Article Content
Abstract
Chemical analysis is a fundamental concept in chemistry, which is characterized by polysemy observed in both scientific and everyday language, which are both employed in science classrooms. Our work aims at developing a conceptual profile model of chemical analysis as a methodological tool for describing the heterogeneity of students’ ways of thinking about the concept of chemical analysis. In this study, we present our findings resulting from an inductive qualitative analysis of secondary data obtained from the historical and epistemological literature (sociocultural domain) and students’ alternative conceptions and thinking (ontogenetic domain) in a dialogic way. A preliminary classification of the ways of thinking about chemical analysis is proposed: chemical analysis as (a) everyday analysis, (b) alchemical analysis, (c) empirical analysis, (d) classical analysis, (e) classical instrumental analysis, and (f) a contemporary tool for society. Proposed categories of ways of thinking are established and stabilized by ontological, epistemological, and axiological commitments, which were identified during data analysis.
Explore related subjects
Discover the latest articles and news from researchers in related subjects, suggested using machine learning.
- Analytical Chemistry
- Chemistry Education
- Conceptual Analysis
- History of Chemistry
- Philosophy of Chemistry
- Chemistry In Society
1 Introduction
One of the main goals of education in the twenty-first century worldwide is to foster scientific thinking (Fischer et al., 2014; National Research Council, 2013; Zhou et al., 2016). Aligning with this goal, chemical education fosters chemical thinking that has been conceptualized as “the development and application of chemical knowledge and practices with the main intent of analyzing, synthesizing, and transforming matter for practical purposes” (Sevian & Talanquer, 2014). Numerous studies on chemical thinking have shown that students’ conceptualizations and ways of thinking about different chemical concepts vary widely (Caspari et al., 2018; Cooper et al., 2016; Moon et al., 2017; Talanquer, 2013; Yan & Talanquer, 2015), which poses significant challenges for chemistry education (Talanquer, 2019). A tool which not only assists teachers in recognizing the many ways themselves and students think and speak in science classrooms but also helps students broaden their thinking by incorporating new scientific ideas is the conceptual profile framework (da Silva Costa & dos Santos, 2022; Mortimer et al., 2012, 2014a; Orduña Picón et al., 2020).
The conceptual profile research program initially aimed at the determination of the zones of conceptual profile models of ontoconcepts like matter, energy, and life. These are actually very wide categories that pertain to key fundamental categories of the contemporary scientific world view. Later, the research program has been expanded, and the ontoconcepts in question gradually evolved into more specific concepts (El-Hani et al., 2023). According to the literature review, there are many scientific and chemistry concepts for which conceptual profile models have been proposed. We indicatively mention life (life, living organisms and adaptation) (Mortimer et al., 2012), energy (Aguiar et al., 2018) (heat, entropy, spontaneous physical and chemical processes) (do Amaral et al., 2014) and thermal physics in general (Aguiar, 2014), matter (particle models of matter, atoms, molecules) (Mortimer et al., 2012), substance (do Amaral et al., 2018; Orduña Picón et al., 2020), chemical bonding (Baltieri et al., 2021), chemical reactions (chemical change) (Solsona et al., 2003), equilibrium (da Silva Costa & dos Santos, 2022), and chemistry as a discipline (Freire et al., 2019). Our literature review indicates that a conceptual profile model of chemical analysis has not yet been built; therefore, its development fills a gap in the pertinent body of literature.
Our research project’s main goal is to develop a conceptual profile model for chemical analysis. A concept should meet the following criteria so that a conceptual profile model could be constructed for it: (1) it must be central and not peripheral to a given science, (2) it must be polysemous so that there are several ways of thinking about it, and (3) it must be used in both everyday and scientific language so that one can derive a model of heterogeneity of ways of thinking and speaking that can be used to analyze students’ discussion in a science classroom (Mortimer et al., 2014b).
We argue that chemical analysis is a central concept in the discipline. As Simon (2002) describes, Diderot’s Encyclopedia, which dates back to the Enlightenment era, provides a characterization of analysis as a “key concept” in chemistry. Furthermore, Klein (2012) claims that, prior to atomic theories, the conceptual network of chemistry consisted of concepts like chemical compounds, affinity, and analysis. As this conceptual network expanded to include notions such as molecules, atoms, and ions, chemical analysis remained a valuable part of it.
It is important to highlight that chemical analysis is a complex concept that includes both material entities (the targets and/or the endpoints of analysis) and analytical procedures (Chi et al., 1994; Klein & Ragland, 2014; Sevian & Talanquer, 2014). Many times throughout history, emphasis has been placed primarily on the methods and utility of analysis while downplaying its conceptual meaning, which fundamentally lies in complex bodies breaking down into their constituent parts (Simon, 2002). On closer inspection, however, analysis and synthesis are likely the areas where theory and practice have been most closely linked throughout chemistry’s history (Klein & Ragland, 2014).
Chemical analysis has always been connected to the knowledge about matter and chemical substance. The answer to the fundamental question of analysis “What is the world and matter around us made of?”, i.e., the last point that analysis can reach (Weisberg et al., 2019), was determined by the current understanding of matter in a certain historical period. In antiquity, certain philosophers, such as Thales and especially Aristotle, answered this question using fundamental material and observable entities: earth, water, air, and fire (Curd, 2007; Klein & Ragland, 2014; Weisberg et al., 2019), whereas others, such as Leucippus and Democritus, employed an “atomic theory” (Weisberg et al., 2019). Later, conceptions of matter such as the principles sought by alchemists affected ideas about chemical analysis (Klein & Ragland, 2014; Leicester, 1971). Since Lavoisier’s time, with the development of chemistry and atomic theories of matter, elements have been viewed as the endpoint of analysis which has different properties than the material under analysis (Simon, 2002; Weisberg et al., 2019). As chemistry and technology have advanced, it has been possible to target nuclei and subatomic particles (Weisberg et al., 2019) as well as to collect large amounts of data and multidimensional information about entities through instrumental analysis (Karayannis & Efstathiou, 2012; Zolotov, 2020).
Additionally, the characterization of analysis as chemical is unrelated to the methods and techniques employed (Zolotov, 2020) to determine the chemical composition of materials and substances in terms of both quantity and quality (Skoog et al., 2007). The word “chemical” refers to the chemical entities that comprise the last point of analysis, namely, principles, elements, atoms, ions, parts of molecules, or molecules (Klein & Ragland, 2014; Zolotov, 2020), which are all the chemical entities discovered and known even since antiquity. Interestingly enough, it was not uncommon for antiquity’s philosophers or for academics of the nineteenth and twentieth centuries to overlook practical analysis (e.g., analysis of mineral waters, metallurgy techniques) (Simon, 2002).
Chemical analysis is of particular importance as a core, fundamental, and invaluable component of chemistry (Hudson, 1992; Breslow, 1997; Karayannis & Efstathiou, 2012), and chemistry relies on analysis both as the system of knowledge we discussed in the previous paragraphs and as a field of professional work (Vershinin & Zolotov, 2009). Because of its many applications, chemical analysis is a highly demanding field that employs a large number of individuals. In fact, substances can never be used for a specific purpose without first being analyzed to determine their components (Szabadváry & Robinson, 1980). Analytical methods are always required when examining a novel material, whether it is organic or mineral-based, or when examining the products of a chemical reaction (Hudson, 1992; Breslow, 1997; Karayannis & Efstathiou, 2012). Process control and high-tech manufacturing procedures are also examples of the application of chemical analysis (Cammann, 1992).
Chemical analysis is also used to address society’s problems concerning both the entities that are present and the analytical procedures used to detect those entities in a variety of contexts, such as economy, trade, detection of essential trace elements, nutrition, environmental protection, fuels, agriculture, defense, safety, health, quality control of medical supplies, biomedicine, forensic chemistry, food quality and safety, archeology, space science, ethics, health care, and industrial processes (Karayannis & Efstathiou, 2012; Zolotov, 2020). Apart from chemists, chemical analysis is a useful and familiar concept to other scientists, such as physicists, geologists, health scientists, and environmental scientists (Skoog et al., 2007).
Additionally, the concept of analysis is characterized by a worthwhile polysemy. The oldest definition of analysis is mathematical, which refers to “the resolution or the simplification of a whole into its parts” (Simon, 2002), which is similar to the philosophical decompositional conception of analysis (Beaney & Raysmith, 2024). The concept of analysis is applied in distinct scientific fields, like statistical analysis, data analysis, mathematical analysis, system analysis (in computer science), psychoanalysis, natural philosophy, epistemology, and the dissection of the human body (Simon, 2002). In most of these domains, analysis means breaking down a complex whole (concept, material, etc.) into simpler parts. Also, chemical analysis is a concept broadly used in everyday life, both consciously and unconsciously and either imparting a scientific content (e.g., “the analysis of liquid waste showed high levels of drugs”) or not (e.g., “he analyzed the event thoroughly”), while the phrase “in the final analysis” indicates a primary cause of a phenomenon or situation (Cambridge University Press & Assessment, n.d.). In fact, chemical analysis is frequently referred to simply as just “analysis” in the press, allowing the public to associate it with whichever meaning they are more comfortable with. This diversity of notions can also be observed in a science classroom, due to the fact that students are not always familiar with the chemical analysis concept, which, as a complex concept, is connected with a variety of entities and procedures. So they often just follow a “recipe” for performing an experiment of chemical analysis without understanding exactly what they are doing (Tan et al., 2002).
From the above, it is deduced that the concept of chemical analysis meets the prerequisites that are required for the construction of a conceptual profile model associated with it. The purpose of this paper is to suggest a preliminary classification of various ways of thinking about chemical analysis based on historical and epistemological sources (sociocultural domain) and students’ alternative conceptions (ontogenetic domain), as part of our research project aiming at developing a conceptual profile model of chemical analysis.
2 Theoretical Framework
The fundamental assumption of the conceptual profile framework is that individuals think in various ways and apply different ways of thinking in various contexts (Mortimer et al., 2014a). A conceptual profile model of a given concept consists of zones, which portray the different ways of thinking about the concept (Mortimer et al., 2014a), so it can be used as a methodological tool in order to characterize the heterogeneity of students’ thinking in a science classroom (Mortimer et al., 2014b). According to the theoretical-methodological grounds of the conceptual profile framework and to Wertsch’s interpretation of Vygotsky’s work (Mortimer et al., 2014b), building a conceptual profile model includes taking into account a wide range of meanings that can be given to a concept in all of the contexts in which the concept makes sense, which suggests that at least three of the genetic domains that Vygotsky proposed should be considered, namely, the sociocultural, ontogenetic, and microgenetic domains (Mortimer et al., 2014b). Through research in these domains, distinct ways of thinking are defined, established, and stabilized by ontological, epistemological, and axiological commitments about their meaning (Mortimer et al., 2014b). Each way of thinking about a concept corresponds to a particular way of speaking about it (Aguiar et al., 2018; da Silva Costa & dos Santos, 2022).
The conceptual profile framework, as developed by its pioneers, is founded on the following assumptions: (a) heterogeneity in thinking for a particular concept is found both in the population and in individuals and, (b) when it comes to data analysis, ways of speaking are regarded as indicative of ways of thinking (da Silva Costa & dos Santos, 2022; Mortimer et al., 2014a).
To be more precise, people have unique conceptual profiles for various concepts which are defined by the different weight given to each zone. Such differences originate from the person’s background, which provides different levels of potential for using each zone in different circumstances (Mortimer et al., 2012) and making sense of the concept in specific contexts (Orduña Picón et al., 2020). Due to their exposure to comparable social and cultural circumstances, different people might share similar ways of thinking and speaking (Mortimer et al., 2012), known as supra-individual systems of thinking and speaking (Orduña Picón et al., 2020).
Furthermore, one concept can have two or more distinct meanings that can be revealed via speech in suitable contexts. After all, science itself is not a homogeneous body of knowledge and can offer several perspectives on the same event (Mortimer & Amaral, 2014). Influenced by the dialectical grounds of Vygotsky’s thinking, concept learning is a dynamic process that entails a dialectical relation between an internal and an external plane of functioning. The conceptualization process shifts inwards when an internal plane of functioning is constructed by dialectical interaction with an external plane of social interactions and outwards when one employs learned concepts to understand experiences. Interaction with others is critical in both cases (El-Hani et al., 2023; Mortimer et al., 2014b). Additionally, everyday speech may sometimes employ meanings of scientific terms that are inconsistent with scientific meanings. This means that one or more ways of thinking that are incompatible with the scientific ones may be present in a person’s conceptual profile (Mortimer et al., 2012).
