Article Content
Introduction
Gynecologic malignancies account for approximately 11% of new cancer diagnoses in the United States, with ovarian, cervical, and endometrial cancers being the most commonly reported both nationally and globally. Among these, ovarian cancer (OC) remains one of the deadliest, ranking high in cancer-related mortality worldwide [1,2,3]. Developing alternative therapeutic approaches is a priority, particularly for persistent, advanced-stage, and recurrent gynecologic cancers. Standard chemotherapy for these malignancies typically involves a combination of carboplatin, which modifies DNA, and paclitaxel, which targets microtubules [4]. While these agents have led to significant treatment success, resistance to chemotherapy inevitably develops, necessitating new therapeutic strategies. One promising approach focuses on proteins involved in apoptosis regulation and drug resistance, as chemotherapy primarily induces cell death through apoptotic pathways. Ral-interacting/binding protein (RLIP/RalBP1) has gained attention in this context due to its role in protecting cancer cells from apoptosis-related stress. Research has shown that RLIP is highly expressed in multiple tumors, including OC. Studies indicate that suppressing or eliminating RLIP function can induce cell death in various solid and hematologic cancers, particularly in OC cells [5,6,7].
In a B16 mouse melanoma model, RLIP depletion demonstrated cancer cell selectivity, as normal cells remained unaffected during antisense treatment, which led to complete tumor regression [5]. These findings suggest that inhibiting RLIP, in combination with conventional chemotherapy, could enhance treatment efficacy in human cancers. In fact, previous studies have demonstrated that combination therapies involving RLIP inhibition can prolong survival, highlighting the potential synergy between these treatment components [7]. To investigate this, we conducted studies using human OC cell cultures and nude mouse xenograft models. Our research explored whether reducing RLIP expression through antisense mechanisms could promote tumor shrinkage in xenograft mice undergoing OC treatment. Additionally, we examined the impact of combining carboplatin with RLIP antibodies, hypothesizing that this approach could enhance therapeutic outcomes.
Materials and methods
Materials
Carboplatin was obtained from Selleck Chemicals LLC (Houston, TX, Sylvanfield Drive). RLIP antibodies were procured from Santa Cruz Biotechnology (Columbus, OH), while β-actin antibodies were sourced from Bioss Antibodies (Woburn, MA). Cambrex Bio-Science (Walkersville, MD) supplied Keratinocyte medium, serum-free medium (K-SFM), and the EGM-2 bullet kit medium. Essential cell culture reagents, including Dulbecco’s Modified Eagle Medium (DMEM), RPMI-1640, phosphate-buffered saline (PBS), fetal bovine serum (FBS), penicillin/streptomycin (P/S) solution, trypan blue, and trypsin-EDTA, were acquired from GIBCO-BRL Inc. (Grand Island, NY). Bio-Rad Laboratories (Hercules, CA) provide SDS-PAGE components. Sigma-Aldrich (St. Louis, MO) supplied the MTT reagent (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide), while the TUNEL Fluorescence Apoptosis Detection kit was obtained from Promega (Madison, WI). Anti-mouse peroxidase-conjugated secondary antibodies and HRP-conjugated anti-rabbit antibodies were purchased from Cell Signaling Technologies (Danvers, MA). The cell invasion assay kit was sourced from Cell Biolabs, Inc. (San Diego, CA), and the cell counting kit-8 (CCK-8), used for viability assays, was supplied by Dojindo Molecular Technologies (Rockville, MD). The Universal Mycoplasma Detection Kit was obtained from the American Type Culture Collection (ATCC; Manassas, VA).
Cell lines and culture conditions
A collection of human OC cell lines, including A2780, HEYA8, CAOV3, OVCAR3, OVCAR4, OVCAR8, MDAH2774, SW626, and SKOV3, were generously provided by Dr. Edward Wang from the Department of Medical Oncology at City of Hope National Medical Center and Beckman Research Institute (Duarte, CA). Human umbilical vein endothelial cells (HUVECs) and human aorta vascular smooth muscle cells (HAVSMCs) were obtained through the courtesy of Dr. Fiemu Nwariaku at the University of Texas Southwestern Medical Center (Dallas, TX). The OC cell lines were cultured at 37 °C with 5% CO2 in a humidified incubator using RPMI-1640 medium. HUVECs were maintained in EGM-2 bullet kit medium, while HAVSMCs were cultured in DMEM. All culture media were supplemented with 10% heat-inactivated fetal bovine serum (FBS), 1% penicillin/streptomycin (P/S), 10 mmol/L HEPES, 2 mmol/L L-glutamine, 1 mmol/L sodium pyruvate, 1.5 g/L sodium bicarbonate, and 4.5 g/L glucose. To ensure cell line authenticity, short tandem repeat (STR) profiling was performed at the City of Hope Integrative Genomics Core (Duarte, CA). Additionally, mycoplasma contamination screening was conducted every three months to maintain culture integrity.
RLIP antisense Preparation
A segment of RLIP amino acids from 171 to 185 (nucleotides 510 to 555, starting from an AUG codon in the open reading) was chosen for phosphorothioate DNA synthesis. The addition of sulfur atoms in place of phosphate oxygen molecules makes modified DNA more resistant to nucleases although it extends its electrical charge to prevent membrane penetration. The selected DNA underwent BLAST testing of the NCBI database against EST libraries to establish that it targeted only the RLIP gene. The research utilized phosphorothioate DNA (desalted) originally acquired from Biosynthesis Inc. (Lewisville, TX). A non-target specific 21-nucleotide DNA sequence containing phosphorothioate modifications went through BLAST verification to check its inhibitory role on RLIP cDNA prior to use. The targeted cDNA sequence (AAGAAAAAGCCAATTCAGGAGCC) ranged from nucleotide position 507 to position 527 and had its corresponding complementary phosphorothioate DNA sequence as GGCTCCTGAATTGGCTTTTTC. The prototype sequence for the scrambled control reaction was CATCGAAATCGTTGCAGTTAC. Maxfect transfection reagent (MoleculA) was employed for transfection, while gene silencing evaluation was conducted 24–48 h post-transfection.
