcine oocytes, treatment with EGF/GT1b significantly increases the expression of both total and phosphorylated ERK1/2 proteins [130].
Stimulation by FSH and LH activates the PKA- and p38 MAPK-C/EBP pathways, leading to increased expression of TACE/ADAM17. This enzyme releases the EGF domains of EGF-like factors, subsequently activating the EGFR-ERK1/2-C/EBP pathway, whose activation results in elevated expression levels of TACE/ADAM17 and enhanced Ptgs2 mRNA expression in GCs and cumulus cells during ovulation [127, 131,132,133,134]. TACE/ADAM17, a protease, cleaves EGF-like factors to release their EGF domains, thereby activating the EGFR-ERK1/2 pathway [132, 133, 135, 136]. Using selective inhibitors of TACE/ADAM17 (such as TAPI-2) or siRNA to reduce TACE/ADAM17 activity suppresses luteinization, cumulus expansion, and oocyte maturation in porcine GCs [132]. PGE2 acts on cumulus cells through its receptor PGER2 to maintain cAMP levels, thereby supporting the expression of EGF-like factors and TACE/ADAM17 in a positive feedback loop that is crucial for sustained activation of the ERK1/2 pathway [137,138,139]. The initial expression of EGF-like factors and TACE/ADAM17 is induced in a gonadotropin-dependent manner, whereas their sustained expression relies on the PGE2-PGER2 pathway [139, 140].
FSH promotes HA synthesis and accumulation in porcine cumulus cells by activating ERK1/2 [141]. Activation of EGFR leads to ERK1/2 activation, which in turn regulates the expression of genes involved in HA synthesis and accumulation [142,143,144]. When COCs are exposed simultaneously to FSH and IGF1, phosphorylation of AKT and ERK1/2 is significantly increased [145]. IGF1 enhances FSH-induced HA synthesis and retention in the cumulus cell matrix through PI3K/AKT- and ERK1/2-dependent mechanisms [145]. The use of the ERK1/2 inhibitor U0126 markedly reduces HA synthesis and retention stimulated by the combined action of FSH and IGF1 [145], further confirming the critical role of ERK1/2 in cumulus cell expansion [141].
Disruption of ERK1/2 signaling in GCs and cumulus cells underscores the critical role of ERK1/2 activation in mediating LH action during the ovulatory stage of follicular development in vivo [120, 143]. Pharmacological or gene-targeting approaches to downregulate the ERK1/2 pathway result in significant inhibition of COC expansion and oocyte maturation [124]. Inhibition of ERK1/2 significantly reduces the phosphorylation of HDAC2, prevents the deacetylation of H3K27Ac, and disrupts cumulus expansion in mice [146]. Furthermore, inhibition of ERK1/2 activity markedly decreases LH-induced expression of genes associated with COC expansion, such as Has2, Tnfaip6, and Ptx3, which play crucial roles in this process [147]. Treatment with TSA significantly blocks ERK1/2 activation in mouse cumulus cells, indicating that TSA inhibits cumulus expansion by suppressing ERK1/2 activity [148]. Inhibition of the ERK1/2 signaling pathway leads to altered expression of LH-induced genes, including those associated with COC expansion, such as EGR1 and ADAMTS1 [149]. Knockout of ADAMTS1 in mice results in subfertility due to abnormal extracellular matrix remodeling of the follicle wall [149,150,151].
p38 MAPK differentially regulates the induction of EGF-like factors in cumulus and GCs, with p38 inhibition blocking COC expansion and the expression of specific genes, including Ptgs2, which controls PGE2 synthesis. The p38-specific inhibitor SB203580 also inhibits COC expansion and the expression of genes induced by FSH, PGE2, or IL-6 [152,153,154,155]. Additionally, SB203580 significantly reduces PGE2-induced expression of AREG, EREG, HAS2, PTGS2, and TNFAIP6, highlighting the importance of p38 in the gene expression associated with cumulus expansion [156]. Treatment with SB203580 also prevents COC expansion in pigs, indicating the crucial role of p38 MAPK in this process [126, 157]. Studies on p38 gene knockout mice reveal that p38 specifically alters gene expression profiles in GCs and COCs. Specific deletion of p38 in GCs leads to upregulation of Areg and Ereg in GCs but impairs their expression in COCs. This compensatory upregulation in GCs may help maintain the fertility of p38 GC-/- mice. Despite impaired responsiveness of COCs to FSH, forskolin, or PGE2 under in vitro conditions, co-culture with AREG allows normal COC expansion, suggesting that GC-derived AREG and EREG can bypass the requirement for p38 signaling in PGE2-induced Areg/Ereg expression in COCs, thus maintaining COC expansion [158], which implies that while p38 is not absolutely essential for pre-ovulatory follicle development or ovulation and luteinization-related events in vivo, it does impact gene expression. Moreover, treatment with traditional Chinese medicine components BSTJ-II-D and BSTJ-III-D increases serum FSH levels and the expression of p-p38/p38 and p-ERK1/2/ERK1/2 proteins in mice, suggesting that these compounds may promote cumulus cell expansion by regulating FSH levels and activating the MAPK signaling pathway [159].