In terms of methods of instruction and learning objectives, from the perspective of the conceptual profile framework, learning science means further developing students’ conceptual profiles and making them aware not only of their possession of the conceptual profiles (Mortimer et al., 2012) but also of the contexts in which the scientific way of thinking is the most appropriate choice (Aguiar et al., 2018; Mortimer et al., 2014b). Particularly, the relative significance of each zone could change as a result of enhancing students’ conceptual profiles by supporting their understanding of scientific ways of thinking, which are typically inaccessible to them in some way other than school (Mortimer et al., 2012). Following that, educators help students in the metacognitive process of becoming conscious of their own unique ways of thinking as well as the principles or standards that can influence their selection of viewpoints to solve certain problems, i.e., which ones are appropriate for each context (El-Hani et al., 2014). Hence, the purpose of science education is not to replace all other ways of knowing (Mortimer et al., 2012) but to help individuals realize that each way of thinking and speaking is not equally strong in addressing a particular problem (Aguiar et al., 2018; Mortimer et al., 2014b).
3 Methodology
According to Mortimer et al. (2014a), for a concept to be thoroughly studied, specific ways of thinking about the concept (zones) are determined, and a conceptual profile model is created. Due to the fact that the zones of a conceptual profile model depend on the sociocultural context studied, the concept should be examined in a variety of contexts in which the concept makes sense (Orduña Picón et al., 2020). Data should be collected from different sources and not in a linear but in a dialogic way, in the sense that all groups of data are simultaneously in interaction with one another (Mortimer et al., 2012). Sources that can be used are the following: (a) secondary literature related to the history of science and epistemological research on the concept, in order to achieve an understanding of the sociocultural genetic domain of the concept, the difficulties and changes in the way of thinking about the concept, and the effect of these changes; (b) literature on alternative conceptions combined with data from classroom teaching and learning, which facilitate the investigation of the ontogenetic domain and, specifically, how the concept is learned and developed, thus representing the process of knowledge construction in everyday life; and (c) authentic/primary data collected by interviews, questionnaires, or video recordings of interactions in a variety of contexts and environments where the concept has a meaning, such as science classrooms, so as to investigate both students’ alternative perspectives (ontogenetic domain) as well as the micro-processes that occur during interactions in short periods of time in these environments (microgenetic domain) (da Silva Costa & dos Santos, 2022; Mortimer et al., 2012, 2014b).
In this study, we present our findings from the exploration of the concept of chemical analysis within the sociocultural and the ontogenetic domains. An inductive qualitative analysis approach was adopted to analyze data obtained from these two genetic domains. Based on how chemical analysis is viewed, the various ideas and definitions of the concept detected through the investigation of the sociocultural and ontogenetic domains were classified into expanded categories which represent the different ways of thinking about chemical analysis (Fig. 1).
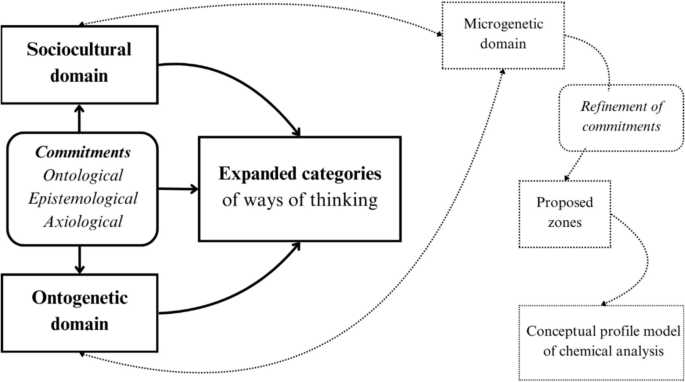
The investigation of sociocultural and ontogenetic domains along with the identification of commitments will result, after refinement through the exploration of the microgenetic domain, in the determination of the zones of the proposed conceptual profile model of chemical analysis. The steps that are not presented in this paper are indicated by the dotted lines and boxes
More specifically, the concept of chemical analysis was initially studied within the sociocultural genetic domain through investigation of pertinent secondary historical and epistemological literature. Data collection was conducted via search in the Web of Science, Scopus, and Google Scholar databases using the following keywords: “chemical analysis,” “chemical identity,” “chemical properties,” “chemical substance” combined with the keywords “history,” “philosophy,” and “epistemology.” After a thorough examination of the results and of relevant publications indicated by their bibliographical references, we chose a total of 51 of them to constitute the data of the sociocultural domain. Our selection criterion was the coherence of publications with the history and/or epistemology of chemical analysis, as indicated by the title or by reading the abstract and conclusion. Some of the publications analyzed were retrieved and studied in a later stage when we needed to clarify a specific historical or epistemological aspect that was troubling us. The majority of the sources refer to history and philosophy of chemistry in the Occident. We did not limit our search period in order to find as many old and new sources as possible, so the years of publication of our sources range from 1897 to 2024.
The following are some examples of the body of secondary historical and epistemological literature, starting with books about the history and epistemology of chemistry (Brock, 2016; Hudson, 1992; Leicester, 1971; Partington, 1989; Szabadváry & Robinson, 1980). We also studied conference proceedings about the history of chemical analysis (Belcher et al., 1977) and historical and epistemological research on the evolution of chemical analysis (Baiulescu, 2000; Kim, 2014; Pérez-Bustamante, 1997; Siegfried, 1988; Simon, 2002; Slater, 2002; Štulík & Zýka, 1992; Valcarcel, 1992; Vershinin & Zolotov, 2009; Zolotov, 2020; Zuckerman, 1992). Some dictionaries were also used, as well as Analytical Chemistry textbooks (Skoog et al., 2007).
The same procedure (i.e., search of databases and selection of the resulting publications) was followed to gather the data regarding the ontogenetic domain, which refers to research about students’ thinking (Mortimer et al., 2014a). A total of 35 publications constituted the data set of this domain. In this case, we used the presence of descriptions of students’ ideas, alternative conceptions, and ways of thinking about chemical analysis as our criterion for including a publication in the analytical corpus. For example, we could mention research papers on students’ thinking and alternative conceptions about chemical analysis from the research groups of Tan et al. (2002), Doménech-Carbó et al. (2009), and Milligan and Wink (2024). Again, we did not limit our search period in order to find as many old and new sources as possible, so the years of publication of our sources range from 1983 to 2024.
It should be pointed out that due to the fact that the literature regarding students’ misconceptions about chemical analysis as a whole was found to be limited, some of the above-mentioned publications pertain to research about students’ alternative conceptions on specific aspects of chemical analysis, such as chemical identity and chemical substance (Krnel et al., 1998; Liu & Lesniak, 2006; Mas et al., 1987; Ngai & Sevian, 2017; Ngai et al., 2014; Orduña Picón et al., 2020; Stavy, 1991) and on chemistry in general (Garnett et al., 1995; Horton, 2007; Sevian & Talanquer, 2014; Taber, 2019; Talanquer, 2006). As argued in the introduction, chemical analysis has always been connected to specific ways of thinking about matter identity and chemical substance. The answer to the fundamental question of analysis “What is the world and matter around us made of?”, i.e., the last point that analysis can reach (Weisberg et al., 2019), was determined by the current understanding of matter in each historical period and is analogous to questions regarding chemical identity, such as “What types of matter are there?” and “What cues are used to differentiate matter types?” (Sevian & Talanquer, 2014). In fact, as mentioned earlier in this work, chemical analysis yields information about the chemical composition of the sample. Specifically, qualitative analysis reveals the chemical identity of the substances in a sample (Talanquer, 2019). Also, chemical identity theory is founded on the idea that every substance may be distinguished from other substances by at least one property (Ngai & Sevian, 2017) which is crucial for chemical analysis since ancient times. Due to the fact that the concepts of chemical identity and substance are related to the concept of chemical analysis, we argue that some of the students’ alternative conceptions may be shared among them. It is very important to clarify that we did not use these results as a whole; instead, we retained only the chemical analysis–related information, and for this reason, the contribution of the ontogenetic field is somehow restricted compared to that of the sociocultural domain.
The publications that comprise the analytical corpus of the sociocultural domain are presented accompanied by an asterisk (*) in the “References” section while those of the ontogenetic domain by two asterisks (**). In cases where a publication offered historical and epistemological information on the concept as well as data on students’ alternative perceptions, it was used as a source for both the sociocultural and ontogenetic domains, and it is presented as follows: (*/**).
We should mention that this preliminary classification of ways of thinking in a conceptual profile model is not only about categorizing extracts from the data (although it typically includes this stage). Instead, a researcher should probe deeper into the passages retrieved from the relevant literature and the respondents’ claims in order to interpret them in terms of their ontological, epistemological, and axiological commitments, which are not often clearly articulated either orally or in writing, and according to the theory’s pragmatist realist grounds, they are presumed to codetermine one another (Mortimer et al., 2012, 2014b). A conceptual profile model aims to represent possible genetic paths for the development of different meanings for a concept. Therefore, commitments have no hierarchical order (Mortimer et al., 2014a), and they should be seen from a dynamic perspective, i.e., that they simultaneously set limits (causing difficulties for the construction of new meanings) but also provide possibilities for expanding the meaning of a concept (Mortimer et al., 2012). In order to identify the ontological, epistemological, and axiological commitments that differentiate each separate way of thinking from others, the following questions were used, in accordance with Orduña Picón et al. (2020):
- 1.What kind of entities and/or analytical procedures does one commit to consider present when thinking about chemical analysis? (ontological question)
- 2.On what basis does one construct his/her knowledge about the entities and/or analytical procedures considered present when thinking about chemical analysis? (epistemological question)
- 3.How one evaluates and affectively judges the entities and/or analytical procedures considered present when thinking about chemical analysis? (axiological question)
The final result of the above-described process is the proposition of the expanded categories of ways of thinking making up a conceptual profile model of chemical analysis.
4 Results and Discussion
The history of chemical analysis is mainly related to the history of chemistry which can be divided into the following five periods: (a) prealchemy (until the fourth century AD), (b) alchemy (fourth to sixteenth centuries), (c) unification (sixteenth to eighteenth centuries), (d) quantitative laws (the first 60 years of the nineteenth century), and (e) the modern period (since 1870 until today), according to historians of science (Vershinin & Zolotov, 2009). These historical periods concern different ways of conceptualization of chemical analysis. Our qualitative analysis of the historical and epistemological sources is consistent with and extends these conceptualizations of chemical analysis. In addition, our analysis of students’ alternative conceptions and thinking about chemical analysis revealed analogies with historical conceptualizations (Liu & Lesniak, 2006). It is important to clarify that research in this work aimed to describe the genesis of the categories of ways of thinking that will eventually comprise a conceptual profile model of chemical analysis and not to create a detailed historical report (Mortimer & Amaral, 2014). It is also important to have in mind that analytical chemistry is a branch of chemistry with not as long a history as chemical analysis (Vershinin & Zolotov, 2009). Chemical analysis forms the theoretical foundation of analytical chemistry (Zolotov, 2020); however, the two concepts are not interchangeable.
The qualitative analysis of the sources from the sociocultural and ontogenetic domains resulted in the preliminary classification of ways of thinking in a total of six categories. These categories are listed in Table 1, accompanied by their corresponding ontological, epistemological, and axiological commitments. The main historical and epistemological findings (sociocultural domain) as well as students’ alternative conceptions (ontogenetic domain) which explain and justify each way of thinking are subsequently discussed. The categories are not numbered in order to show that there is no hierarchy between them (de Mattos, 2014).
4.1 Everyday Analysis
4.1.1 Ontological Commitment
According to historical and epistemological sources mainly referring to the pre-alchemical period (Leicester, 1971; Vershinin & Zolotov, 2009), chemistry from its beginning has been connected with analysis in the most part (Zuckerman, 1992). This primitive concept of chemical analysis was possibly based on the corresponding philosophical view of it, which compares chemical analysis to philosophical analysis in the light of isolating what is more elementary from what is more complex by any method (Beaney & Raysmith, 2024), influenced by Aristotle’s analysis of causation. Matter, as the last point of analysis, is dissolved in its tiniest particles (minima) during the process of diacrisis, while during the process of syncrisis, these particles are reorganized to create another type of matter (Brock, 2016).
Analytical practices were evolved independently of these theoretical ideas (Simon, 2002). Analysis of that kind undoubtedly preceded both chemistry as we know it today, and alchemy (Hudson, 1992) was often characterized as “art” (Partington, 1989; Simon, 2002) or “ars probandi” (art of assaying) (Belcher et al., 1977) and was not anticipated as science (Szabadváry & Robinson, 1980). Basic analytical procedures for isolation and separation of matter were also involved, mostly employing human senses (Leicester, 1971; Partington, 1989; Strathern, 2000), such as distillation, crystallization, evaporation, and filtration (Szabadváry & Robinson, 1980). Even though these methods have existed since antiquity, they were occasionally applied until the seventeenth century (Vershinin & Zolotov, 2009).
By posing the ontological question, we argue that, from a sociocultural perspective, the ontological commitment for this way of thinking includes views of matter that can be separated into its basic parts that can be isolated. These simple procedures involve the use of human senses.
