Article Content
Abstract
Background
Recurrent pregnancy loss (RPL) affects 1–2% of women worldwide and poses diagnostic and therapeutic challenges due to its multifactorial causes. Preimplantation genetic testing for aneuploidy (PGT-A) aims to improve outcomes by selecting euploid embryos, but its benefits in RPL patients remain uncertain. This study compared the effectiveness of PGT-A versus conventional in vitro fertilisation/intracytoplasmic sperm injection (IVF/ICSI) in improving cumulative live birth rates (CLBRs) and explored the effects of maternal age and miscarriage frequency on treatment efficacy.
Methods
This study included RPL patients who underwent their first oocyte retrieval and at least one single-blastocyst transfer between June 2016 and June 2022. Patients were divided into an IVF/ICSI group (n = 156) and a PGT-A group (n = 198). Primary outcomes included the CLBR, live birth rate, miscarriage rate, time to live birth, and perinatal outcomes.
Results
After three single-blastocyst transfer cycles, no significant difference was observed in the conservative CLBR between the PGT-A and IVF/ICSI groups (Cycle 1: adjusted odds ratio [aOR] = 0.78, 95% confidence interval [CI]: 0.49–1.23; Cycle 2: aOR = 0.81, 95% CI: 0.51–1.29; Cycle 3: aOR = 0.96, 95% CI: 0.60–1.53; all P > 0.05). Similarly, the optimal CLBR after three transfer cycles showed no significant difference between the two groups (P > 0.05). However, the time to live birth was significantly longer in the PGT-A group than in the IVF/ICSI group (adjusted hazard ratio = 0.56, 95% CI: 0.42–0.75, P < 0.05). Other outcomes were comparable between the two groups.
Conclusion
PGT-A did not significantly improve the CLBR or shorten the time to live birth in RPL patients. Further research is needed to elucidate its role and identify potential subgroups within the RPL population that may benefit from PGT-A.
Introduction
Globally, an estimated 1–2% of women experience recurrent pregnancy loss (RPL) [1], a condition defined by the European Society of Human Reproduction and Embryology (ESHRE) as the experience of two or more miscarriages [1]. RPL is a complex clinical issue with multifactorial causes, including genetic, anatomical, endocrine, and immunological factors, which complicate its diagnosis and management.
Preimplantation genetic testing for aneuploidy (PGT-A) is a diagnostic procedure that involves taking a biopsy of the trophectoderm layer of blastocysts generated via standard in vitro fertilisation (IVF) or intracytoplasmic sperm injection (ICSI) to discern euploid embryos [2]. Numerous studies have indicated that PGT-A can potentially improve pregnancy and live birth rates, concurrently reducing the likelihood of miscarriage [3,4,5]. Moreover, it has been linked to an increase in the cumulative live birth rate (CLBR) and a decrease in the number of transfer cycles needed to achieve a live birth [6]. However, the reported benefits of PGT-A remain inconsistent, particularly when considering different patient populations and clinical contexts.
For women with favourable prognoses, evidence from randomised controlled trials indicates that PGT-A does not significantly improve live birth rates [7]. Meanwhile, for RPL patients, prospective studies have suggested that although PGT-A may reduce the number of transfer cycles required to achieve a live birth, it does not substantially improve the overall live birth rates [8]. The use of PGT-A remains controversial due to several unresolved issues. Although it is primarily recommended for cases with advanced maternal age (AMA), RPL, or recurrent implantation failure (RIF), critics have highlighted its potential limitations, including procedural risks, high costs, and the possibility of misdiagnosis due to mosaicism in embryos [9]. Furthermore, ethical concerns have been raised regarding the prioritisation of embryos based solely on genetic testing, which may conflict with patient preferences or cultural values. Many studies have examined the effectiveness of PGT-A in cases with AMA and RIF [6, 8, 10, 11], but studies addressing RPL are often constrained by their limited scope and narrow focus on the outcomes of initial transfers while ignoring the outcomes of subsequent embryo transfers [8, 12]. Considering that numerous patients may require multiple cycles to achieve a live birth [13], there is an increasing emphasis on the significance of the CLBR [7, 14]. According to the ESHRE guidelines, the CLBR, live birth rate, and pregnancy loss rate are crucial outcomes when evaluating treatment strategies in RPL patients [1]. Additionally, measures such as the conservative CLBR, optimal CLBR, and conditional live birth rate have been proposed to account for potential biases and provide a more comprehensive assessment of treatment outcomes [15,16,17,18].