Expression of RLIP
The single 1968 bp human RLIP cDNA sequence including the open reading frame was first isolated from a λgt11 human bone marrow library by using anti-DnpSG ATPase antibodies for immunological screening. The cDNA was inserted into the pET-30a (+) (Novagen) prokaryotic expression vector to create pET30-RLIP without any extra sequences. The plasmid was then transferred into E. coli BL21 (DE3) cells for protein expression using 0.4 mM IPTG at 30 °C [8].
Purification of Recombinant RLIP
DNP-SG affinity resin was applied as described in [8] to purify RLIP from 0.4 mM IPTG-induced E. coli BL21 (DE3)-RLIP pellet fractions to near homogeneity. The purification method proceeded at 4 °C under all steps with the use of metal-contaminant free water and buffers that contained Chelex-100 resin treatment. Filter-sterilized buffers were freshly prepared prior to the study. The lysing buffer utilized 1% (w/v) polidocanol (C12E9 non-ionic detergent), 10 mM Tris-HCl (pH 7.4), 1.4 mM β-mercaptoethanol, 100 µM EDTA, 50 µM BHT, and 100 µM PMSF together with 10 mM Tris-HCl (pH 7.4) and 1.4 mM β-mercaptoethanol. RLIP was extracted from the membrane fraction by applying it to DNP-SG Sepharose 4B affinity resin after gentle shaking for 16 h, followed by sonication and centrifugation at 27,000 × g for 30 min. The established protocols enabled the removal of contaminant proteins followed by RLIP elution with 10 mM ATP, 10 mM MgCl₂ and 0.025% C12E9 in lysis buffer. An Amicon Centriprep concentrator was used to reduce the solution volume, which was then dialyzed against progressively changing lysis buffer solutions containing 1% DE-52 and 0.01% SDS, followed by a lysis buffer mixed with 1% (w/v) Chelex resin. The process included two exchanges through 2 L of buffer during each 24 h step.
Isolation of crude membrane fractions from mouse tissues
The temperature needed for all procedures remained at 4 °C except when explicitly stated otherwise. A 22-gauge syringe was used to infuse PBS into tissues prior to homogenization as part of blood contamination reduction measures. A solution containing 10 mM Tris-HCl buffer (pH 7.4) with 1.4 mM β-mercaptoethanol was utilized to prepare 20% (w/v) homogenates from mouse liver, lung, kidney, brain, heart, and spleen tissues. The supernatant fluid was collected from the 8,000 × g centrifugation run after the 45 min period using vigilant techniques to dispose of the pellet. Membranes were obtained from the ultracentrifugation of the supernatant at 100,000 × g for 60 min. The pellet was solubilized using a lysis buffer containing 10 mM Tris-HCl (pH 7.4), 0.1 mM PMSF, 0.05 mM BHT, 0.1 mM EDTA, and 1.4 mM β-mercaptoethanol. The extract also included 0.5% polidocanol as an addition. Three rounds of sonication were performed at 50 W for 30 s each, with intermittent shaking at 4 °C for 4 h, which increased membrane solubilization. A second round of ultracentrifugation at 100,000 × g for 2 h at 4 °C was executed following the incubation step. The supernatant solution obtained after discarding the pellet was retained.
ELISA-based quantification of RLIP depletion
ELISA-based assays were used to assess RLIP depletion in cellular and tissue samples. While RLIP purification remains the most precise method for analysis, ELISA procedures on crude homogenates provide a reliable alternative. A carbonate-bicarbonate buffer solution (pH 9.6) containing 0.01% Tween 20 and 1% BSA was used to coat 96-well plates, followed by incubation at 37 °C for 2 h. Each well was then loaded with 100 µl of protein solution (0.1–4 µg per well) prepared in the same buffer and incubated at 37 °C for another 2 h. After washing out the reaction solution, blocking was performed by adding 100 µL of 10% goat serum at room temperature for 30 min. A 100 µg/mL solution of RLIP primary antibody was applied to each well and incubated at 4 °C for 2 h following PBS-Tween washes. Subsequently, a secondary antibody solution (1:1000 dilution) in PBS-Tween with 1% BSA was added (100 µl per well) after multiple washes and incubated at room temperature for 30 min. After additional PBS-Tween washes, 100 µl of HRP solution was added to each well and incubated on a shaker for 10–20 min until detection was complete. Absorbance was measured at 405 nm using an ELISA plate reader, with a standard reference curve constructed using purified recombinant RLIP (rec-RLIP) protein.
MTT cell viability assay
The MTT assay was used to evaluate the RLIP depletion effects on cell survival throughout all OC cell lines (OVCAR8, MDAH2774, SW626, CAOV3, SKOV3, OVCAR3, OVCAR4, A2780, and HEYA8) and normal cell lines (HAVSMC, HLBEC, and HUVEC). Transfection with RLIP antisense (0–10 µg/mL) was carried out using Maxfect Transfection Reagent (MoleculA, Inc.) according to the product specifications in plates containing 5,000 cells per well. The testing solution containing 10 µL of 5 mg/mL MTT solution was added to each well following 48 h, and plates were kept at 37 °C for another 4 h. A solution of 100 µL DMSO was used to dissolve the formazan crystals, which had been produced following PBS washing. Samples were measured at 570 nm wavelength using a Microplate XMark spectrophotometer from Bio-Rad, Hercules, CA. The survival percentage of cells was determined by establishing the Absorbance Treatment/Absorbance Control ratio and converting it to a percentage based on background-subtracted absorbance values. The readings used for calculations were derived from absorbance measurements with background subtraction. The results show mean value calculations with standard deviation bars from three replicate experiments containing four technical duplicate readings per condition (n = 12). Western blot examination confirmed the success of RLIP reduction as a result of antisense treatment.
