Article Content
Introduction
Invasive Gram-positive infections including bacteremia, prosthetic joint infections, and osteomyelitis are associated with a substantial burden to the healthcare system in both the inpatient and outpatient setting. Most of these infections are caused by Staphylococcus and Streptococcus species [1,2,3]. Continuation of treatment for invasive Gram-positive infections commonly requires outpatient use of intravenous (IV) antibiotics for several weeks with vancomycin or daptomycin as the standard of care (SOC) on the basis of the methicillin-resistant Staphylococcus aureus (MRSA) Infectious Diseases Society of America (IDSA) guidelines [4].
Vancomycin and daptomycin have proven efficacious for these infections; however, treatment success is not possible without patient adherence. Several weeks of daily parenteral antibiotic administration and continuous maintenance of intravenous (IV) lines can be cumbersome to patients in the outpatient setting. Outpatient parenteral lines can have complications, such as thrombosis and line infections, resulting in a potential increase of readmission rates and negative patient outcomes [5]. Owing to these potential factors, antibiotic selection for treatment continuation of invasive Gram-positive infections has recently focused on available agents with less frequent administrations and no need for outpatient parenteral lines.
Dalbavancin (DBV) is an intravenous lipoglycopeptide antibiotic approved to treat Gram-positive organisms such as MRSA with an FDA indication for the treatment of acute bacterial skin and skin structure infections (ABSSSIs) [6]. DBV has many benefits, including a prolonged terminal half-life of approximately 346 hours—resulting in less frequent administration; thus, outpatient parenteral lines are not required. Typical dosing for ABSSSI treatment is well-established with either a two-dose regimen (1000 mg followed by 500 mg 7 days later) or a one-dose regimen (1500 mg) [5, 6]. However, the optimal dosing strategy for the off-label treatment of invasive Gram-positive infections has yet to be established.
Several studies examine DBV’s unique pharmacokinetics with different regimens showing prolonged bone and tissue exposure post-infusion. Current literature describes a variety of dosing strategies for DBV including a single-dose regimen of 1500 mg, a two-dose regimen of 1500 mg on day 1 and day 8, a two-dose regimen of 1000 mg on day 1 followed by 500 mg on day 8, and a weekly dosing regimen of 1000 mg on day 1 followed by weekly 500 mg infusions. Overall, data from prior literature display promising clinical cure rates and pharmacokinetic modeling data among the various dosing strategies [7,8,9,10,11,12]. However, there is limited literature focusing on the two-dose DBV regimen of 1500 mg on day 1 and day 8 that is commonly utilized by our institution for the treatment of invasive Gram-positive infections. The purpose of this study was to compare two-dose DBV (1500 mg on day 1 and day 8) to SOC to assess clinical outcomes in the treatment of Gram-positive bacteremia, prosthetic joint infection (PJI), and osteomyelitis (OM).
Patients and methods
Study design
This study was a single-center, retrospective, observational cohort of adult patients diagnosed with either bacteremia, OM, or PJI who were treated and discharged from a 314-bed acute care community hospital (East Alabama Medical Center, EAMC) with outpatient orders for two-dose DBV or SOC. Patients were identified using the electronic medical records of Cerner Millennium (Cerner Corp., Kansas City, MO, USA) and TheraDoc (Premier Inc., Charlotte, NC, USA). Patient electronic medical records were reviewed from the time of patient discharge to 90 days posttreatment completion to assess for the primary and secondary objectives. The study protocol was approved by the East Alabama Health and Auburn University institutional review boards.
Study participants
Patients were included in this study if the following criteria were met: they were at least 19 years of age, were discharged from EAMC with outpatient parenteral antibiotic orders, and received either two-dose DBV or SOC in the outpatient treatment of OM, PJI, or bacteremia between 1 July 2019 and 31 December 2023. OM and PJI were diagnosed at the discretion of the treating physician by radiologic findings (X-ray, computerized tomography, magnetic resonance imaging), histologic findings, and/or clinical documentation stating these diagnoses. Exclusion criteria included vulnerable populations (pregnancy and prisoners), concomitant endocarditis, patients who did not receive both doses of DBV 1500 mg, severe renal impairment defined as creatinine clearance ≤ 30 mL/min (per Cockcroft–Gault) or receiving hemodialysis or other renal replacement therapy, and receipt of outpatient SOC prior to first DBV administration [13]. Patients with polymicrobial infections were permitted to receive either IV or oral therapy for Gram-negative coverage in addition to SOC or DBV.