In the ontogenetic domain, it is suggested that matter is frequently perceived by children as distinct classes of stuff, which are unique and different from one another (Orduña et al., 2020). Βy grouping substances into categories, students can utilize the characteristics or general properties of those categories to decide about substances’ chemical identity (Ngai & Sevian, 2017). Another interesting finding is the fact that, when students try to separate materials from one another, they tend to consider them as objects (“objectivization”), having properties mainly attributed to objects (such as size, shape) (Langley et al., 1987; Horton, 2007; Ngai et al., 2014). This description of matter, resulting from the undifferentiated object/matter notion, regards the components of matter as objects with all of their attributes. For example, many students believe that, given that gases lack mass, they also lack shape (both of which are extensive properties of objects). The same applies to intensive properties, as well (Krnel et al., 1998). Also, students often think that an entity that symbolizes a particular material is formed by the connection of extensive and intensive features. Some of these features are, for example, softness, thinness, brightness, and weight. However, for younger children, it might not be evident what qualities matter the most for identifying an object. In fact, even non-material entities (such as a shadow) may be conceptualized as objects in young children’s minds. Besides, density is perceived by them as a quality that is contingent upon the object’s extensive properties (Krnel et al., 1998). According to this viewpoint, the ontological commitment is broadened by including the above-mentioned perceptions about entities that are considered as the endpoint of analysis.
Thus, people who think of chemical analysis as “everyday analysis” are ontologically devoted to the idea that matter is classified into categories or types of stuff and objects. Matter can be separated into its basic parts with simple procedures mostly employing human senses.
4.1.2 Epistemological Commitment
In the sociocultural domain, color, taste, and smell are examples of classificatory attributes that have historically been the focus of chemistry, and chemical models have placed great emphasis on these qualitative characteristics of matter. Even though modern chemistry includes mostly quantitative and dynamic concepts, these concepts are frequently combined with the above-mentioned descriptive ones (Erduran, 2001). Matter was characterized and described only by properties such as color, hardness, uniformity in appearance, weight/density, brightness, temperature (hot or cold), dryness/humidity, and solubility, without their scientific content as we understand it nowadays (Curd, 2007; Leicester, 1971).
Methods using these properties have been known as organoleptic methods (Vershinin & Zolotov, 2009). Actually, the isolation and separation of matter and its parts were based on direct observation employing the use of human senses (Leicester, 1971; Partington, 1989; Strathern, 2000). Αt these early stages of chemical analysis, instinct, skills, practice, and experience were very much required. Procedures were developed independently of theoretical ideas and were not necessarily conducted in a specific and organized space (i.e., laboratory). Knowledge regarding both matter (endpoint of chemical analysis) and analytical practices was not recorded, and instead, it was verbally passed down from generation to generation (Leicester, 1971).
Subsequently, answering the epistemological question, in the sociocultural domain, knowledge about chemical analysis is constructed via firsthand observation of matter through the employment of the senses (smelling, seeing, touching, etc.), not necessarily in a laboratory and without reference to theoretical concepts. Furthermore, instinct, talent, practice, and experience seem to be really crucial.
The ontogenetic domain’s findings stabilize and refine the epistemological commitment that was first identified through the sociocultural domain’s data analysis. A large amount of data on students’ alternative conceptions about chemical analysis emphasizes the use of senses, as well. Novice learners make decisions on the chemical identity of matter influenced by appearance, perceptual and macroscopic characteristics, and surface similarity (Horton, 2007; Liu & Lesniak, 2006; Ngai et al., 2014). Smell, taste, color, weight, volume, texture, shape, hardness, and tangibility are among the most common explicit properties they use that are detectable by the senses (Stavy, 1991; Ngai & Sevian, 2017; Orduña et al., 2020). Also, when attempting to separate matter, learners frequently place a lot of attention on attributes that are primarily linked to objects (such as size, form, mass, weight, softness, thinness, brightness) (Langley et al., 1987; Krnel et al., 1998; Horton, 2007; Ngai et al., 2014). All these assumptions are powerful cognitive constraints since they cause students to believe that surface similarity and object-related properties may be indicative of materials’ inner structure (Ngai et al., 2014). Consequently, learners assume that the analysis of a given material includes only the recognition of these salient features, and thus, it is argued that their thinking about chemical analysis may be limited to that recognition. It is worth mentioning that the role of instinct, skill, ability, and experience in chemical analysis, which was detected in the sociocultural domain, was not identified in research on students’ thinking; therefore, we refrain from including it in the epistemological commitment.
Thus, we propose that, incorporating ontogenetic domain’s data, the epistemological commitment of this way of thinking states that, when considering chemical analysis as “everyday analysis,” the basis on which people construct their knowledge about matter is direct observation and use of senses (smelling, seeing, etc.) so as to perceive its explicit or object-related properties. These simple organoleptic analytical procedures are employed independently of theoretical ideas about the entities-endpoints of analysis and not necessarily in a laboratory.
4.1.3 Axiological Commitment
According to the history and epistemology of that period, initially, the concept of chemical analysis itself was not as important as its application and outcomes. The analysis was used to satisfy daily and professional needs relevant to ceramics and metals, such as gold and silver (Leicester, 1971; Ruthenberg & Mets, 2020; Vershinin & Zolotov, 2009; Zolotov, 2020) and especially mining and cupellation (the removal of lead in an ore by oxidation) (Brock, 2016; Szabadváry & Robinson, 1980), leather, fabrics, soaps, cosmetics (Szabadváry & Robinson, 1980), and natural pigments (Partington, 1989; Zolotov, 2020).
From the viewpoint of the sociocultural domain, this is associated with the axiological commitment that, in order to meet their everyday and professional demands, people analyze only those materials and use only those analytical procedures that they feel advantageous, such as the ones concerning metallurgy or cosmetics.
Data from the ontogenetic domain shed additional light on the evaluation of the entities under consideration in this way of thinking. To ascertain matter’s chemical identity and whether it has altered, learners often consider its source or typical environment, purpose, or function (Ngai & Sevian, 2017). Younger children’s criteria are mostly limited to simple changes or actions (e.g., it can be bent or it produces sound), while older children consider more complicated and valuable actions or functions, which may show that an object is exemplar of a larger group, e.g., edible materials, construction materials, art materials, and cleaning materials. Also, it is not uncommon for students to identify or classify matter by just considering popular examples of materials widely utilized in our daily lives, such as sugar, glue, wood, iron, clay, and plasticene (Krnel et al., 1998; Stavy, 1991). Hence, the axiological commitment is enriched in its component of the object of analysis, namely, the evaluations and judgments about the entities that individuals find helpful in their daily and professional life and, thus, analyze.
Therefore, considering data from sociocultural and ontogenetic domains in a dialogic way, as described above, the axiological commitment of the way of thinking about chemical analysis as “everyday analysis” is as follows: individuals think that only the materials and objects that they find useful in their daily and professional lives could be analyzed, and this utility lies in the material’s entities origin, habitat, purpose, function, and similarity to valuable exemplar materials. Also, they employ only the analytical procedures they believe to be beneficial, such as those involving metallurgy and cosmetics.
4.2 Alchemical Analysis
4.2.1 Ontological Commitment
According to historical and epistemological data on the very long period when alchemy dominated, primarily in Alexandria, Arabia, China, and later in Europe, during this period significant contributions were made to the discovery and isolation of new substances and elements, the use of reagents, the employment and recording of isolation and synthesis recipes (often too symbolic), and the development of laboratory apparatus (mostly distillers) (Leicester, 1971; Strathern, 2000).
In terms of entities that underlie chemical analysis, until the Middle Ages, alchemists were based on the philosophical views of Thales, Empedocles, Plato, and Aristotle (Pérez-Bustamante, 1997) about the four elements that make up the world around us (Leicester, 1971; Ruthenberg & Mets, 2020; Szabadváry & Robinson, 1980) (or the five elements and two contraries for the Chinese (Kauffman, 1985; Leicester, 1971)). Alchemists argued that the properties of a material were due to the balance of these elements in it (Strathern, 2000; Szabadváry & Robinson, 1980), and therefore, they were often in quest of these elements. Others believed in the “three scholastic principles”, namely, sulfur, mercury, and salt, of which everything consisted (de Berg, 2019). Also, according to the “dualistic sulphur-mercury” alchemical Arabian theory (Kauffman, 1985; Klein, 2007; Pérez-Bustamante, 1997), mercury and sulfur were regarded as the primary substances, the appropriate analogies of which may provide a material incorruptibility (Brock, 2016; Klein, 2007; Szabadváry & Robinson, 1980). The Aristotelian four (or Chinese five) elements, the three philosophical principles, and the primary substances were in fact the essence of a material (for example, what makes gold to be gold), which was the basic entity that endures despite change (“essentialism”) (Hacker, 2004; Shields, 2023).
Regarding analytical procedures, alchemical analysis included simple analytical procedures (isolation, separation, and purity control) (Leicester, 1971; Partington, 1989; Strathern, 2000). Certain procedures were developed or evolved, such as distillation (fire analysis), filtration, and dissolution (Pérez-Bustamante, 1997; Strathern, 2000). The understanding of elements and principles of matter was deeply connected to the knowledge of separation, purification, and combination procedures (Klein & Ragland, 2014).
The foregoing can guide us to establish an ontological commitment to thinking about chemical analysis as “alchemical analysis.” More specifically, entities that constitute the basic essence of materials (“elements”, “principles”, “substances” or “contraries”) are analyzed through specific yet simple isolation and separation procedures.
The ontological commitment pertaining to the entities under analysis is stabilized by research in the ontogenetic domain. Indeed, many students think that matter has an “underlying quality” or “inherent essence” that determines materials’ identity (Forbes, 2017; Newman & Knobe, 2019; Talanquer, 2006) and provides them the potential to be separated. The theory that some objects or materials in general have specific essential features can be generally described as “essentialism” (Robertson & Atkins, 2023). Essentialism can be defined as the impulse to express certain ideas in terms of a more profound, imperceptible characteristic and provides individuals with a subconscious knowledge of key scientific ideas, including the unnoticeable similarities between members of a category, which are more than just apparent characteristics but involve a deeper and more subtle essence (Newman & Knobe, 2019). Furthermore, students often think that the “inherent essence” and therefore the identity are lost when a change results in a transformation into something invisible or “non-material” (such as gases, heat, or light) (Mas et al., 1987; Talanquer, 2006).
Therefore, analysis of secondary data from both genetic domains agrees on the ontological commitment of this way of thinking, namely, that alchemical analysis involves entities (“elements,” “principles,” “substances,” or “contraries”) that constitute the essence of materials. Based on these entities, specific yet simple isolation and separation procedures are designed and used.
4.2.2 Epistemological Commitment
The construction of knowledge about the above-mentioned elements and principles that are considered present during analysis is deeply influenced by the background of alchemy. The sociocultural domain suggests that alchemy combines three distinct approaches—Egyptian technology, Eastern mysticism, and Greek philosophy—to explain the entire world as a single, comprehensive system. Alchemy is therefore not wholly independent of theory; rather, there is a connection between theoretical and practical chemistry (Leicester, 1971). Of course, this does not imply that we refer to a unified theoretical framework and an unbiased application of theory, as it is not unusual for theory to be modified in whole or in part to fit the results or even be totally ignored (Leicester, 1971; Newman, 2011; Read, 1933). Also, it is worth noting that the primitive classifications of metals, gases (spirits), acids, medications, etc. proposed by some alchemists, although not named and without their scientific content as we understand it today, were utilized for many centuries and formed an initial basis for further classifications (Leicester, 1971; Szabadváry & Robinson, 1980).
Alchemical analytical procedures were based on direct observation using senses and were partially supported by the alchemists’ theories and classifications (Leicester, 1971; Partington, 1989; Strathern, 2000). Imagination, instinct, skills, practice, and experience were also considered as necessary (Bolton, 1897; Kim, 2000; Leicester, 1971; Newman, 2011; Szabadváry & Robinson, 1980), and this is why alchemists were characterized as “artists” too (Bolton, 1897; Hopkins, 1925; Newman, 2011). It is remarkable that alchemists were not always aware of what they were discovering (Leicester, 1971; Strathern, 2000), and their predominant assumption in carrying out these analytical procedures was that frequent repeating of a procedure leads to continuous refinement of it, resulting in the anticipated experimental result (Leicester, 1971). Moreover, technology was employed, and rudimentary laboratories were established (Read, 1933; Strathern, 2000).
So far, the sociocultural domain has provided us with some insights into the epistemological commitment of the way of thinking about alchemical analysis, which means that knowledge about the entities and procedures of chemical analysis is constructed by modifying, partially using, or independently of theory, along with a primitive categorization of entities under analysis. Procedures of direct observation using the senses are carried out repeatedly in order to achieve the desired outcome (sometimes lacking awareness of the discoveries), usually in a laboratory setting.