The cell density of OC cell lines (SW626, OVCAR4, OVCAR8) and normal cell lines (HUVEC, HAVSMC) was measured using trypan blue exclusion and counting of viable cells with a hemocytometer. A total of 5,000 cells were seeded in 96-well microtiter plates before exposure to 40 µg/ml RLIP antibodies for 24 h. To assess IC50 values, 40 µL aliquots of carboplatin at concentrations ranging from 1 µM to 100 µM were added to eight replicate wells, and the concentration needed to reduce formazan formation by 50% was calculated. After adding 10 µL of MTT solution (5 mg/ml), the plates were incubated at 37 °C for 2 h. The supernatant was discarded, and the plates were washed with PBS. Formazan crystals were dissolved with 100 µL of DMSO, followed by gentle shaking at room temperature for 2 h. Absorbance values were measured using a Microplate XMark™ spectrophotometer (Bio-Rad, Hercules, CA). Cell viability was calculated by multiplying the ratio of treatment absorbance to control absorbance by 100, after background correction. Data points represent the mean and standard deviation from three separate experiments, each with four treatment replicates per condition.
Colony formation assay
The evaluation of RLIP antisense treatment effects on colony formation involved exposing 0.1 × 10⁶ cells to 10 µg/ml final concentrations of either scrambled antisense or RLIP antisense for 48 h through 500 µL treatments. The cells received 50 µL or 100 µL aliquots from 4 mL culture medium for plating into 60 mm petri dishes after treatment. The cells were incubated for seven days before fixing and staining adherent colonies with 0.5% methylene blue solution for 30 min. The Innotech Alpha Imager HP system measured the quantities of developed colonies [9].
Immunohistochemical analysis
To assess RLIP expression in-vivo, C57Bl/6 mice were split into two groups, each receiving a single intraperitoneal (i.p.) dose of either scrambled antisense or RLIP antisense (0.2 mg in 0.2 ml PBS, intraperitoneally). After 48 h, tissue samples were fixed in formalin for 24 h, embedded in paraffin, and sliced into 5 μm thick sections, which were mounted on Poly-L-lysine-coated slides. The sections went through deparaffinization and rehydration, starting with xylene and followed by graded ethanol solutions. H&E staining was done first, followed by RLIP immunostaining using a universal avidin/biotin complex detection kit (Vector Laboratories). Immunoreactivity showed up as dark brown staining, while areas with only background staining were considered non-immunoreactive. Images were captured at 40× magnification using an Olympus DP72 microscope, and antigen staining intensity was analyzed with DP2-BSW digital imaging software.
TUNEL assays for assessing the effects of RLIP antibodies and antisense on apoptosis
The Promega Apoptosis Detection Kit allowed evaluation of apoptotic activity through TUNEL staining of DNA fragments according to manufacturer specifications. One hundred thousand cells were placed on cover slips in 12-well plates, cultured for approximately 12 h, and then received treatment with pre-immune serum and RLIP antibodies (40 µg/ml final conc.) or scrambled or RLIP antisense (10 µg/ml final conc.), carboplatin (20 µM), and their combinations for 24 h. Fluorescence microscopy analysis of apoptotic cells took place after cells received PBS washes. The apoptotic green fluorescence showed detection at 520 nm, while red propidium iodide fluorescence appeared above 620 nm. Cell images were taken at 400× magnification using a Zeiss LSM 510 META laser scanning microscope through laser detection of green fluorescence signal and cell size reduction indicators of apoptosis.
Cell invasion assay
The OVCAR8 OC cells were tested for migration in vitro using a Transwell Boyden chamber under RLIP antisense treatment conditions. OVCAR8 cells at 0.5 × 10⁶ cells per well received either scrambled or RLIP antisense (10 µg/ml) treatment through serum-free medium during 24 h of incubation in the upper chamber of 24-well Transwell plates. The lower chamber contained DMEM, which was supplemented with 10% FBS. The cells received incubation under humid conditions at 37 °C while maintaining 5% CO₂ levels. The stationary cells from the upper chamber were removed at predetermined time points to fix the migratory cells underneath with 4% paraformaldehyde for 15 min, before permeabilizing them with 0.2% Triton X-100, staining them with 0.2% crystal violet, and observing them under a light microscope at 200× magnification. Five random selected fields were used for image capture. After washing, the inserts were moved to a separate well, followed by the extraction of crystal violet stain using 200 µl of extraction buffer. The absorbance at 560 nm was measured to quantify invasive cells, comparing the RLIP antisense-treated group to the scrambled antisense control. This assay was repeated in triplicate.
In-Vivo xenograft studies
A mouse model was used to evaluate the potential of therapeutic agents for anticancer treatment through in vivo studies. The research involved Hsd: Athymic nude nu/nu female mice, sourced from The Jackson Laboratory (Bar Harbor, ME). All experimental animal procedures obtained institutional review board approval from the Institutional Animal Care and Use Committee (IACUC). Forty, 8-week-old female mice received specific treatments including pre-immune serum, scrambled antisense, RLIP antibodies, RLIP antisense, and carboplatin treatments (Figs. 5 and 6). The mice were divided into eight groups with five mice in each group. The mice received 1 × 10⁶ OVCAR8 OC cells diluted in 100 µl of PBS through subcutaneous injection following their one-week acclimation process. Tumor observation started on day 18 post-injection, after the tumors became visible at approximately 40 mm² surface size. The study used RLIP antibodies at 4 mg/kg body weight (b.w.) and RLIP antisense at 4 mg/kg b.w. combined with carboplatin at 20 mg/kg b.w., and RLIP antisense + carboplatin, RLIP antibodies + carboplatin, and the combination of RLIP antisense + RLIP antibodies + carboplatin. The control groups received pre-immune serum or scrambled antisense (4 mg/kg b.w.) as their treatment. During eight weeks of treatment all solutions were given through i.p. injections once a week for the duration of the study while maintaining regular measurements of tumor volume changes. The study measured tumor dimensions along with body weight using calipers at weekly intervals throughout days 1 through 56. Photographs captured tumor progression on the designated dates. Mice were euthanized by cervical dislocation followed by CO₂ asphyxiation at the experiment’s end point. Tumor images were recorded then. Statistical tests for tumor weight differences were performed through an unpaired Student’s t-test across groups.