This retrospective study evaluated the comparative effectiveness of PGT-A versus conventional IVF/ICSI in improving the CLBR, time to live birth, and perinatal outcomes in RPL patients. Subgroup analyses based on maternal age and miscarriage frequency were conducted to further explore potential modifiers of treatment efficacy.
Materials and methods
Patients
This retrospective cohort study included RPL patients who underwent their initial oocyte retrieval cycle followed by at least one single fresh or frozen-thawed blastocyst transfer cycle as part of IVF/ICSI or PGT-A between June 2016 and June 2022 at the Reproductive Medicine Center of the Third Affiliated Hospital of Zhengzhou University. RPL patients voluntarily chose PGT-A or IVF/ICSI after comprehensive counselling by our specialised genetic counsellors, who provided detailed explanations of the advantages, limitations, and potential outcomes of both approaches. In the PGT-A group, only euploid embryos were selected for transfer, ensuring the exclusion of both aneuploid and mosaic embryos. Patients were required to meet strict inclusion and exclusion criteria. The inclusion criteria were as follows: (1) patients diagnosed with RPL, defined as having two or more clinical pregnancy losses documented by ultrasound or histopathology; (2) first oocyte retrieval cycle during the study period; and (3) at least one single-blastocyst transfer cycle following IVF/ICSI or PGT-A, with a single blastocyst transferred in each cycle. The exclusion criteria included known uterine anomalies, untreated uterine septa, adenomyosis or submucosal fibroids, endometrial polyps or intrauterine adhesions, contraindications to pregnancy, use of donor sperm, presence of autoimmune diseases, and second live births in the same patient. Patients who underwent cleavage-stage embryo transfers, double-blastocyst transfers, or combined IVF/ICSI and PGT-A cycles were also excluded, significantly narrowing the pool of eligible participants. Patients with incomplete clinical or follow-up data were also excluded to minimise potential bias (Fig. 1). Missing data were handled using multiple imputation techniques to ensure the robustness of the analyses. The final cohort comprised 196 PGT-A patients and 158 IVF/ICSI patients. Although the small sample size limits the statistical power of the subgroup analyses, it reflects a focus on rigorous inclusion and exclusion criteria to minimise confounding factors. The subgroup analyses were conducted based on maternal age and miscarriage frequency. These factors were chosen as they represent key clinical variables influencing RPL outcomes and potential modifiers of treatment efficacy. For example, AMA is associated with higher rates of aneuploidy, while the number of previous miscarriages may indicate the severity of RPL. This study complied with the ethical guidelines of China’s ‘Ethical Review Measures for Life Sciences and Medical Research Involving Humans’. Ethical approval was obtained from the Ethics Committee of the Third Affiliated Hospital of Zhengzhou University (approval number: 2024-052-01; Human Ethics and Consent to Participate declarations: not applicable; Clinical trial number: not applicable).
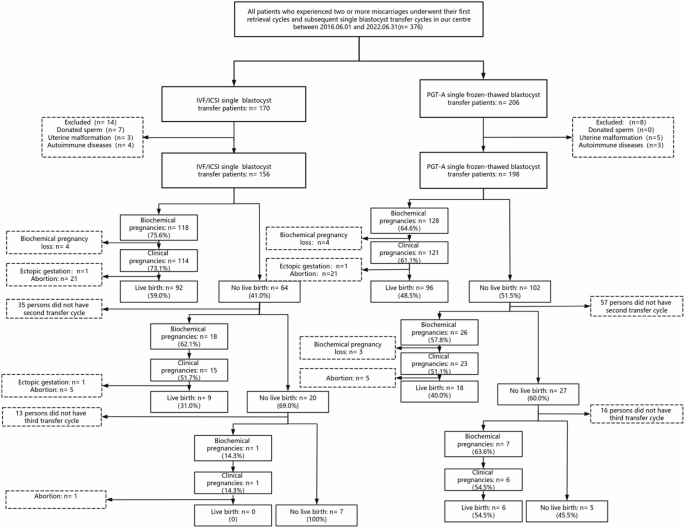
Study enrolment and exclusion flowchart
PGT-A and IVF/ICSI treatment
Ovarian stimulation, transvaginal ultrasound-guided oocyte retrieval, IVF/ICSI, embryo culture, blastocyst morphological assessment, embryo vitrification or thawing, embryo transfer, luteal phase support, trophectoderm biopsy, and three frozen embryo transfer protocols were performed as previously described by our centre’s researchers [19,20,21,22]. A good-quality blastocyst was defined as one with a score of 4BB or higher based on Gardner and Schoolcraft’s classification [23]. Biochemical pregnancy was diagnosed based on a positive human chorionic gonadotropin test at 14 days post-embryo transfer but the absence of a gestational sac on ultrasound at 6–7 weeks post-transfer. Clinical pregnancy was confirmed by the presence of an intrauterine gestational sac on ultrasound at 6–7 weeks of gestation. Miscarriage or termination of pregnancy was defined as pregnancy loss before 28 weeks of gestation. Live birth was defined as the delivery of a foetus with signs of life at ≥ 28 weeks of gestation, with multiple births counted as one live birth.