Once more, the ontological domain provided some insight into the characteristics of entities that are the last point of analysis. The “essentialism” of novice students actually implies that there are imperceptible commonalities among members of a category, namely, their essential properties. These similarities go beyond mere appearances and entail a more subtle and profound nature (Newman & Knobe, 2019; Robertson & Atkins, 2023). This essence defines and distinguishes materials from others (Forbes, 2017; Newman & Knobe, 2019; Talanquer, 2006), and it also establishes materials’ eligibility for a category (Newman & Knobe, 2019). These essential attributes appear to exist independently of the materials and regardless of any physical or sensory changes. For instance, even though silver turns dark when it comes into contact with air, it remains silver (Nieswandt, 2001). Also, “additivity” perceptions of students about these properties, such as assumptions that the properties of a mixture are the average properties of the “principles” or “elements” which it is composed, are not uncommon (Ngai et al., 2014). The epistemological commitment is thus stabilized in its component about the entities considered present in materials during analysis. Furthermore, the “substantializing” model may also influence students’ thinking of substances as components. For example, heat is thought to possess the characteristics of matter or substance and to be able to reside in a body or move between bodies like a fluid (caloric-like notions of heat) (Horton, 2007; Taber, 2019).
Hence, knowledge is constructed using senses partially by direct observation of essential and additive properties of materials which define and differentiate them from each other, along with a primitive categorization of entities. There is often a lack of awareness of some of the “experimental” findings and theoretical ideas are manipulated and sometimes partially used or even not used at all. Constant repetition of procedures seems crucial in order to get the desired outcome.
4.2.3 Axiological Commitment
The philosophical, mystical, spiritual, and supernatural background of alchemy, which is its inheritance from Greek philosophy, Egyptian technology, and Eastern mysticism (Leicester, 1971), had a significant influence on the way of thinking about chemical analysis as alchemical analysis (Bolton, 1897; de Berg, 2019; Leicester, 1971; Partington, 1989; Pérez-Bustamante, 1997; Ragai, 1992; Read, 1933, 1995). For example, the purposes of alchemical analysis differed significantly from those of practitioners (e.g., metallurgists), since the former aimed at the discovery of the philosopher’s stone, the creation of the elixir of immortality, and the transformation of all metals into gold (Hopkins, 1925; Kauffman, 1985; Leicester, 1971; Ragai, 1992; Read, 1933; Strathern, 2000).
Without a doubt, alchemy contributed greatly to the development of chemistry in general and chemical analysis in particular, as a large number of compounds and elements were discovered during this period and many analytical methods of isolation and separation were developed (Leicester, 1971; Strathern, 2000). Indeed, alchemists are credited with preserving and developing alchemy until it was systematized and evolved into the science of chemistry (Leicester, 1971). Nonetheless, judgments and attitudes toward alchemical analysis differ depending on the time and societal acceptability of alchemical activities and were mainly based on magical and mystical thought that were integral parts of alchemical culture (Fors, 2022). At the same time, within the framework of the natural philosophy of the era, the transformation of common metals into gold—chrysopoeia—was a rational pursuit. Therefore, judging alchemy as purely superstitious, irrational, and deceptive (Bolton, 1897; Partington, 1989; Strathern, 2000) is an unfair and historically flawed perspective (Fors, 2022).
Therefore, the axiological commitment that stabilizes this way of thinking relates to the opposite perspectives (positive and negative) on the contribution of alchemical analysis to the development of chemistry as well as the influence of the mystical, spiritual, and philosophical background of alchemy in the purposes of analysis.
In the ontogenetic domain, while alchemical analysis has ceased to concern the scientific community from a certain period of time onwards, remnants can still be found in ways of thinking about chemical analysis, synthesis, and transformation. Like alchemists who believed that changing living and non-living matter includes similar entities and procedures, the lines separating the various fields of human experience and knowledge are often blurred for students (Freire et al., 2019). It should also be noted that nowadays it is not uncommon for children to hold alternative conceptions on chemistry, such as that it is an alchemical pseudo-science and sorcery, while chemists are often portrayed as mad scientists (Schummer et al., 2007), which may make students doubtful and suspicious of all chemicals. In contrast, it is not rare for some chemists to be labeled as magicians (Laszlo, 2006), implying perhaps a more favorable attitude of young learners toward the field.
Consequently, integrating information that is in agreement from both genetic domains, we conclude that the axiological commitment of the way of thinking about chemical analysis as “alchemical analysis” is related to evaluations and beliefs, influenced by the origins of alchemy, about the entities and procedures that fuse the different domains of human experience and knowledge. Attitudes toward alchemical analysis vary, from positive (attractive, important due to its contribution to the study of substances and the development of analytical procedures) to negative (suspicion about its purposes and results).
4.3 Empirical Analysis
4.3.1 Ontological Commitment
Historical and epistemological data suggest that in the eighteenth century, apart from compounds, the endpoints of analysis were elements as well (elementary analysis) (Simon, 2002). Also, a distinction was established between one kind of substance (pure substance) and two or more kinds of substances (mixture), as components of the surrounding world (Klein, 2012). Nevertheless, the lack of a comprehensive theoretical framework (Leicester, 1971; Siegfried, 1988; Strathern, 2000) often led to conceptual misunderstandings. For instance, concerning entities as structural units, “chemical principles” (i.e., the tangible materials discovered by chemical analysis) were distinct from the supposedly “natural elements” that were posed philosophically (Kim, 2000).
In terms of processes, from a certain time onwards, chemical analysis procedures were related to simple experiments (Leicester, ; Partington, 1989; Strathern, 2000; Vershinin & Zolotov, 2009). By using fire (distillation) or by solvent analysis (using mostly aqueous solutions) chemical analysis intended to reduce matter into its most basic components (Kim, 2000; Simon, 2002). In particular, analysis aimed at the determination of the composition of objects or solutions with different properties and/or the concentration of elements and compounds as their constituents using simple titrimetric or gravimetric techniques (Siegfried, 1988; Karayannis & Efstathiou, 2012). The use of solutions truly represented the advent of qualitative analysis (Kim, 2014; Szabadváry & Robinson, 1980). In order to be helpful in this case, analysis should either assist in the preparation of these solutions or in the identification of their components (Simon, 2002).
From a sociocultural perspective, the ontological commitment of thinking about chemical analysis as “empirical analysis” relates to the components of objects or solutions, namely, elements and compounds, which can be determined by simple experiments.
Data from the ontogenetic domain suggests that, instead of thinking about elements and compounds as just labels for different types of objects and materials, students sometimes believe that they are the fundamental components of everything in the world (Ngai et al., 2014; Stavy, 1991), without completely distinguishing elements and chemical compounds (Stains & Talanquer, 2007). Nonetheless, when it comes to this simplistic model of matter composition, our study of the literature on chemical identity of entities showed that conceptual misunderstandings are analogous to those of scientists in the past, as students frequently misinterpret how scientific terminology is used in everyday contexts. For instance, in common parlance, a “particle,” as a component of matter, is a tiny yet observable fragment of solid matter (Garnett et al., 1995). Therefore, the ontological commitment of this way of thinking is stabilized in terms of entities considered present during analysis.
Data from both domains suggest that people who think of chemical analysis as “empirical analysis” are ontologically committed to the idea that the endpoints of analysis are the elements and compounds from which materials, objects, and solutions are composed. Elements and compounds can be determined by simple experimental procedures.
4.3.2 Epistemological Commitment
In the sociocultural domain, chemistry gradually separated from physics and had its own primitive theoretical constructs. Following that, chemical analysis aimed at scientific development and simultaneously relied on it (Kim, 2014; Strathern, 2000). It is worth mentioning that some scientists, such as Lavoisier, made a significant contribution to experimental chemistry through their practical, pragmatic, and operational description, categorization, and recording of the components of matter (Pérez-Bustamante, 1997; Simon, 2002). Additionally, the fact that “the whole is more than the sum of the parts” (Luisi, 2002; Simon, 2002) was observed in the case of compounds and their elemental components, because a compound (a group of elements) has characteristics that none of the single elements does, namely, emergent properties (Simon, 2002; Weisberg et al., 2019).
Certain aspects of the scientific method appeared in the procedures. For example, the concept of experiment was consciously introduced, for which the word “test” was initially used (Kim, 2014; Strathern, 2000). It is suggested that the simple experiments for the determination of the components of matter were mostly carried out in laboratories and vaguely recorded (Leicester, 1971; Strathern, 2000). Empirical analysis differs from everyday and alchemical analysis in that procedures were systematized and evolved and relied on theory (Kim, 2014; Strathern, 2000). However, intuition, skills, practice, and experience remained quite important (Siegfried, 1988; Karayannis & Efstathiou, 2012), while results were still in some cases based on direct observation through the employment of senses (Belcher et al., 1977; Leicester, 1971; Strathern, 2000).
From a sociocultural point of view, the epistemological commitment of this way of thinking is that the process of knowledge construction about the entities with emergent properties being analyzed includes an operational categorization of elements and compounds as well as experiments which are conducted in laboratories and rely on theories and the scientific method.
Data from the ontogenetic domain focus more on the process of knowledge construction about the components of matter. Novice learners struggle to understand and predict the presence of emergent properties in chemical systems. Therefore, when considering chemical compounds or elements, most inexperienced students use an additive framework. Also, the coexistence of additive and emergent explanations is not unusual (Talanquer, 2008). Students have difficulty employing emergent explanations of matter instead of additive ones (Chi, 2005).
Thus, combining data from both genetic domains in a dialogic way, thinking of chemical analysis as “empirical analysis” is constructed based on theories and operational categorizations of components (elements, compounds) of matter, with some of those (compounds) having emergent properties, as well as on simple experiments involving aspects of the scientific method and carried out in laboratories.
4.3.3 Axiological Commitment
Data from the sociocultural domain indicate that many scientists, such as Boyle, were deeply concerned about the accuracy and validity of chemical analysis (Kim, 2000; Strathern, 2000). Therefore, compliance with the scientific method was considered necessary, in the sense that events should be tested, broken down into their components, and measured. Only then would it be possible to compare and combine similar events, and one should make generalizations (Strathern, 2000). In order to articulate the axiological commitment of this way of thinking, these findings could be coupled with those of the ontogenetic domain that students sooner or later realize that, in some cases, perception is limited when it comes to recognizing or differentiating components of materials. As a result, they acknowledge that certain differentiating features of these components must be tested experimentally so that a decision can be made about their identity (Ngai et al., 2014).
Thus, the axiological commitment of the way of thinking about chemical analysis as “empirical analysis” lies in judgments and evaluations of the entities and analytical procedures based on theory and their compliance with the scientific method.
4.4 Classical Analysis
4.4.1 Ontological Commitment
As shown by historical and epistemological findings, the development of chemical analysis resulted in the clarification of many chemical concepts (Szabadváry & Robinson, 1980). Thus, following the Lavoisier era, people thought about chemical analysis in terms of entities such as elements, compounds, mixtures, and solutions (Simon, 2002; Szabadváry & Robinson, 1980). Furthermore, the definition of substance identity was based on its composition (atoms, molecules) (Schummer, 2002) that was represented by its chemical formula. Compounds were divided into groups according to their composition, such as salts, acids, alkalis, and gases (Szabadváry & Robinson, 1980; Simon, 2002; Weisberg et al., 2019).
Regarding analytical procedures, classical analysis involves simple experimental methods (Skoog et al., 2007), which either were guided by or aimed at substances’ composition, and both qualitative and quantitative information was obtained mainly on organic compounds (Slater, 2002), minerals, metals, mineral waters, and textile ingredients (Belcher et al., 1977; Coley, 1982). During qualitative analysis the composition of a sample was identified by means of discrete components reacting with particular reagents and followed by product identification, whereas during quantitative analysis the concentration of compounds or elements with a particular composition was determined using gravimetric or titrimetric techniques (Stock, 1977; Pérez-Bustamante, 1997; Skoog et al., 2007; Karayannis & Efstathiou, 2012; Klein, 2012). Classical gas analysis was barely used (Zolotov, 2020) as well as methods of microanalysis (Leicester, 1971). In general, the development of analytical methods in a specific field led to its further advancement (Leicester, 1971; Slater, 2002; Vershinin & Zolotov, 2009), thus making possible for chemists to address topics that were previously completely disregarded (Siegfried, 1988).
Therefore, from a sociocultural viewpoint, people who think about chemical analysis as “classical analysis” are ontologically committed to the presence of elements and compounds having specific composition (atoms, molecules), divided into groups and determined through simple experimental procedures, which can be qualitative or quantitative.
The ontogenetic domain also highlights the consequences of introducing the composition of elements and compounds, which is one of the main differences with the previously presented ways of thinking. There is research evidence that science and chemistry students make sense of classical analysis procedures as experimental methods that separate substances which have different chemical compositions (Orduña Picón et al., 2020). The composition and structure of matter appear to be notions that students, at least at the primary level, comprehend. Students do, however, sometimes believe that structures are just static groups of microscopic components (atoms, molecules, etc.) (Orduña Picón et al., 2020). Due to the fact that novice learners ignore the dynamic interactions between particles at the microscopic level, it is not uncommon for them to transfer properties from macro-level to micro-level in order to make sense of simple analytical procedures. For example, dissolving (micro-level) is often regarded as a type of breaking (macro-level) (Krnel et al., 1998; Liu & Lesniak, 2006). Likewise, it is not unusual for students to combine explanations of substantialisation with microscopic body structure: microscopic entities retain the macroscopic characteristics of their surroundings. For example, copper sulfate’s blue color is attributed to the presence of blue particles in it (Sanmarti et al., 1995).