Statistical analysis
The results are presented as mean ± standard deviation (SD) from at least three independent experiments unless stated otherwise. Body weight data and tumor size measurements are depicted as scatter plots. Statistical significance was determined using a threshold of p ≤ 0.05, applying either paired t-tests or two-way ANOVA. All statistical analyses were two-sided, and error bars represent the standard error derived from triplicate sample measurements.
Ethics statement
The research investigation excluded studies on human participants. Animal research operated under guidelines set by the City of Hope Animal Care and Ethics Committee through IACUC protocol #22,011. The study protocol ensured humane euthanasia for mice exhibiting signs of distress, pain, or tumor-related complications.
Results
RLIP-targeting agents and carboplatin reduce viability of OC cells
Research investigations showed that the combination treatment with RLIP-targeting agents and carboplatin leads to decreased viability of OC cells. To study RLIP suppression effects on OC cells, we utilized RLIP antibodies as inhibitors and RLIP antisense to decrease protein levels. The growth of OC cells decreased only when they received a 48 h treatment of RLIP antibodies at 40 µg/ml yet maintained normal rates under preimmune serum (Fig. 1, panel A). These results suggest that inhibiting RLIP does not affect the growth of HUVEC and HAVSMC cells, which are non-cancerous in nature. An MTT assay evaluated the antiproliferative actions of carboplatin which represents a standard treatment for OC. When OC cells received carboplatin treatment at concentrations between 0 and 100 µM for 48 h, their viability decreased in a dose-responsive manner. The exposure to carboplatin did not cause cellular growth repression in non-malignant HUVEC and HAVSMC cultures. The data illustrates IC50 values and cell survival percentages for carboplatin treatment in the presence of RLIP antibodies is presented (Fig. 1, panel A).
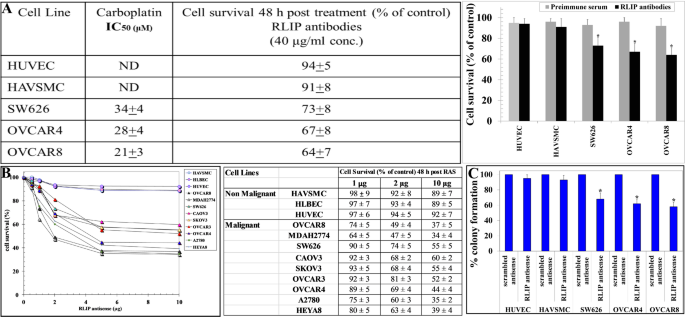
Impact of RLIP antibodies, RLIP antisense, and carboplatin on normal and OC cells. The experimental cells derived from OC received 48 h carboplatin exposure at different concentration ranges from 0 to 100 µM. The administration of RLIP antibodies led to specifically diminished survival rates among OC cells but not normal cells. The multiwell plate contained 40 micrograms per milliliter of RLIP antibodies. The data reveals the IC50 value (µM) of carboplatin together with the numbered survival rate of cells that received RLIP antibody exposure. ND indicates no detection. The analysis of Student’s t-test established statistical significance at *p < 0.03 compared to matching control groups (panel A). The process of RLIP antisense treatment for malignant cells required Maxfect transfection reagent (MoleculA Inc.) administration in accordance with the manufacturer guidelines. The MTT cytotoxicity assay used for survival assessments of cells occurred 48 h after treatment. We investigated the effects of RLIP antisense concentrations from 0 to 10 µg/ml in a 96-well plate. The measurements are displayed as the mean ± SD from three independent experiments along with four replicates (n = 12) throughout (panel B). The evaluation of colony-forming efficiency in OC and non-malignant cells used methylene blue staining on antisense-treated cells followed by counting through Innotech Alpha Imager HP. Panel C shows statistically significant results (*p < 0.04) against each control group
To facilitate antisense-mediated downregulation of RLIP, specific sequences, particularly nt508–528, were analyzed to identify unique regions optimal for RLIP targeting. We evaluated RLIP cytotoxicity on OC cells by using RLIP phosphorothioate antisense for RLIP reduction and examined the impacts using scrambled antisense as the reference group. The cell growth of OC cells decreased significantly when antisense tagged with RLIP sequences was used for 48 h, compared to the cells treated with scrambled antisense or left untreated. The decrement of RLIP levels did not influence the growth pattern of non-cancerous HUVEC, HLBEC, and HAVSMC cells. RLIP antisense treatment reduced OC cell survival in a dose-dependent manner, with the results shown in Fig. 1, panel B. Further research into how RLIP depletion affects both OC cell survival and proliferation was conducted through clonogenic potential testing. RLIP antisense treatment successfully reduced the ability of cells to form colonies, as shown by results from the colony-forming assay (Fig. 1, panel C).
Antineoplastic effects of RLIP depletion by RLIP antisense
The evidence of recent findings validates that malignant cells possess more sensitivity toward RLIP-targeting agents when compared with nonmalignant cells [6, 7]. We evaluated how RLIP antisense affects RLIP protein levels in the organs of living mice through this study. Non-tumor bearing animals injected with a single 0.2 mg dose of RLIP antisense were euthanized at 24 h and 48 h after treatment. The assessment of RLIP protein levels in crude homogenates from liver, lung, kidney, brain, spleen, and heart tissues was conducted using both ELISA and Bradford assays, while the purified rec-RLIP served as an ELISA standard (Fig. 2, panel A). RLIP content in all tested organs reported at a steady rate of 0.4 ± 0.05% compared to total extractable protein levels except in the spleen (Fig. 2, panel B), where the reported value was 0.6% similar to previous measurements in OC cells [6]. The administration of RLIP antisense resulted in significant reduction of RLIP so that levels declined to 0.01% after 48 h (Fig. 2, panel C). The immunohistochemistry tests generated results that matched the ELISA data, demonstrating tissue-wide RLIP protein reduction (Fig. 3).