Perinatal outcomes
Data on PGT-A and IVF/ICSI treatment cycles were retrieved from our hospital’s electronic medical record system. Data on pregnancy and perinatal outcomes for clinical pregnancies delivered at our hospital were also collected from the electronic medical record system. For patients who received treatment at our centre but delivered elsewhere, trained nurses contacted them to confirm pregnancy outcomes within 2 months of the expected delivery date. Data on RPL patients and their singleton live births achieved via PGT-A and IVF/ICSI at our centre were collected. Pregnancy-induced hypertension was defined as systolic pressure ≥ 140 mmHg and/or diastolic pressure ≥ 90 mmHg recorded on two separate occasions at least 4 h apart after 20 weeks of gestation in previously normotensive women [24]. Pre-eclampsia was defined as the occurrence of proteinuria with systolic pressure ≥ 140 mmHg and/or diastolic pressure ≥ 90 mmHg after 20 weeks of gestation in previously normotensive women [24]. Gestational diabetes was defined according to ACOG Practice Bulletin number 190 [25]. Hypothyroidism during pregnancy was defined according to the criteria established by Gietka-Czernel and Glinicki [26]. Conditions such as premature rupture of membranes; placenta previa; caesarean section; gestational age (weeks); preterm birth (defined as delivery before 37 weeks of gestation) and very preterm birth (defined as delivery between 28 and 31 weeks of gestation); birth weight (grams), including low birth weight (< 2,500 g) and very low birth weight (< 1,500 g); birth height (cm); congenital defects (as defined by Ran et al. [22]); and Apgar scores at 1 and 5 min were also collected.
Sample size determination and sensitivity analyses
A priori sample size calculation determined that a minimum of 174 participants per group was required to detect a 10% absolute difference in the CLBR (e.g., 60% vs. 50%) with 80% power at a significance level of 0.05. This calculation assumed a 1:1 allocation ratio and accounted for an anticipated 10% loss to follow-up, leading to a target sample size of 192 participants per group. Although the final cohort included 196 PGT-A patients and 158 IVF/ICSI patients– slightly below the target for the IVF/ICSI group– post-hoc sensitivity analyses were conducted to address this limitation. Sensitivity analyses were performed to evaluate the robustness of the results under various assumptions about missing data and sample size. These included complete-case analyses and multiple imputation models to address potential biases introduced by excluded data. Additionally, subgroup-specific sensitivity analyses stratified by maternal age and miscarriage frequency were conducted to assess outcomes, given their roles as key clinical modifiers. For example, AMA is strongly associated with increased rates of aneuploidy, while the number of previous miscarriages reflects the severity and potential aetiology of RPL. Across all scenarios, the sensitivity analyses yielded consistent findings, supporting the reliability of the primary conclusions despite the slightly smaller-than-expected sample size in the IVF/ICSI group.
Statistical analysis
All statistical analyses yielded consistent results, underscoring the reliability of the primary conclusions. The statistical analyses were conducted using SPSS 25.0 (IBM, Chicago, IL). Categorical variables are presented as frequencies and percentages and compared using the chi-square test or Fisher’s exact test. Continuous variables are presented as means ± standard deviations and compared using Student’s t-tests. Conservative and optimal CLBRs were calculated. The conservative CLBRs assumed that women who did not continue treatment never achieved live birth, whereas the optimal CLBRs assumed that patients who discontinued treatment had the same live birth probability as those who continued treatment. Differences in optimal CLBRs were compared using the log-rank test, and hazard ratios (HRs) and 95% confidence intervals (CIs) were determined using Cox regression adjusted for endometrial thickness, maternal body mass index (BMI), number of miscarriages, number of retrieved oocytes, and infertility diagnosis. Adjusted odds ratios (aORs) and 95% CIs for the CLBRs were determined using multivariable logistic regression analyses while adjusting for endometrial thickness, maternal BMI, number of miscarriages, number of retrieved oocytes, and infertility diagnosis. P < 0.05 was considered to indicate statistical significance.