Study intervention
DBV administrations were scheduled and performed at EAMC’s non-oncology infusion center where a 30-min infusion of 1500 mg DBV occurred, with orders indicating a dosing schedule of day 1 and day 8. Upon audit of 28 charts, over 90% of patients received their second dose on day 8, plus or minus 1 day. Patients who received a single dose of DBV were excluded. Our institutional protocol has been in place since 2019, and while the two-dose regimen was initially intended for the treatment of osteomyelitis, it has expanded to include other invasive infections. SOC infusions of vancomycin and daptomycin were based on the recommendations of the infectious diseases (ID) physician; they were nonstandardized and administered via various home infusion companies.
Objectives
The primary outcome was clinical success, defined as no known clinical or microbiological evidence of recurring infection and no additional antibiotic therapy required for the infection within 90 days from hospital discharge. Secondary outcomes included 90-day infection recurrence, additional antibiotic therapy within 90 days, 90-day infection-related readmission, 90-day all-cause mortality, treatment-emergent adverse reactions, infection-related emergency department (ED) visits, the percentage of patients switched to oral antibiotics, rate of infectious disease follow-up, and duration of inpatient antibiotics. Clinical recurrence of OM and PJI was defined as worsening of infection, new purulence, unplanned amputation, or further debridement within 90 days from discharge. Microbiological recurrence of osteomyelitis and PJI was defined as new positive cultures 42 days or longer after the initial positive culture. Clinical and microbiological recurrence of bacteremia was defined as recurring positive blood cultures at least 7 days from clearance of index bacteria. Infectious-related readmissions were defined as readmissions occurring secondary to clinical recurrence as defined above, a line-related complication, requirement for further surgical intervention, or an antibiotic-related adverse drug reaction.
Statistical analysis
All statistical tests were performed using SPSS® statistics software (IBM Corp). Primary and secondary outcomes were assessed using Pearson chi-squared or Fisher’s exact test for categorical variables and Student’s t-test for continuous data. Normality of data was assessed by Shapiro–Wilk tests. Statistical significance for all tests was defined as a two-tailed p-value of < 0.05.
Results
Patient selection and demographics
A total of 196 patients were screened for inclusion. After applying exclusion criteria, 148 patients were included in the analysis, with 86 patients within the SOC group and 62 patients within the DBV group (Fig. 1). The average age in both groups was 60 years old with a mean weight of approximately 95 kg. The DBV group had a higher median Charlson Comorbidity Index (CCI) score of 4.5 compared with 3 in the SOC group (p = 0.016). Within the SOC group, bacteremia was the most common indication for antibiotics, while the majority of DBV patients carried an indication of OM or PJI. Most of the SOC group had staphylococcal infections, while polymicrobial infections or lack of cultures predominated in the DBV group (Table 1).
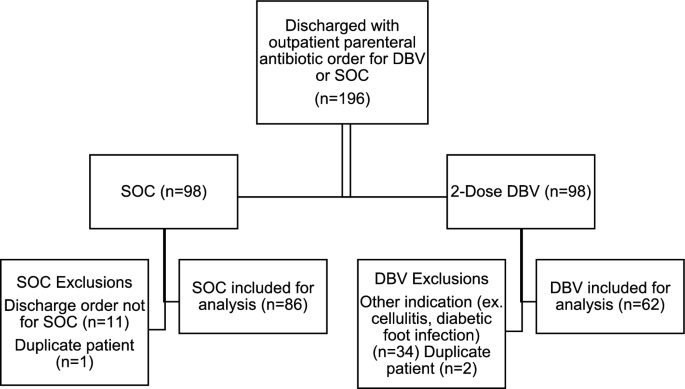
Inclusion pathway
Outcomes
Clinical success was achieved by 79 of 86 patients (92%) in the SOC group compared with 55 of 62 (89%) patients in the DBV group (p = 0.518). Among patients with OM and PJI, clinical success was achieved by 87% (34/39) and 88% (49/56) of patients in the SOC and DBV groups, respectively (p = 0.963). Among patients with bacteremia, the clinical success rate was 96% (51/53) and 100% (16/16) for the SOC and DBV groups, respectively (p = 0.587).