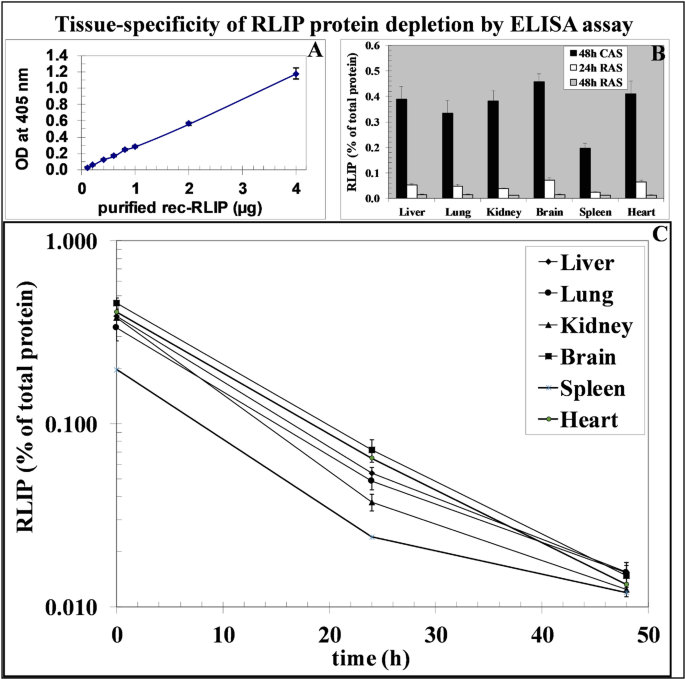
Effect of depletion of RLIP in C57Bl/6 mouse tissues by RLIP antisense. Single i.p. injections of 200 µg of RLIP antisense (or scrambled control) were given to C57Bl/6 mice that were then euthanized at 24 h and 48 h after administration. The ELISA standard curve received its standard from purified rec-RLIP (panel A). Tissue homogenates were used for ELISA-based protein quantification (panels B and C). CAS denotes scrambled antisense whereas RAS stands for RLIP antisense. The measurement showed 0 h represented 48 h after CAS treatment
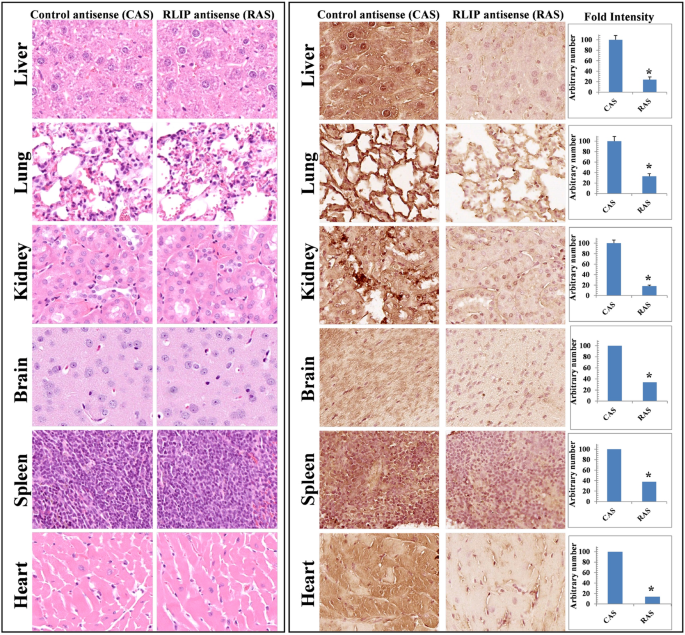
Effects on tissue morphology in excised organs after RLIP depletion (left panel) and RLIP depletion in C57Bl/6 mouse tissues by RLIP antisense, analyzed through immunohistochemistry using RLIP antibodies (right panel). Tissues received treatment with RLIP antisense and control antisense. The image shows representative magnified (400×) results of each treatment group. The experiment utilized RAS for RLIP antisense and CAS for control scrambled antisense. Error bars represent the mean with standard statistical deviation (n = 5). Results were considered statistically significant through a two-tailed analysis of the Student’s t-test method, *p < 0.02
To assess the possible toxic outcomes of RLIP antisense, we analyzed mouse tissue sections from liver, lung, kidney, brain, spleen, and heart via H&E staining. Tissues were prepared for analysis by fixing in 4% paraformaldehyde, embedding in paraffin, and sectioning into 5 μm slices, followed by H&E staining as outlined in Fig. 3. Canalicular dilation between the treatment groups, including RLIP antisense-treated samples, appeared similar with no visible harmful effects on tissue structure (Fig. 3, left panel). Tissues treated with RLIP antisense exhibited reduced RLIP expression, as demonstrated by immunohistochemical analysis (Fig. 3, right panel). The in vitro findings are further supported by in vivo results, showing that RLIP depletion inhibits cellular proliferation and induces programmed cell death.
Effect of RLIP depletion on apoptosis
To thoroughly investigate the impact of RLIP suppression on OVCAR8 cell proliferation, we performed apoptosis analysis. Apoptosis induction served as the third assessment method to evaluate RLIP suppression effects. Additionally, clonogenic assays revealed a significant reduction in colony-forming potential (Fig. 1, panel C). The analysis of apoptosis through TUNEL assessment was conducted on cells treated with either RLIP inhibitor or depletion procedures while receiving carboplatin treatments. The combination analysis of RLIP antisense and RLIP antibodies alongside carboplatin exposed OVCAR8 cells to a collaborative apoptotic reaction for the TUNEL assay (Fig. 4, panel A). The treatments using preimmune serum together with scrambled antisense did not trigger any changes in apoptosis. Our results matched the information from studies about tumor growth suppression.