Hence, the ontological commitment can be more effectively expressed by integrating the ontogenetic domain’s findings with those of the sociocultural domain, as follows: the way of thinking about chemical analysis as “classical analysis” involves elements and compounds with specific chemical composition (atoms, molecules) and static structures, which are divided into groups and determined through simple experimental procedures, which can be qualitative or quantitative.
4.4.2 Epistemological Commitment
History and epistemology of chemical analysis suggest that at the time the physical properties of materials and compounds were considered as important as the chemical ones (Leicester, 1971). In terms of entities, chemical composition was operationally connected to both experimental synthesis and analysis (Schummer, 2002). In addition, thinking about chemical analysis was often based on common properties and composition that facilitated grouping (Strathern, 2000). Therefore, the composition of a material and its corresponding properties enable the choice of the most suitable analytical procedure.
In terms of procedures, the dynamic integration of synthesis and analysis was exemplar, and chemical practices varied from those of physics (Ruthenberg & Mets, 2020). Simple experimental analytical methods were based on core chemistry concepts, several physical properties (color, odor, solubility in a range of solvents, boiling or melting point, optical activity or refractive index, etc.) and chemical reactions in solutions (Skoog et al., 2007; Vershinin & Zolotov, 2009) (oxidation/reduction, thermal decomposition, acid/base reactions, double replacement, etc.) (Karayannis & Efstathiou, 2012). A typical and particularly important application of chemical analysis was the interpretation of different isomers’ properties based on their structures, which were determined by analytical procedures (Leicester, 1971). In general, during the determination of the composition of a sample (qualitative analysis), the discrete components (elements or compounds) reacted with specific reagents, and their products could be identified by their physical properties. During the determination of the concentration (quantitative analysis) of specific entities with different properties, the mass of an entity or of one of its products (gravimetric techniques) or the volume of a standard reagent (titrimetric techniques) in the laboratory was measured (Stock, 1977; Pérez-Bustamante, 1997; Skoog et al., 2007; Karayannis & Efstathiou, 2012; Klein, 2012). Concurrently, error awareness was developed (Leicester, 1971).
It is worth mentioning that all chemists could benefit from the advancement of science. For instance, laboratories specifically for chemical analysis and applied chemistry instruction were developed as the starting point of chemical analysis training (Belcher et al., 1977). Also, the continuous improvement and development of experimental equipment as well as the clear and easily understandable way of recording in writing the elements and compounds undergoing analysis along with the analytical procedures undoubtedly played a part in this. Method records were shared through publications in journals edited by scientific societies, such as the Royal Society of London and the Academy of Sciences in Paris (Strathern, 2000), or published books (Baiulescu, 2000; Belcher et al., 1977), thereby becoming broadly accessible.
Based on the foregoing, we may suggest an epistemological commitment. To be more specific, thinking of entities and procedures of chemical analysis as “classical analysis” is built on core chemistry concepts as well as the physical and chemical properties of elements and compounds, which are determined by their structure, their compositional categorization which allows for the selection of the appropriate analytical method, and the possibility that the results of the analysis are accessible to all.
In the ontogenetic domain, students understand that chemical analysis is mostly based on the composition and properties of elements and compounds (Orduña Picón et al., 2020). Students’ ideas about composition and structure can guide them in choosing an appropriate chemical reaction or physical property to separate and identify a substance. For example, they sometimes believe that structures are static groups of microscopic entities (molecules, atoms, etc.) which result in fixed properties (Orduña Picón et al., 2020). When explaining and predicting the properties of matter, students place more emphasis on centralized causality over emergence and chemical composition over molecular structure (Talanquer, 2017).
These views of structure could influence their thinking about chemical analysis, with regard to the selection of the differentiating property or characteristic that separates a material from another and the criteria that help them categorize different materials based on how similar their structures are. For example, lots of students have a tendency to simplify a problem of identifying depicted elements or compounds with fixed structures based only on a single cue or property. The majority of inexperienced students typically make use of explicit characteristics (i.e., the quantity of atoms in chemical formulas) as opposed to implicit ones (i.e., the nature of chemical bonding) (Ngai et al., 2014).
However, what is perhaps most important is that when analysis ceases to be regarded as merely empirical practices and a comprehensive scientific background is introduced, a combination of procedural and propositional knowledge in addition to manipulating and inferential skills is required (Tan et al., 2001). Having this in mind, when learners try to determine a substance’s chemical identity, they sometimes suggest conducting specific experiments or measuring properties which they assume the substance should have (such as its boiling point) (Ngai & Sevian, 2017), greatly influenced by the assumption of simple linear causality even in the most complex interacting systems (Taber, 2019). Thus, they often put too much focus on experiments, which however may be disproportionate to their understanding and deepening of the corresponding knowledge (Tan et al., 2001). Due to the theoretical background of chemical analysis, there is also a chance that pupils do not comprehend what they are actually performing when working on practical assignments (Tan et al., 2001).
The epistemological commitment of this way of thinking is enriched by ontogenetic domain data mainly in terms of the properties of the entities under analysis, namely, the elements and compounds. Knowledge construction about the entities and procedures of chemical analysis as “classical analysis” is based on core chemistry concepts as well as the fixed and mostly explicit physical and chemical properties of elements and compounds along with a categorization based on their composition (structure–property relationships), enabling thus the choice of the most suitable analytical procedure. There is also a disproportionate emphasis on experimentation in comparison to theory and the potential that the results of the analysis are accessible to all, both in terms of the endpoints of analysis and the methods employed.
4.4.3 Axiological Commitment
In the sociocultural domain, we find that, because of the Chemical Revolution (Langley et al., 1987; Siegfried, 1988; Pérez-Bustamante, 1997), chemical analysis was included in an extensive theoretical framework of chemistry, as a separate scientific topic with a unified background and core concepts rather than just a compilation of discrete pieces of information (Strathern, 2000). More and more chemists believed that theory should be based on experiments and experimental data and should not be envisaged beforehand (de Berg, 2019; Strathern, 2000). Also, one of the results of the accumulation of this body of knowledge on chemical analysis was the establishment of the branch of analytical chemistry, as a distinct branch of chemistry presenting unique theoretical and methodological challenges of its own (Baiulescu, 2000; Zuckerman, 1992). From now on, for a method or technique to be employed, it should be aligned with the scientific framework of analytical chemistry (Strathern, 2000). Many analytical methods were applicable only to specific compounds, e.g., organic (Leicester, 1971; Slater, 2002). Societies agreed on international standards for analysis (Campbell, 1979), and even though absolute values were not determined and random empirical values were ascertained instead (Belcher et al., 1977), the accuracy, repeatability, and systematization of analytical procedures were increased (Strathern, 2000). Methods were less destructive than those that were largely in use before, such as distillation (Kim, 2014).
To formulate the axiological commitment of this way of thinking, we must consider the ontogenetic domain’s smaller but significant contribution to judgments and evaluative assessments of entities and procedures. In particular, data from students’ alternative conceptions on chemical analysis suggest that students consider analysis as an extremely difficult and tedious memory work based on abstract, intricate, and highly interconnected concepts about the elements and compounds, which are considered present in the materials under analysis (Tan et al., 2001).
Therefore, the axiological commitment of the way of thinking about chemical analysis as “classical analysis” includes judgments and evaluations that are based on (a) core chemical entities (elements and compounds) and their structures, (b) standards that should adhere to the theoretical framework of chemistry, and (c) analytical procedures that are thought to be complex and interconnected.
4.5 Classical Instrumental Analysis
4.5.1 Ontological Commitment
In the sociocultural domain, the advancement of physical chemistry changed the views on materials and their components as well as their properties and interactions. An example of this can be found in the description of the forces of attraction and repulsion among components of matter (e.g., molecules, atoms or ions which consist of subatomic particles, such as nuclei, protons, electrons) (Weisberg et al., 2019). The materials differ in terms of their density, form, texture (de Berg, 2019), and energetic state of their components (Cammann, 1992), which interact dynamically with each other, forming new chemical entities, which have different properties from those of their constituent parts. These properties are called emergent properties since they are solely present in the new entities with a specific level of structural complexity and not in the lower-complexity components. This is frequently summed up by the widely used assertion that “the whole is more than the sum of the parts” (Luisi, 2002; Simon, 2002), which was also discussed in a previous way of thinking, but in this case we may also predict and explain why this occurs through dynamic interactions of entities and their constituent particles.
In terms of analytical procedures, methods involve isolated instruments in the laboratory (Karayannis & Efstathiou, 2012). Under certain conditions, physical or physicochemical properties based on their constituent entities of the samples (such as conductivity, electrode potential, light absorption or emission, mass-to-charge ratio, fluorescence) are measured (Skoog et al., 2007), employing a variety of instruments including polarographs, photometers, potentiometers, mass spectrometers, fluorimeters, chromatographs, NMR spectrometers (Laszlo, 1998; Valcarcel, 1992), and radiometers (Pérez-Bustamante, 1997). It should be noted that when a very simple instrument for measuring the volume or the mass, like a balance, is used, analysis is not considered instrumental. As far as qualitative analysis is concerned, the purpose of analysis is still to ascertain the sample’s composition, while, regarding quantitative analysis, analysis aims at the determination of the amount of the entities with similar or different physical or physicochemical properties (Skoog et al., 2007; Karayannis & Efstathiou, 2012).
With the foregoing in mind, the ontological commitment of this way of thinking encompasses views of the last point of analysis as dynamically interacting molecules, atoms, and ions made up of dynamically interacting particles (e.g., protons, electrons). These components of matter can be determined using isolated instruments in a laboratory setting.
Our proposed ontological commitment is further strengthened by data from the ontogenetic domain. In particular, thinking of molecules, atoms, and ions as dynamic components of matter that consist of entities such as electrons and protons, whose interactions result in emergent physical properties (“emergence”), is a prerequisite for students to fully understand the theoretical background of instrumental analysis. It is suggested that experienced students can understand the relationship between microscopic particles and their corresponding macroscopic properties by employing the idea of emergence (Sieroka et al., 2024). For example, they are able to acknowledge that a material’s physical characteristics (such as density and melting point) are emergent properties produced by the interactions of an enormous number of particles that make up the material. Meanwhile, they go beyond the sum of the features that define any given components (Ashkenasy et al., 2017; Ngai et al., 2014; Wang, 2014) and explain chemical properties (e.g., acid–base and redox) by means of interactions between subatomic particles of single components (Liu & Lesniak, 2006; Ngai et al., 2014; Orduña Picón et al., 2020).
Therefore, the ontological commitment of the way of thinking of “classical instrumental analysis”, as determined by our secondary data analysis across both genetic domains, relates chemical analysis to dynamically interacting molecules, atoms, and ions composed of dynamically interacting entities (e.g., protons, electrons). Procedures for the determination of these components employ isolated instruments in a laboratory setting and are developed based on the dynamic interactions between the components of matter.
4.5.2 Epistemological Commitment
Historical and epistemological data suggest that the theoretical foundations of (physical) chemistry and the comparison of sample signals with standard (which contain a known concentration of the entity being analyzed) signals formed the basis of this way of thinking about chemical analysis (Leicester, 1971; Skoog et al., 2007; Karayannis & Efstathiou, 2012). A unified theoretical framework was used to deductively explain and make predictions based on both experimental results and theory (O’Connor, 1983). Apart from the structure, more information was needed for the characterization of the components of matter (molecules, atoms, ions), including sample preparation and results of elemental analysis. Also, knowledge about physical properties, visual characteristics, chemical reactivity (Schummer, 2002), and elemental mass proportions was required. These are mostly emergent properties (Luisi, 2002) that endure across chemical changes, in contrast to the majority of chemical properties (Ruthenberg & Mets, 2020). We can easily understand that not only have chemical and physical properties been considered equally important (as mentioned in the previous way of thinking, i.e., “classical analysis”), but the relationship between them was suggested, due to the collaboration of physical chemistry and other chemistry domains (Leicester, 1971). For example, according to Beer’s law, in spectrometry, the absorption of a monochromatic radiation is proportional to the concentration of the absorbing particle (Skoog et al., 2007). Also, chemical analysis deals not only with the choice of method but also with the interpretation of analytical results, i.e., how analytical signals are translated into a specific structure (Zuckerman, 1992) or property (Laszlo, 1998) and how the comparison between the signal of a sample and the signal of a standard provides analytical qualitative or quantitative information.
Thus, findings within the sociocultural domain suggest that knowledge is constructed by experiments based on the emergent physicochemical properties of the molecules, atoms, and ions, as well as on the transformation of the analytical signal into chemical information about entities or the comparison of the signals of samples and standards. In any case, explanations and predictions should be made in a deductive way and based on the relevant theory.