Clinical and microbiological recurrence were less than 10% in both groups (Table 2). The DBV group received additional antibiotics within 90 days more frequently than the SOC group (31% versus 15%; p = 0.024). The most used additional agent was oral levofloxacin, seen among 13 patients undergoing SOC and 10 patients taking DBV. The SOC group had a longer mean duration of inpatient antibiotics compared with the DBV group (10.1 days versus 6.5 days; p < 0.001). Within the SOC group, eight patients (9%) were eventually transitioned to oral therapy; all eight patients received oral linezolid. No patients in the DBV group were transitioned to oral therapy. Infection-related readmission within 90 days was more common in the SOC group compared with the DBV group (19% versus 5%; p = 0.011). No patients in either group experienced mortality within 90 days. Two patients in the SOC group experienced a treatment-related adverse event, with both patients experiencing hypersensitivity to daptomycin. Infection-related ED visits occurred in 6% of the SOC group compared with 0% of the DBV group, with the majority of these visits consisting of catheter-related issues (p = 0.063). More patients within the DBV group completed an outpatient infectious disease follow-up compared with the SOC group (90% versus 70%; p = 0.003).
Discussion
In the studied sample, 92% of the SOC group and 89% of the DBV group achieved the primary endpoint of clinical success, which is consistent with recent primary literature displaying similar clinical cure rates using various DBV dosing strategies. A retrospective, observational study of 48 patients who mostly received 1–2 doses of DBV for various Gram-positive invasive infections found 97% of the DBV group versus 88% of the SOC group achieved clinical cure at day 42 [8]. Molina and colleagues found no difference in 90-day clinical failure among a retrospective cohort of 225 patients treated with either SOC or 1–2 doses of DBV for S. aureus bacteremia [9]. Furthermore, a randomized, open-label, comparator-controlled trial of 80 patients with osteomyelitis treated with either SOC or DBV (1500 mg on day 1 and day 8) saw clinical cure at day 42 in 97% of the DBV group versus 88% of the SOC group [10]. This study reflected the most similar population to our study in terms of DBV dosing (1500 mg on day 1 and day 8), and the similar clinical cure rates were reassuring.
Clinical success in our study was demonstrated among 87% (n = 34) and 88% (n = 49) of patients with OM/PJI in the SOC and DBV groups, respectively. Success rates vary among literature assessing OM and PJI. Among a small cohort of patients with diabetes-related foot OM, 9 of 11 (82%) patients evaluable at 6 months experienced remission [14]. Of note, DBV dosing varied within this study, at a dose of 1500 mg times one, two, or three doses depending on duration of treatment. Within another study in which patients received two doses of 1500 mg, with possibility for regimen extension with further monthly doses, cure rates by indication were 93% for ABSSSI, 100% for bacteremia, 90% for acute osteomyelitis, 0% for chronic osteomyelitis, 75% for native joint septic arthritis, and 33% for prosthetic joint infection [15]. However, most bone and joint treatment failures occurred in patients without source control. Among a small French cohort of patients with PJI, 8 of 17 patients (47%) achieved clinical cure [16]. In total, eight patients in this study received two 1500 mg doses, with half of these patients experiencing treatment success [16].