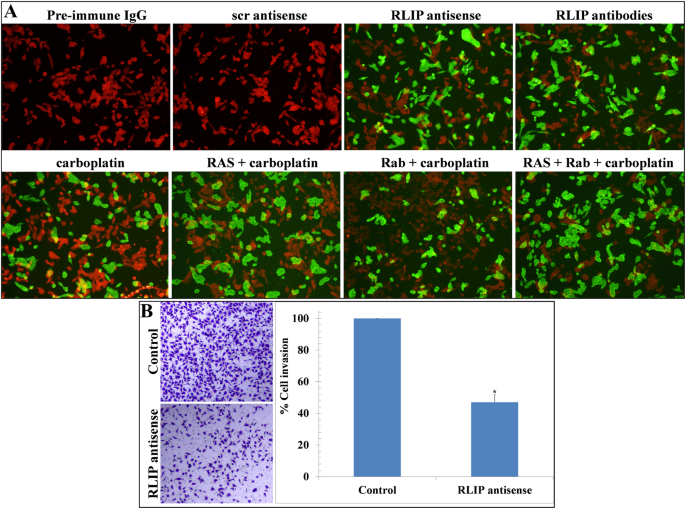
Effect of RLIP antisense, RLIP antibodies, and carboplatin on apoptosis as determined by TUNEL assay. Human OVCAR8 OC cells received treatment from control scrambled (scr) or RLIP antisense (10 µg/ml final concentration), preimmune IgG or RLIP antibodies (40 µg/ml final concentration), and carboplatin (20 µM) for 24 h followed by PBS wash. Cells were counted to 1 × 106 and laid on Poly-L-lysine-coated slides for TUNEL apoptosis assessment through Promega Apoptosis Fluorescence Detection Kit methods. Microscope analysis of slides occurred through Zeiss LSM510 META (Germany) equipment using filters at 520 nm and beyond 620 nm. The fluorescence images under identical exposure (×400 magnification) revealed apoptotic cells to have both green fluorescence and cell shrinkage characteristics (panel A). OC cell invasion was evaluated after 24 h treatment with 10 µg/ml final concentration of either scrambled control antisense or RLIP antisense on OVCAR8 cells placed inside serum-free medium transwell inserts (8 μm pore size). The cells that received treatment were fixed with 4% paraformaldehyde solution before the crystal violet staining procedure. Invasive cells were imaged using a light microscope at 200× magnification power. The number of invading cells was calculated by normalizing their data to the control group percentages. Student’s t-test revealed a statistically substantial difference (*p < 0.02) between treated samples and controls (panel B). This experiment was repeated three times, with a representative image as shown
Effect of RLIP depletion on invasion
Our findings demonstrate that RLIP depletion exerts anti-migratory and anti-invasive effects on OC cells in vitro. Metastasis is a multi-step process involving cell migration and invasion, facilitating the spread of tumor cells from their site of origin to distant organs. The cell invasion evaluation under RLIP reduction conditions took place after OVCAR8 OC cells received RLIP antisense treatment (10 µg/ml) for 24 h through Matrigel® invasion chambers. The number of cells that infiltrated the treated cells reduced significantly according to crystal violet staining following treatment with RLIP antisense in comparison to control treatment. These findings demonstrate that RLIP deficiency diminishes the in vitro motility ability of OC cells (Fig. 4, panel B). Because scientists have established that overexpression of RLIP is crucial for tumor progression, we next investigated RLIP expression levels between control and RLIP antisense-treated OVCAR8 cells. The control and scrambled antisense-treated cells presented elevated RLIP protein levels, but the cells treated with RLIP antisense showed reduced protein levels [7].
Anti-neoplastic effects of RLIP depletion/inhibition and carboplatin
The combined anti-neoplastic effects of RLIP inhibitor with carboplatin were evaluated using fixed drug doses in OC cell lines through recent studies. The Chou-Talalay analysis served to detect synergistic effects by evaluating the combination effects of three agents at specified doses. The research established that RLIP antibody combined with RLIP antisense treatment along with carboplatin (Rab + RAS + carbo) created a synergistic interaction between the drugs. Laboratory testing of these experiments led to successful confirmation through the OVCAR8 OC cell line xenografts performed on nude-mice. In vivo studies of drug response and metastasis patterns make use of human cell transplants implanted into mice. The subcutaneous inoculation procedure made it possible for researchers to track tumor growth easily during the examination of treatment responses. The established OVCAR8 subcutaneous tumor mice received 100 µl of weekly i.p. injections containing Rab at 4 mg/kg b.w. and RAS at 4 mg/kg b.w., while carboplatin was administered at 20 mg/kg b.w. or various combination dosages for eight weeks (Figs. 5 and 6). The experimental groups receiving Rab + carbo and RAS + carbo drugs demonstrated better tumor response than either drug used alone. Animals treated with Rab or RAS experienced faster treatment outcomes compared to animals receiving carboplatin individually. Animals treated with a combination of all three agents experienced the fastest tumor regression (Fig. 5, panels A and B). The measured tumor weights on day 56 after initial treatment proved substantial for the reduction of tumor size (Fig. 5, panel C). The weight of animals remained equal in all treatment groups, thus validating the accepted safety aspects of the adopted interventions (Fig. 5, panel D). All animals received a beneficial tumor growth reduction after the researchers depleted and inhibited RLIP along with no weight-related adverse effects observed. No tumors were detected outside the treatment sites, as per microscopic examinations done on all animal groups.