In the ontogenetic domain, the epistemological commitment is stabilized for its most part. With regard to the property of emergence, older learners believe that analyzing the interactions between molecules, atoms, and ions and their constituent particles (e.g., protons, electrons) as well as the factors that influence them is a better way of determining their identity than emphasizing only the characteristics of the separate constituents. Nevertheless, many students face challenges in forming an emergent understanding of the characteristics and interactions of matter. Thus, we could acknowledge the concept of emergence as another crucial concept for students when they try to think about chemical identity (Ngai et al., 2014). Attention should be paid when young learners describe emergent properties because they should deductively make predictions based not only on their observations but also on the corresponding theoretical framework (O’Connor, 1983).
Thus, the way of thinking about chemical analysis as “classical instrumental analysis” relies on the epistemological commitment that knowledge is constructed through deductive explanations and predictions within a certain theoretical framework, which are based on the emergent physicochemical properties of entities, the transformation of the analytical signal into chemical information about entities, and the comparison of the signals of the unknown samples with the signal of the standard.
4.5.3 Axiological Commitment
In the sociocultural domain, before the development of instrumental analysis, there had been a significant development of the theoretical foundations of classical—mostly chemical—analysis and an abundance of novel procedures (Vershinin & Zolotov, 2009). As a result, the contribution of instruments to the advancements of chemical analysis is sometimes overlooked by historians of science because of their traditional focus on theory as the driving force of scientific progression (Chamizo, 2014; Slater, 2002). However, instrumental analysis has become so important that, after the proposition of a structure using conventional chemical analysis, the structure predicted must definitely be verified by comparing the result with the spectra of known (structurally defined) substances, mostly obtained through NMR. Nowadays, there are very few instances (mainly in the chemistry of natural products) in which researchers expressly state that they have ascertained the (relative) configuration via just chemical analysis (Schummer, 2002; Slater, 2002). In general, methods have been enhanced in terms of accuracy, sensitivity, and other indexes (Vershinin & Zolotov, 2009), such as non-destructiveness, speed, and detection limits (James, 1983; Leicester, 1971; Slater, 2002). However, the expense of purchasing and particularly of maintaining, housing, and operating these devices is substantial (Slater, 2002).
The aforementioned indicates that the axiological commitment of this way of thinking rests on the significance of instrumental analysis, thereby making it essential as a confirmation tool as well. Methods have low detection limits, accuracy, sensitivity, non-destructiveness, and speed, but they are expensive.
Research on students’ alternative conceptions about chemical analysis indicates that misconceptions pertaining especially to instrumental analysis arise from the fact that many concepts, terms, and symbols employed in this field have ambiguous or interchangeable meanings, such as absorption versus absorbance (Doménech-Carbó et al., 2010) or technique versus method (Karayannis & Efstathiou, 2012). Other alternative conceptions concern the equipment. Instruments are frequently seen as “black boxes” and overly sophisticated (Milligan & Wink, 2024), always accurate and independent from one another. In addition, the operational principles of the apparatus and the basis of the instrumental analysis are typically seen as incidental or subsequent aspects of analysis (Doménech-Carbó et al., 2009).
Incorporating data from both genetic domains, the axiological commitment that underpins this way of thinking relates to evaluations about instrumental analysis as an important and vital confirmation tool of chemical entities’ structures. The respective methods are assessed as accurate, sensitive, non-destructive, fast, and with low detection limits; they are also expensive and complicated in terms of the underlying entities (molecules, atoms, ions, subatomic particles) and the theoretical principles that guide the operation of equipment.
4.6 Contemporary Tool for Society
4.6.1 Ontological Commitment
In this way of thinking, entities are treated in terms of their relationship to social, environmental, etc. issues rather than their chemical composition (for example, consisting of dynamically interacting subatomic components as in the previous way of thinking). As a contemporary tool for society, chemical analysis provides solutions to problems about chemical entities related to society, environment, and economy as well as research and development (Karayannis & Efstathiou, 2012). Together with the utilization of mostly coupled instruments in the laboratories, field work is also conducted (Vershinin & Zolotov, 2009). Furthermore, the barrier between qualitative and quantitative analysis fades as methods grow more comparable and quantitative analysis takes precedence (Štulík & Zýka, 1992). The arrival of computers and especially microcomputers (Valcarcel, 1992) is crucial to the differentiation between this and the previous ways of thinking.
Regarding the sociocultural domain, this particular way of thinking is stabilized by the ontological commitment that the endpoint of chemical analysis is thought to be entities which are related to social, economic, or environmental problems, which are determined through the employment of instrumental analytical methods including the use of coupled instruments in the laboratory or in fieldwork.
A limited amount of data on students’ ontological commitments to chemical analysis as a contemporary tool for society is available to us from the ontogenetic domain. To be more precise, entities, notions, and procedures concerning social demands, such as environmental analysis and quality assessment in the manufacturing sector, are not always clear for students (Doménech-Carbó et al., 2009). In addition, it is not uncommon for students to have a view of analytical methods as a linear series of analytical activities (sampling, preliminary tasks, use of instruments) executed only once (Doménech-Carbó et al., 2009). Additionally, they sometimes fail to understand that instrumental measurement is merely the last and simpler step in an analytical protocol and that there are more steps after the measurement phase (Karayannis & Efstathiou, 2012).
Therefore, chemical analysis as a “contemporary tool for society” concerns entities which are generally related to social, economic, or environmental problems and determined by linear approaches involving coupled instruments in the laboratory or in the field.
4.6.2 Epistemological Commitment
Data from the sociocultural domain reveal that modern chemical analysis focuses on basic and applied research on entities as well as exploration of techniques and methods of chemical analysis (Štulík & Zýka, 1992; Valcarcel, 1992; Karayannis & Efstathiou, 2012). The current approach emphasizes the interplay of a variety of problem-solving methods as well (Štulík & Zýka, 1992), which relies on the incorporation of microcomputers along with the collaboration with a variety of scientific disciplines, including physics, mathematics, informatics, nanotechnology, and robotics (Leicester, 1971; Vershinin & Zolotov, 2009, Karayannis & Efstathiou, 2012) as well as chemometrics (e.g., pattern identification and fingerprinting techniques) (Zolotov, 2020), while metrological support is always provided (Vershinin & Zolotov, 2009). Among other things, chemical analysis aims to reduce analytical error, collect more data, and provide multifaceted information about entities with similar or different properties, and instead of entities alone, total indexes are sometimes determined (Vershinin & Zolotov, 2009; Karayannis & Efstathiou, 2012). A strong connection between the sampling technique and the analyzing apparatus is mentioned (Baiulescu, 2000).
So knowledge about chemical analysis relies on large data about entities and analytical procedures aided by microcomputers, chemometrics, and the collaboration of chemistry with other scientific disciplines.
According to the findings of the ontogenetic domain, contemporary chemical analysis includes laws, procedures, specific vocabulary, rules and so forth, in addition to entities and procedures. The majority of these notions and procedures relate to analytical properties (precision and accuracy, reliability, etc.), operational terms (separation, identification), and mathematical tools (linear correlation, integration and derivation). Many of these concepts are not just chemical entities or procedures with clarified and strict descriptions; hence, alternative ideas about them are called metaconceptions (Doménech-Carbó et al., 2009). For example, metrological terminology, such as traceability and uncertainty, which is crucial for contemporary analytical methods, is not fully understood by students (Karayannis & Efstathiou, 2012). It also appears that societal demands arising from diverse real-world conditions have no effect on which analytical methods students choose. Learners often analyze data in an automatic manner, ignoring alternatives or complexities (Doménech-Carbó et al., 2010).
Combining data from both domains, we can somehow extend the epistemological commitment of the way of thinking about chemical analysis as a “contemporary tool for society.” Knowledge construction is related to large data regarding the entities and the analytical procedures along with the integration of microcomputers in all instruments, the application of chemometrics, and the collaboration between other related scientific fields, such as physics, mathematics, and informatics.
4.6.3 Axiological Commitment
According to the sociocultural domain, the selection of entities examined is influenced by society and its current demands (Karayannis & Efstathiou, 2012). For example, during the COVID-19 pandemic, chemical analysis aimed at the identification of the genetic material of SARS-CoV-2 in waste products, while in other situations it targets specific drugs or environmental indicators.
Data from both domains concur in terms of analytical terminology. According to the sociocultural domain, reliability, sensitivity, selectivity, optimization, specialization, and lower detection limits are only some of the characteristics which are taken into consideration for the choice of the most appropriate method. In practice, analysts will make the required compromises between sensitivity, precision, and analysis cost in order to reduce the error and select the most effective analytical methods for a given sample (Cammann, 1992; Karayannis & Efstathiou, 2012).
Regarding students’ ideas and alternative conceptions, it is suggested that learners are unaware that they are compromising analytical properties in order to reduce analytical error, even if they actually make such trade-offs (Karayannis & Efstathiou, 2012). Also, it is not uncommon for students to have mental models involving an error-free belief for data obtained from instruments and experimentation in general and a pseudo-empiricist assumption of scientific practice, namely, that theory-independent measurements convey analytical information (Doménech-Carbó et al., 2009).
Another interesting finding in the ontogenetic domain indicates that students express a variety of attitudes toward contemporary chemical analysis, from positive (for example, because of the more accurate results) or uncertain (for example, because of the many calculations) to negative (for example, because of the complexity of steps) (Milligan & Wink, 2024). Furthermore, learners’ intuitive reasoning presents an obvious dominance of instrumental over classical chemical analysis, in terms of goals, methodology, and analytical performance (Doménech-Carbó et al., 2012). Thus, many of them believe that classical qualitative and quantitative analysis should be abolished from university curricula, because of the development of instrumental analysis, and that microcomputer control should be introduced on almost all analytical instruments since any analysis can thus be carried out very easily, quickly, and accurately (Karayannis & Efstathiou, 2012). This variety of attitudes also relates to the impact of chemical analysis on society and the environment. Many students believe that chemical practices, in general, help solve environmental issues and prolong human life (Salta & Tzougraki, 2004). On the contrary, there is a generalized dislike of chemistry, which has caused many to associate the word “chemical” (with a negative connotation) with artificial and perhaps hazardous materials, such as poisons, chemical warfare, and environmental pollutants (Freire et al., 2019; Laszlo, 2006; Schummer et al., 2007). Students have unfavorable opinions of the discipline and lack a critical, well-rounded understanding of the advantages, disadvantages, and threats associated with analyzing, manipulating, and using chemicals in activities and goods (Freire et al., 2019).
Therefore, the axiological commitment that stabilizes this way of thinking is the variety of views (positive, neutral, negative) considering not only the complexity of chemical analysis itself but also its interplay with society, economy, and environment. Compromises between sensitivity, precision, and analysis cost are made in order to minimize analytical error. Due to the emphasis placed on instruments and experimentation, a false empiricist assumption sometimes prevails, thus diminishing the role of theory in the interpretation of results.
5 Conclusions
This study describes the heterogeneity of thinking emerging from the sociocultural and ontogenetic domains as a part of a research project aiming at building a conceptual profile model of the “chemical analysis” concept. Due to the fact that the different ways of thinking about a concept are dependent on the sociocultural context studied (Orduña Picón et al., 2020), our findings result from an inductive qualitative analysis of secondary data obtained from different genetic domains in a dialogic way (Mortimer et al., 2012). The sociocultural domain was investigated through historical and epistemological literature on the evolution of the concept of chemical analysis. As far as the ontogenetic domain is concerned, we explored how the concept of chemical analysis is developed in everyday life and learned or, in other words, how students construct their knowledge about the concept (Mortimer et al., 2014a). As suggested by the founders of the theory of the conceptual profile (Mortimer et al., 2012), students’ alternative conceptions and thinking about chemical analysis share some similarities with historical conceptualization (Liu & Lesniak, 2006). The purpose of data analysis in the current work was not to simply categorize the data or to create a detailed historical report but to delve more deeply into the statements in order to understand them in light of their ontological, epistemological, and axiological commitments, which are not often expressed explicitly either orally or in writing. These commitments establish, stabilize, and differentiate each way of thinking from others (Mortimer et al., 2014b; Orduña Picón et al., 2020).
One limitation of our work is related to the data analysis strategy in general. In order to infer the categories of ways of thinking and, in a next stage of research, the zones, we started with the historical, philosophical, and alternative conceptions sources to build a conceptual profile model (the second type of strategy) and not with authentic data as in the first type of strategy (Mortimer et al., 2014a). Primary empirical data, however, may be richer than the articulation reached in this paper. Also, the potential for bias in the interpretation of the primary data is another issue (Mortimer et al., 2014a). In the subsequent stage of our research, ongoing dialogue between all three data sources, that is the sociocultural, ontogenetic, and the microgenetic domains, will help to mitigate these risks.