Patients in the DBV group were more likely to receive additional antibiotics along with or after DBV treatment compared with the SOC group (31% versus 15%; p = 0.024), most commonly, oral levofloxacin. On the basis of physician progress notes and practice patterns at our institution, the authors conclude that this is likely related to more of the DBV group (50% versus 23%) having a polymicrobial infection or lack of cultures compared with the SOC group. This led to the addition of Gram-negative coverage in more patients. The higher rate of additional antibiotic use and the higher prevalence of polymicrobial infections in the DBV group are potential confounding variables within our study, among several differences in baseline characteristics. The additional antibiotics may have influenced clinical success rates. Similarly, the presence of polymicrobial infections could complicate the interpretation of the results, as these infections might require different treatment approaches compared with monomicrobial infections. Patients in the DBV group also tended to be older (mean age of 63 years versus 59 years), have a higher CCI score (median 4.5 versus 3), and have osteomyelitis or PJI versus bacteremia involving another source.
More infection-related readmissions and ED visits were observed among the SOC group (19%) versus the DBV group (5%). Veve and colleagues conducted a retrospective cohort study of patients with invasive Gram-positive infections receiving DBV or SOC. Infection-related readmission incidence among DBV patients was 17% versus 28% for SOC patients, and DBV used was associated with lower infection related readmission (adjusted odds ratio [adjOR], 0.10; 95% confidence interval [CI], 0.04–0.31). Within their study, the most common reason for drug discontinuation or readmission was peripherally inserted central catheter (PICC)-line complications in the SOC group [17]. In contrast, Cain and colleagues found no difference among infection-related readmission rates between patients with osteomyelitis who received DBV (31%) versus SOC (31.3%) [5]. Additionally, Molina and colleagues found similar readmission rates among their patients with S. aureus bacteremia who received SOC (26.7%) versus DBV (24.4%); however, this does not take into account that 36.6% of the SOC group was discharged to another healthcare institution, which could skew the potential readmission rates [9]. Frazier and colleagues compared DBV with SOC in patients unable to receive outpatient parenteral microbial therapy. Patients in the SOC group completed therapy at a post-acute care facility to complete antibiotics, which differs from our study and could impact readmission rates. Nonetheless, readmission rates for the DBV and SOC groups were 19% and 22% at 90 days, respectively [18]. The rate of reported adverse drug reactions (ADRs) within our study—for example, infusion reactions—was low in both groups, likely owing to the outpatient nature of this study; the authors had limited ability to assess reactions in this setting unless noted at the follow-up ID physician visit, ED visit, or subsequent hospitalization. Several ADRs may not have been adequately documented in a manner viewable by investigators and, therefore, could have been missed.
On average, patients in the DBV group received less inpatient parenteral antibiotics. This finding is consistent among other studies [5, 9, 10, 17]. We did not assess the potential cost savings of this result; however, previous studies found this to be monetarily beneficial for their institution [8, 19, 20]. The facilitation of earlier discharge and lower readmission rates with DBV administration could provide further financial benefit for institutions.
Overall, the small sample size and difference in baseline characteristics limit the application of this study. The differences in baseline characteristics, such as CCI, pathogen distribution, and additional antibiotics, may be reflective of selection bias in this retrospective analysis. Further, microbiological cultures were not collected for every patient. Assessment of ADRs, clinical success, and whether or not patients were lost to follow-up was limited by the retrospective nature of the study, as not all patients had scheduled follow-up visits with the ID physician. It is a reasonable assumption that, as the only hospital within an approximately 30-mile radius, most patients would return to our facility for follow-up care in the event of an infection relapse, recurrence, or ADR; however, information about encounters at other institutions was not available to study investigators. On a positive note, this study does reflect a “real-world” design where patients often have a variety of pathogens or lack of cultures. This is expected to increase external validity. The high rate of clinical success is consistent with results in other studies that utilize the same dosing strategy. Additional studies with larger sample populations and balanced baseline characteristics are needed to further establish the role of two-dose DBV in the treatment of invasive Gram-positive infections.
Conclusions
Similar rates of clinical success were found among SOC versus DBV patients. The differences in baseline characteristics, including 50% of the DBV group not having any cultures or having polymicrobial infection, limits the interpretation and application of this study. However, the high rates of clinical success seen in the DBV group are consistent with the results in similar studies utilizing the same dosing regimen. Future studies with larger sample populations and balanced microbiological data are needed to further establish the role of DBV in the treatment of invasive infections.