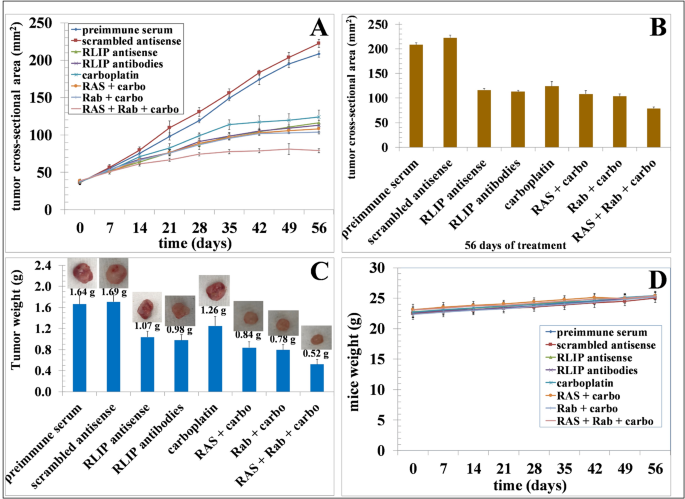
Effect of RLIP antibodies, RLIP antisense, and carboplatin on OVCAR8 OC progression in vivo. The experimental groups of Athymic nu/nu nude mice with OVCAR8 OC cells received different treatments consisting of (i) preimmune serum, (ii) scrambled antisense, (iii) RLIP antisense at 4 mg/kg body weight (b.w.), (iv) RLIP antibodies at 4 mg/kg b.w., (v) carboplatin at 20 mg/kg b.w., (vi) RLIP antisense combined with carboplatin, (vii) RLIP antibodies combined with carboplatin, and (viii) RLIP antibodies combined with RLIP antisense and carboplatin. Tumors grew more slowly in subjects treated with the combination of RLIP antibodies or RLIP antisense with carboplatin but without other treatment ingredients, among other tests (panel A). The experimental groups differed significantly from the control groups based on tumor cross-sectional area measurements (mm²) at the endpoint on day 56 (panel B). The experimental group tumors demonstrated reduced average wet tumor weights toward the end of the study compared to controls because controls weighed 1.64 g and 1.69 g but experimental groups weighed 1.07 g, 0.98 g, 1.26 g, 0.84 g, 0.78 g, and 0.52 g, respectively (panel C). The administration of RLIP antibodies together with RLIP antisense or carboplatin to mice revealed no indication of treatment-related stress or toxicity through assessments of movement abilities and posture as well as digestive functions, tissue redness, and swelling. The control group mice as well as the treated mice groups showed no variation in their body weights during both the beginning and completion stages (panel D). This research establishes crucial evidence of tumor growth control by combination treatment of RLIP antibodies and RLIP antisense with carboplatin in OC models in vivo. Mean ± SD statistical data points from treatments were obtained from each group consisting of five mice
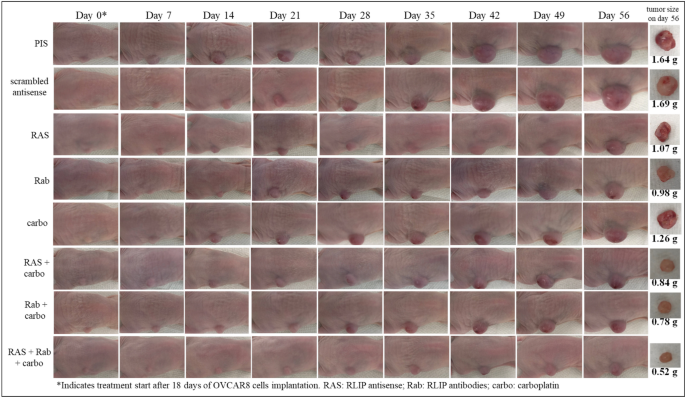
Effect of RLIP antibodies, RLIP antisense, and carboplatin on the size of subcutaneously implanted human OC (OVCAR8) cells in nude mice. Athymic nude nu/nu Hsd mice were obtained from The Jackson Laboratory, Bar Harbor, ME and received one week of room adjustment before experimental procedures. The Institutional Animal Care and Use Committee approved all animal procedures through IACUC #22,011. Eight different groups of 8-week-old mice with five animals per group received changing i.p. treatments including preimmune serum, scrambled antisense, RLIP antisense at 4 mg/kg body weight (b.w.), RLIP antibodies at 4 mg/kg b.w., carboplatin at 20 mg/kg b.w., combination of carboplatin and RLIP antisense, carboplatin and RLIP antibodies, and combination of carboplatin with RLIP antibodies and RLIP antisense. A 100-microliter cell suspension was administered subcutaneously, which contained 1 × 10⁶ OVCAR8 cells placed in one flank for each of the 40 mice. After tumors grew to achieve a size of around 40 mm² in 18 days the laboratory mice received their randomized experimental treatments. The experiment administered 100 µl volume treatments including RLIP antibodies, RLIP antisense, and carboplatin as well as their various combinations through i.p. delivery once a week over 8 weeks. The control groups received either preimmune serum or control antisense that contained scrambled sequences through administration of 100 µl solutions. Biweekly tumor size measurements used calipers on two axes and the researchers documented body weight changes. The photograph documentation process for animals and tumor images occurred from days 1 through 56 post-treatments along with specific capture days at 1, 7, 14, 21, 28, 35, 42, 49, and 56. Photographs of tumors were also taken on day 56 after treatment
Studies have shown that carboplatin demonstrated an additional effect of enhancing tumor regression. The experimental results demonstrate that membrane transporters are the primary target of RLIP blockade, however, the use of carboplatin enhances its toxicity levels. According to the results, the anti-tumor activity of RLIP antibodies matches the antisense treatment when their transport function maintains RLIP’s anti-apoptotic characteristic. A treatment combination of MAP pathway blocker and carboplatin shows potential to become an effective medical solution for OC. The use of RLIP antisense resulted in a considerable reduction of PI3K and Akt phosphorylation within examined tumors. The study evaluated the expression patterns of vital regulatory factors Bax and E-cadherin alongside cyclin B1 and CDK4 because these components directly shape cancer cell division and death processes. Tumor tissue lysates from mice treated with RLIP antisense showed reduced levels of vimentin, cyclin B1, and CDK4, while pro-apoptotic Bax and tumor-suppressive E-cadherin were significantly elevated (p < 0.05) [6]. These findings suggest that tumor growth inhibition is associated with the disruption of key cancer signaling pathways.