Results
Cohort analysis
From June 2016 to June 2022, 24,817 patients underwent PGT-A or IVF/ICSI treatment at our centre. Following the inclusion and exclusion criteria, 198 RPL patients who received PGT-A treatment and 156 RPL patients who underwent IVF/ICSI treatment were included in the final analysis.
Significant differences were observed between the PGT-A and IVF-ICSI groups in terms of maternal BMI, number of miscarriages, oocyte yield, endometrial thickness on the day of transfer, and infertility diagnosis type (female factors and other factors) (P < 0.05, Table 1). However, no significant between-group differences were observed in maternal age, paternal age, paternal BMI, maternal delivery history, duration of infertility, baseline antral follicle count, or the number of high-quality embryos transferred (P > 0.05, Table 1).
Subgroup analysis revealed additional stratified differences based on maternal age and the number of miscarriages.
For patients aged ≥ 35 years, significant differences were found in the number of previous miscarriages, oocyte yield, endometrial thickness, and infertility diagnosis between the PGT-A and IVF/ICSI groups (P < 0.05, Supplementary Table 1). Among patients aged < 35 years, only infertility diagnosis showed a significant difference between the two groups (P < 0.05, Supplementary Table 1).
For patients with two previous miscarriages, significant differences were observed in endometrial thickness on the day of transfer and infertility diagnosis between the two groups. In contrast, for patients with at least three previous miscarriages, significant between-group differences were noted in maternal BMI and infertility diagnosis (P < 0.05, Supplementary Table 2). All other baseline characteristics remained comparable between the two groups across all subgroup analyses for other variables.
Comparison of embryo outcomes
A total of 198 patients in the PGT-A group and 156 patients in the IVF/ICSI group completed their first transfer cycle, with 45 and 29 patients proceeding to a second transfer cycle and 12 and seven patients proceeding to a third transfer cycle in the respective groups.
Although the time to live birth was significantly longer in the PGT-A group than in the IVF/ICSI group, no significant between-group differences were observed in the pregnancy rate per transfer (aOR = 0.76; 95% CI: 0.50–1.16; P > 0.05, Table 2), miscarriage rate per pregnancy (aOR = 0.81; 95% CI: 0.42–1.55; P > 0.05, Table 2), or live birth rate per pregnancy (aOR = 0.90; 95% CI: 0.60–1.35; P > 0.05, Table 2). The prolonged time to live birth in the PGT-A group (adjusted HR [aHR] = 0.56; 95% CI: 0.42–0.75; P < 0.05, Table 2) may be attributable to the additional procedural steps required for PGT-A treatment, such as embryo biopsy, vitrification, and genetic analysis, which tend to extend the treatment cycle duration. This delay could impose a considerable burden on patients with AMA or diminished ovarian reserve.
Among patients aged ≥ 35 years, no significant differences were observed in the pregnancy rate, miscarriage rate, or live birth rate between the PGT-A and IVF/ICSI groups (P > 0.05, Supplementary Table 3). Similarly, for patients aged < 35 years, no significant between-group differences were found in the live birth rate, pregnancy rate, or miscarriage rate (P > 0.05, Supplementary Table 3). However, the time to live birth in those aged < 35 years was significantly longer in the PGT-A group than in the IVF-ICSI group (aHR = 0.46; 95% CI: 0.33–0.65; P < 0.05, Supplementary Table 3). These findings suggest that although PGT-A is designed to improve live birth rates by avoiding the transfer of aneuploid embryos, it does not reduce the time to live birth in women aged under 35 years or improve the CLBR in any age group.
For patients with at least three previous miscarriages, the outcomes were comparable between the two groups (P > 0.05, Supplementary Table 4). Similarly, among patients with two previous miscarriages, no significant between-group differences were observed in the live birth rate, pregnancy rate, or miscarriage rate. However, for patients with two prior miscarriages, the time to live birth was significantly longer in the PGT-A group than in the IVF/ICSI group (aHR = 0.59; 95% CI: 0.43–0.83, Supplementary Table 4). This raises concerns about the cost-effectiveness of PGT-A and its applicability for patients requiring timely outcomes.
These limitations should be carefully considered when counselling patients, particularly those with AMA or prior miscarriages.