Another limitation of the current work is related to the fact that there exists a relatively small body of literature on students’ alternative conceptions about chemical analysis. In the immediate future, the incorporation of the primary data obtained from students’ responses is expected to enrich the ontogenetic domain, as the founders of the conceptual profile framework have suggested (Mortimer et al., 2014b).
Due to the restricted contribution of the ontogenetic domain, in a few cases, the sociocultural domain data appears to be dominant, and a temporal classification seems to be derived. This, however, does not apply in our work since the identification of each distinct way of thinking is based on the determination of its ontological, epistemological, and axiological commitments, using the questions mentioned in Sect. 3. In this sense, our data analysis is dialogical rather than sequential, as potential perspectives that emerge in the sociocultural domain are sought out and either validated, enriched, or rejected in the ontogenetic domain. At the same time, there are instances where a way of thinking or a commitment to a way of thinking is detected in the ontogenetic domain; in these cases, the equivalent is then sought in the sociocultural domain. Furthermore, the lack of relevant research in the ontogenetic field highlights the importance of developing a conceptual profile model for chemical analysis, as this will fill the gap in the literature and may serve as the foundation for future research on students’ conceptions and thinking.
Six categories of ways of thinking about the concept of chemical analysis resulted from our preliminary classification. Thinking about chemical analysis as “everyday analysis” entails perceptions of matter as stuff or objects. In addition, it includes views of separating materials via practices which are based on human senses and on explicit or object-related properties. Thinking regarding procedures of chemical analysis, which are not necessarily conducted in a laboratory, is independent of theoretical ideas. Individuals think that only the materials and objects that they find useful in their daily and professional lives are worth analyzing, and this utility lies in the material’s origin, habitat, purpose, function, and similarity to valuable exemplary materials.
The way of thinking “alchemical analysis” involves thoughts of entities as “elements”, “principles”, “substances” or “contraries” that constitute the essence of materials, which are analyzed using specific yet simple isolation and separation procedures. Knowledge about these entities and procedures is constructed using senses by direct observation of essential and additive properties of materials. There is often a lack of awareness of some of the “experimental” findings, and theoretical ideas are manipulated and sometimes partially used or even not used at all. Constant repetition of procedures seems crucial in order to get the desired outcome. Alchemical analysis thinking includes various evaluations and beliefs, which are influenced by the origins of alchemy and are either positive or negative.
Continuing with “empirical analysis”, those who think in this way regard elements and compounds from which materials, objects, and solutions are composed as the endpoints of analysis. They also think that elements and compounds can be determined by simple experimental procedures based on theories as well as on operational categorizations of the elements and compounds having emergent properties. Judgments and evaluations of the entities and analytical procedures are based on theory and on their compliance with the scientific method.
Τhe “classical analysis” way of thinking involves elements and compounds with specific chemical composition and static structures, which are determined through simple experimental qualitative or quantitative procedures. This way of thinking is based on mostly explicit physical and chemical properties of elements and compounds as well as on their categorization via structure–property relationships, addressing thus the choice of the most suitable analytical procedure. Also, judgments and evaluations rely on chemical structures, theoretical standards, and complex analytical procedures.
Moving on to the “classical instrumental analysis” way of thinking, this is related to perceptions about dynamically interacting entities and about procedures for the determination of these entities which depend on their interactions. Knowledge is constructed through deductive explanations and predictions within a certain theoretical framework, which are based on the emergent physicochemical properties of entities and the transformation of the analytical signal into chemical information. These methods are evaluated as an important confirmation tool of chemical entities’ structures, as well as accurate, sensitive, non-destructive, fast, and with low detection limits. However, they are considered expensive and complicated in terms of the underlying entities and the theoretical principles which lie behind the operation of the equipment.
Finally, the way of thinking that mainly focuses on social, economic, or environmental problems, which are related to entities-targets of chemical analysis regardless of their chemical composition and are determined by linear approaches involving coupled instruments in the laboratory or in the field, is described as a “contemporary tool for society”. Knowledge construction is related to large data regarding the entities and the analytical procedures along with the integration of microcomputers in all instruments, the application of chemometrics, and the collaboration between other related scientific fields, such as physics, mathematics, and informatics. Those who think of chemical analysis in this way possess favorable, impartial, or unfavorable opinions of the complexity of chemical analysis itself and its interplay with society, economy, and environment, while analytical properties (such as sensitivity, specialization, accuracy, cost) and the compromises between them are frequently under evaluation in order to minimize analytical error. Quite often, a false empiricist assumption prevails, and the role of theory in the interpretation of results is diminished.
The boundaries of the categories of ways of thinking of the proposed conceptual profile model may not always be clear because the identification of these categories was not based on the complete replacement of ideas or techniques by others (Vershinin & Zolotov, 2009) but on the differentiation of the ontological, epistemological, and axiological commitments. However, this overlap between the expanded categories of ways of thinking highlights the dynamic character of the conceptual profile theoretical framework (Liu & Lesniak, 2006).
We wish to clarify that the proposed expanded categories of ways of thinking should not be regarded as zones because no data from the microgenetic domain has yet been integrated (Mortimer et al., 2014a). As we have already mentioned, in the following phase of the undertaken research, the herein proposed conceptual profile model of chemical analysis will be enhanced with data from the microgenetic domain. The collection and analysis of such empirical data will provide final determination of the zones (and their corresponding commitments) constituting a conceptual profile model of chemical analysis, whose initial proposal was made in this work for the first time. The development of a conceptual profile model of chemical analysis will provide a firm basis for a deeper understanding of students’ ideas on chemical analysis and a pivotal point for the design of novel evidence-based educational interventions for the advancement of chemistry teaching.
Data Availability
Not applicable.
References
-
Aguiar, O., Sevian, H., & El-Hani, C. N. (2018). Teaching about energy: Application of the conceptual profile theory to overcome the encapsulation of school science knowledge. Science & Education, 27(3), 863–893. https://doi.org/10.1007/s11191-018-0010-z
-
Aguiar, O. G. (2014). The implications of the conceptual profile in science teaching: An example from a teaching sequence in thermal physics. In E. Mortimer & C. El-Hani (Εds.), Conceptual Profiles: A Theory of Teaching and Learning Scientific Concepts (pp. 235–259). Springer. https://doi.org/10.1007/978-90-481-9246-5_9
-
**Ashkenasy, G., Hermans, T. M., Otto, S., & Taylor, A. F. (2017). Systems chemistry. Chemical Society Reviews, 46(9), 2543-2554. https://doi.org/10.1039/C7CS00117HG
-
*Baiulescu, G. E. (2000). Analytical chemistry, past, present and future. Analytical Letters, 33(4), 571–577. https://doi.org/10.1080/00032710008543074
-
Baltieri, R. S., Bego, A. M., & Cebim, M. A. (2021). Why the covalent bond is such a complex concept: A conceptual profile proposal. International Journal of Science Education, 43(6), 1–18. https://doi.org/10.1080/09500693.2021.1949070
-
*Beaney, M., & Raysmith, T. (2024). Analysis. In E. N. Zalta & U. Nodelman (Eds.), The Stanford encyclopedia of philosophy (Fall 2024 ed.). Stanford University, Metaphysics Research Lab. Retrieved February 12, 2024, from https://plato.stanford.edu/archives/fall2024/entries/analysis/
-
*Belcher, R., Szabadváry, F., & Peden, J. D. (1977). Annual chemical congress: History of analytical chemistry. Proceedings of the Analytical Division of the Chemical Society, 14(7), 161-171. https://doi.org/10.1039/AD9771400161
-
*de Berg, K. C. (2019). The iron (III) thiocyanate reaction: Research history and role in chemical analysis. Switzerland: Springer Cham. https://doi.org/10.1007/978-3-030-27316-3
-
*Bolton, H. C. (1897). The revival of alchemy. Science, 6(154), 853-863. https://doi.org/10.1126/science.6.154.853
-
Breslow, R. (1997). Chemistry today and tomorrow: The central, useful, and creative science. Jones & Bartlett Learning.
-
*Brock, W. H. (2016). The history of chemistry: A very short introduction. Oxford: Oxford University Press.
-
Cambridge University Press & Assessment. (n.d.). Analysis (noun). In Cambridge Dictionary. Retrieved August 12, 2024, from https://dictionary.cambridge.org/dictionary/english/analysis
-
*Cammann, K. (1992). Analytical chemistry — Today’s definition and interpretation. Fresenius’ Journal of Analytical Chemistry, 343, 812–813. https://doi.org/10.1007/BF00328560
-
*Campbell, W. A. (1979). Fact and fantasy in chemical analysis. Endeavour, 3(1), 38-41. https://doi.org/10.1016/0160-9327(79)90008-5
-
Caspari, I., Kranz, D., & Graulich, N. (2018). Resolving the complexity of organic chemistry students’ reasoning through the lens of a mechanistic framework. Chemistry Education Research and Practice, 19(4), 1117–1141. https://doi.org/10.1039/C8RP00131F
-
*Chamizo, J. A. (2014). The role of instruments in three chemical’ revolutions. Science & Education, 23(4), 955-982. https://doi.org/10.1007/s11191-014-9678-x
-
Chi, M. T., Slotta, J. D., & De Leeuw, N. (1994). From things to processes: A theory of conceptual change for learning science concepts. Learning and Instruction, 4(1), 27–43. https://doi.org/10.1016/0959-4752(94)90017-5
-
**Chi, M. T. H. (2005). Commonsense conceptions of emergent processes: Why some misconceptions are robust. Journal of the Learning Sciences, 14(2), 161–199. https://doi.org/10.1207/s15327809jls1402_1
-
*Coley, N. G. (1982). Physicians and the chemical analysis of mineral waters in eighteenth-century England. Medical History, 26(2), 123-144. https://doi.org/10.1017/S0025727300041120
-
Cooper, M., Kouyoumdjian, H., & Underwood, S. M. (2016). Investigating students’ reasoning about acid–base reactions. Journal of Chemical Education, 93(10), 1703–1712. https://doi.org/10.1021/acs.jchemed.6b00417
-
*Curd, P. (2007). Presocratic philosophy. In E. N. Zalta & U. Nodelman (Eds.), The Stanford encyclopedia of philosophy (Fall 2023 ed.). Stanford University, Metaphysics Research Lab. Retrieved February 12, 2024, from https://plato.stanford.edu/archives/fall2023/entries/presocratics/
-
da Silva Costa, M. B., & dos Santos, B. F. (2022). The conceptual profile of equilibrium and its contributions to the teaching of chemical equilibrium. Chemistry Education Research and Practice, 23(1), 226–239. https://doi.org/10.1039/D1RP00039J
-
de Mattos, C. R. (2014). Conceptual profile as a model of a complex world. In E. Mortimer & C. El-Hani (Εds.), Conceptual profiles: A theory of teaching and learning scientific concepts (pp. 263–292). Springer. https://doi.org/10.1007/978-90-481-9246-5_10
-
do Amaral, E. M. R., da Silva, J. R. R. T., & Sabino, J. D. (2018). Analysing processes of conceptualization for students in lessons on substance from the emergence of conceptual profile zones. Chemistry Education Research and Practice, 19(4), 1010–1028. https://doi.org/10.1039/C8RP00050F
-
do Amaral, E. M. R., Mortimer, E. F., & Scott, P. (2014). A conceptual profile of entropy and spontaneity: Characterising modes of thinking and ways of speaking in the classroom. In E. Mortimer & C. El-Hani (Εds.), Conceptual profiles: A theory of teaching and learning scientific concepts (pp. 201–234). Springer. https://doi.org/10.1007/978-90-481-9246-5_8
-
Doménech-Carbó, A., Gimeno-Adelantado, J. V., & Bosch-Reig, F. (2009). Misconceptions and metaconceptions in instrumental analysis/Concepções errôneas e metaconcepções em análise instrumental. Acta Scientiae, 11(1), 73–87.
-
Doménech-Carbó, A., Gimeno-Adelantado, J. V., & Bosch-Reig, F. (2010). Black boxes in analytical chemistry: University students’ misconceptions of instrumental analysis. US-China Education Review, 7(7), 15–29.