Ovarian tumor sections receiving RLIP antisense treatment exhibited reduced expression of RLIP protein alongside decreased Vimentin, Ki67, and CD31 protein but elevated E-cadherin protein. The expression of E-cadherin exhibited higher levels than before treatment. Empirical laboratory tests support earlier in vitro results, which demonstrate that RLIP reduction extends to cellular proliferation suppression and apoptotic promotion in OC. Paramount observations have tested and validated RLIP as a potential therapeutic target while directly supporting our conceptual model of this protective protein through its stress-resistant transport capacity. The research established that RLIP antibodies display similar anti-neoplastic properties to carboplatin fixed doses by acting independent from human xenograft models of OC. The dual effect of using RLIP antisense or RLIP antibodies in combination with carboplatin is likely derived from their shared and separate binding sites on RLIP for glutathione-electrophile conjugates (GS-Es). When used together with RLIP antisense along with carboplatin the inhibition of physiological ligand efflux becomes more effective.
Discussion
Ovarian carcinoma functions as one of the most lethal gynecological cancer types worldwide because patients often receive a late-stage diagnosis with resistant tumors that developed from chemotherapy treatments. The PI3K/Akt signaling pathway becomes excessively active in OC, contributing powerfully to cancer cell resistance against chemotherapy treatment [10]. The development of antibody-drug conjugates (ADCs) attracts significant interest because these pharmaceutical compounds enable the precise delivery of cytotoxic drugs to cancer cells together with targeted antigen recognition and reduced damage to normal tissues. Mirvetuximab soravtansine represents an FDA-approved ADC that has drawn significant research interest due to its potential capabilities in penetrating drug resistance in ovarian carcinoma [11].
Epithelial ovarian cancer (EOC) continues to be the main cause of gynecological-related fatalities; however, treatment improvements in chemotherapy with surgery have produced better survival responses but still affect less than 25% of long-term survival among patients. Data indicates that PARP inhibitors show enduring therapeutic benefits for treating advanced EOC with BRCA mutations within the 20% of all EOC cases. The inhibitory effect of these drugs is less pronounced in EOC cases lacking BRCA mutations and those with platinum-resistant disease. The most common EOC subtype, high-grade serous ovarian cancer (HGSOC), usually presents at later stages (III and IV), resulting in severe clinical complications because the cancer has metastasized within the peritoneal cavity [12, 13]. The delayed diagnosis of the disease plays a primary role in patients’ poor survival outcomes because patients who reach stages III and IV survive for 5 years at rates of 35% and 24%. The current treatment combination of cytoreductive surgery together with chemotherapy fails to achieve satisfactory results because of high mortality rates, which require better therapeutic alternatives conferring to statistical data [14]. Thus, the development of better treatment plans for EOC requires a comprehensive knowledge of its biological traits.
RLIP functions as a stress-responsive protein that transports GS-E and various drugs and serves as an overexpressed transporter in malignant cells. Research demonstrates that this transporter displays a prominent anti-apoptotic function particularly in cancer cell contexts [15]. Studies have focused on RLIP expression in tumors because this activity promotes both cell migration and enables human cancer cell metastasis [16]. RLIP facilitates cell migration and spreading by modulating the Rac1 and Arf6 signaling network [17]. Treatment of cells with antibody drugs or antisense therapy blocks RLIP’s protective functions, which creates conditions for oxidative stress damage of tissues and cells. The increased presence of electrophiles and RLIP protein expression in cancer cells indicates that cancer cells depend more heavily on RLIP than regular tissues. Furthermore, the loss of RLIP, via antibody or antisense therapy, increases the cells’ sensitivity to radiation and chemotherapy [18, 19].
Our research showed that RLIP antibody or RLIP antisense treatment applied intraperitoneally to nude mice created sustained remission of OVCAR8 OC xenograft tumors. We conclude that OC cells need RLIP as a survival protein because its inhibition or depletion makes tumors regress while causing minimal harm to the animals. Because the treatment effects between RLIP antibodies and antisense remained equivalent, it can be concluded that the functionality of RLIP as an anti-apoptotic and stress-protective protein tightly correlates to its transport characteristics. RLIP functions as an exceptional anticancer target because it operates as both a survival protein and an anti-apoptotic factor that transforms cells into multidrug transporters that aid in cancer cell proliferation. A new understanding about drug resistance in OC has emerged from this study while offering potential solutions for better treatment of drug-resistant OC.
Conclusions
RLIP functions as a vital protein that operates during tumor evolution within different body regions where it exists, including ovarian and other tissues. OC stands as the most fatal gynecological condition causing death because drug resistance creates substantial barriers in medical treatment. The increased use of new drugs in medical treatment makes treating drug-resistant OC an increasing medical challenge. The critical oncogenesis signaling route known as PI3K/Akt operates in the pathway of 70% of OC patients show pathologic disruptions. The PIK3CA gene mutation, which controls the catalytic subunit, demonstrates its presence in 10% of OC cases [20].
RLIP protein serves as a necessary bridge between pathways that process foreign compounds and GSH reserves and paths involved in stress response activation. RLIP acts as a protective cell component that protects normal cells from dangerous agents while avoiding cancer cell death through apoptosis. Tumor regression occurs when RLIP is inhibited and depleted in cases of RLIP overexpression, making carboplatin treatment more effective. The tissue sections of mice treated with RLIP antisense showed reduced RLIP protein levels along all tissues compared to mice receiving control antisense treatment based on immunohistochemical analysis. Our findings establish RLIP as an essential factor during ovarian carcinogenesis. Targeted RLIP blockage through inhibition methods and depleting activities leads to apoptosis in OC cells, thus creating an effective therapeutic target. The laboratory findings support ongoing investigations which will examine RLIP depletion or inhibition as treatment for human OC. RLIP represents a critical component in cancer cell survival mechanisms because its targeted interruption creates antitumor effects in OCs.