-
**Doménech-Carbó, A., Gimeno-Adelantado, J. V., & Mateo-Castro, R. (2012). Classical vs. instrumental analytical chemistry: The role of metaconceptions. In L. Gómez Chova, A. López Martínez & I. Candel Torres (Eds.), INTED2012 Proceedings (pp. 5805–5812). IATED. https://library.iated.org/view/DOMENECHCARBO2012CLA
-
El-Hani, C. N., da Silva-Filho, W. J., & Mortimer, E. F. (2014). The epistemological grounds of the conceptual profile theory. In E. Mortimer & C. El-Hani (Εds.), Conceptual profiles: A theory of teaching and learning scientific concepts (pp. 35–65). Springer. https://doi.org/10.1007/978-90-481-9246-5_2
-
El-Hani, C. N., do Amaral, E. M. R., & Mortimer, E. F. (2023). Heterogeneity and pluralism in science education from the perspective of conceptual profiles. In M. Bélanger, P. Potvin, S. Horst, A. Shtulman & E. F. Mortimer (Eds.), Multidisciplinary perspectives on representational pluralism in human cognition (pp. 181–200). Routledge. https://doi.org/10.4324/9781003189930-12
-
*Erduran, S. (2001). Philosophy of chemistry: An emerging field with implications for chemistry education. Science & Education, 10, 581-593. https://doi.org/10.1023/A:1017564604949
-
Fischer, F., Kollar, I., Ufer, S., Sodian, B., Hussmann, H., Pekrun, R., … & Eberle, J. (2014). Scientific reasoning and argumentation: Advancing an interdisciplinary research agenda in education. Frontline Learning Research, 2(3), 28–45. https://doi.org/10.14786/flr.v2i2.96
-
**Forbes, G. (2017). Essentialism. In B. Hale, C. Wright & A. Miller (Eds.), A companion to the philosophy of language (pp. 881–901). USA: John Wiley & Sons, Inc. https://doi.org/10.1002/9781118972090.ch34
-
*Fors, H. (2022). Alchemy, chemistry, and metallurgy. In D. Jalobeanu & C. Wolfe (Eds.), Encyclopedia of early modern philosophy and the sciences (pp. 1–6). Springer, Cham. https://doi.org/10.1007/978-3-319-20791-9_468-1
-
**Freire, M., Talanquer, V., & Amaral, E. (2019). Conceptual profile of chemistry: A framework for enriching thinking and action in chemistry education. International journal of science education, 41(3), 1-19. https://doi.org/10.1080/09500693.2019.1578001
-
**Garnett, P. J., Garnett, P. J., & Hackling, M. W. (1995). Students’ alternative conceptions in chemistry: A review of research and implications for teaching and learning. Studies in Science Education, 25(1), 69–96. https://doi.org/10.1080/03057269508560050
-
*Hacker, P. (2004). Substance: Things and stuffs. Aristotelian Society Supplementary Volume, 78(1), 41–63. https://doi.org/10.1111/j.0309-7013.2004.00114.x
-
*Hopkins, A. J. (1925). A modern theory of alchemy. Isis, 7(1), 58-76. https://doi.org/10.1086/358298
-
**Horton, C. (2007). Student alternative conceptions in chemistry. California Journal of Science Education, 7(2), 18-38.
-
*Hudson, J. (1992). The history of chemistry. London: Red Globe Press.
-
*James, F. A. J. L. (1983). The establishment of spectro-chemical analysis as a practical method of qualitative analysis, 1854–1861. Ambix, 30(1), 30-53. https://doi.org/10.1179/amb.1983.30.1.30
-
*/**Karayannis, M. I., & Efstathiou, C. E. (2012). Significant steps in the evolution of analytical chemistry – Is the today’s analytical chemistry only chemistry?.Talanta, 102(4), 7-15. https://doi.org/10.1016/j.talanta.2012.06.003
-
*Kauffman, G. B. (1985). The role of gold in alchemy. Part I. Gold bulletin, 18(1), 31-44. https://doi.org/10.1007/BF03214684
-
*Kim, M. G. (2000). Chemical analysis and the domains of reality: Wilhelm Homberg’s Essais de chimie, 1702-1709. Studies in history and philosophy of science, 31(1), 37-69. https://doi.org/10.1016/S0039-3681(99)00033-3
-
*Kim, M. G. (2014). Stabilizing chemical reality: The analytic-synthetic ideal of chemical species. HYLE-International Journal for Philosophy of Chemistry, 20(1), 117-139.
-
*Klein, J. A., & Ragland, E. R. (2014). Introduction analysis and synthesis in medieval and early modern Europe. Ambix, 61(4), 319-326. https://doi.org/10.1179/0002698014Z.00000000065
-
*Klein, U. (2007). Styles of experimentation and alchemical matter theory in the scientific revolution. Metascience, 16(2), 247-256. https://doi.org/10.1007/s11016-007-9095-8
-
*Klein, U. (2012). Objects of inquiry in classical chemistry: Material substances. Foundations of Chemistry, 14(1), 7-23. https://doi.org/10.1007/s10698-011-9122-0
-
**Krnel, D., Watson, R., & Glažar, S. (1998). Survey of research related to the development of the concept of ‘matter’. International Journal of Science Education, 20(3), 257 – 289. https://doi.org/10.1080/0950069980200302
-
*/**Langley, P., Simon, H., Bradshaw, G., & Zytkow, J. (1987). Scientific discovery: Computational explorations of the creative process. Cambridge, MA: MIT Press/Bradford Books.
-
*Laszlo, P. (1998). Chemical analysis as dematerialization. HYLE – An International Journal for the Philosophy of Chemistry, 4(1), 29-38.
-
**Laszlo, P. (2006). On the self-image of chemists, 1950–2000. HYLE – International Journal for Philosophy of Chemistry, 12(1), 99–130.
-
*Leicester H. M. (1971). The historical background of chemistry. New York: Dover Publications, Inc.
-
**Liu, X., & Lesniak, K. (2006). Progression in children’s understanding of the matter concept from elementary to high school. Journal of Research in Science Teaching, 43(3), 320– 347. https://doi.org/10.1002/tea.20114
-
*Luisi, P. L. (2002). Emergence in chemistry: Chemistry as the embodiment of emergence. Foundations of Chemistry, 4(3), 183-200. https://doi.org/10.1023/A:1020672005348
-
**Mas, C. J. F., Perez, J. H., & Harris, H. H. (1987). Parallels between adolescents’ conception of gases and the history of chemistry. Journal of Chemical Education, 64(7), 616-618. https://doi.org/10.1021/ed064p616
-
**Milligan, R. D., & Wink, D. J. (2024). Mass is better than light: Students’ perception of using spectrophotometry and gravimetric analysis to determine the formula of a hydrate in the general chemistry lab. Journal of Chemical Education, 101(5), 1822-1835. https://doi.org/10.1021/acs.jchemed.4c00076
-
Moon, A., Stanford, C., Cole, R., & Towns, M. (2017). Analysis of inquiry materials to explain complexity of chemical reasoning in physical chemistry students’ argumentation. Journal of Research in Science Teaching, 54(10), 1322–1346. https://doi.org/10.1002/tea.21407
-
Mortimer, E. F., & Amaral, L. O. F. (2014). Contributions of the sociocultural domain to build a conceptual profile model for molecule and molecular structure. In E. Mortimer & C. El-Hani (Εds.), Conceptual profiles: A theory of teaching and learning scientific concepts (pp. 103–114). Springer. https://doi.org/10.1007/978-90-481-9246-5_4
-
Mortimer, E. F., Scott, P., & El-Hani, C. N. (2012). The heterogeneity of discourse in science classrooms: The conceptual profile approach. In B. Fraser, K. Tobin & C. McRobbie (Eds.), Second international handbook of science education (pp. 231–246). Springer. https://doi.org/10.1007/978-1-4020-9041-7_17
-
Mortimer, E. F., El-Hani, C. N., Sepulveda, C., do Amaral, E. M. R., Coutinho, F. Â., & Rodrigues e Silva, F. A. (2014a). Methodological grounds of the conceptual profile research program. In E. Mortimer & C. El-Hani (Εds.), Conceptual profiles: A theory of teaching and learning scientific concepts (pp. 67–100). Springer. https://doi.org/10.1007/978-90-481-9246-5_3
-
Mortimer, E. F., Scott, P., do Amaral, E. M. R., & El-Hani, C. N. (2014b). Conceptual profiles: Theoretical-methodological bases of a research program. In E. Mortimer & C. El-Hani (Εds.), Conceptual profiles: A theory of teaching and learning scientific concepts (pp. 3–33). Springer. https://doi.org/10.1007/978-90-481-9246-5_1
-
National Research Council (2013). Next generation science standards: For states, by states. Washington, DC: The National Academies Press. https://doi.org/10.17226/18290
-
**Newman, G. E., & Knobe, J. (2019). The essence of essentialism. Mind & Language, 34(5), 585-605. https://doi.org/10.1111/mila.12226
-
*Newman, W. R. (2011). What have we learned from the recent historiography of alchemy?. Isis, 102(2), 313-321. https://doi.org/10.1086/660140
-
**Ngai, C., & Sevian, H. (2017). Capturing chemical identity thinking. Journal of Chemical Education, 94(2), 137-148. https://doi.org/10.1021/acs.jchemed.6b00387
-
**Ngai, C., Sevian, H., & Talanquer, V. (2014). What is this substance? What makes it different? Mapping progression in students’ assumptions about chemical identity. International Journal of Science Education, 36(14), 2438-2461. https://doi.org/10.1080/09500693.2014.927082
-
**Nieswandt, M. (2001). Problems and possibilities for learning in an introductory chemistry course from a conceptual change perspective. Science Education, 85(2), 158-179. https://doi.org/10.1002/1098-237X(200103)85:23C158::AID-SCE403E3.0.CO;2-3
-
*/**O’Connor, D. J. (1983). Emergent properties. In A. van der Merwe (Ed.), Old and new questions in physics, cosmology, philosophy, and theoretical biology: Essays in honor of Wolfgang Yourgrau (pp. 719–732). Boston, MA: Springer. https://doi.org/10.1007/978-1-4684-8830-2_48
-
**Orduña Picón, R., Sevian, H., & Mortimer, E. F. (2020). Conceptual profile of substance: Representing heterogeneity of thinking in chemistry classrooms. Science & Education, 29(5), 1317-1360. https://doi.org/10.1007/s11191-020-00152-4
-
*Partington, J. (1989). A Short History of Chemistry. New York: Dover.
-
*Pérez-Bustamante, J. A. (1997). Analytical chemistry in the discovery of the elements. Fresenius’ Journal of Analytical Chemistry, 357, 162–172. https://doi.org/10.1007/s002160050131
-
*Ragai, J. (1992). The philosopher’s stone: Alchemy and chemistry. Alif: Journal of Comparative Poetics, 12, 58–77. https://doi.org/10.2307/521636
-
*Read, J. (1933). Alchemy and alchemists. Folklore, 44(3), 251-278.
-
*Read, J. (1995). From alchemy to chemistry. New York: Dover.
-
**Robertson Ishii, T., & Atkins, P. (2023). Essential vs. accidental properties. In E. N. Zalta & U. Nodelman (Eds.), The Stanford encyclopedia of philosophy (Winter 2020 ed.). Stanford University, Metaphysics Research Lab. Retrieved April 19, 2021, from https://plato.stanford.edu/archives/win2020/entries/essential-accidental
-
*Ruthenberg, K., & Mets, A. (2020). Chemistry is pluralistic. Foundations of Chemistry, 22(3), 403-419. https://doi.org/10.1007/s10698-020-09378-0
-
**Salta, K., & Tzougraki, C. (2004). Attitudes toward chemistry among 11th grade students in high schools in Greece. Science Education, 88(4), 535-547. https://doi.org/10.1002/sce.10134
-
**Sanmarti, N., Izquierdo, M., & Watson, R. (1995). The substantialisation of properties in pupils’ thinking and in the history of science. Science & Education, 4, 349-369. https://doi.org/10.1007/BF00487757
-
**Schummer, J., Bensaude-Vincent, B., & Van Tiggelen, B. (Eds.). (2007). The public image of chemistry. Singapore: World Scientific.
-
*Schummer, J. (2002). The impact of instrumentation on chemical species identity: From chemical substances to molecular species. In P. Morris (Ed.), From classical to modern chemistry: The instrumental revolution (pp. 188-211). Cambridge: The Royal Society of Chemistry.
-
Sevian, H., & Talanquer, V. (2014). Rethinking chemistry: A learning progression on chemical thinking. Chemistry Education Research and Practice, 15(1), 10–23. https://doi.org/10.1039/C3RP00111C
-
*Shields, C. (2023). Aristotle. In E. N. Zalta & U. Nodelman (Eds.), The Stanford encyclopedia of philosophy (Winter 2023 ed.). Stanford University, Metaphysics Research Lab. Retrieved June 13, 2024, from https://plato.stanford.edu/archives/win2023/entries/aristotle/
-
*Siegfried, R. (1988). The chemical revolution in the history of chemistry. Osiris, 4(1), 34-50.
-
**Sieroka, N., Lossau, T., & Neudecker, T. (2024). Emergent properties in chemistry ‐ Relating molecular properties to bulk behavior. Chemistry – A European Journal, 30(25). https://doi.org/10.1002/chem.202303868
-
*Simon, J. (2002). Analysis and the hierarchy of nature in eighteenth-century chemistry. The British Journal for the History of Science, 35(1), 1-16. https://doi.org/10.1017/S0007087401004587
-
*Skoog, D. A., Holler, F. J., & Crouch, S. R. (2007). Instrumental analysis . Belmont: Brooks/Cole.
-
*Slater, L. B. (2002). Instruments and rules: R. B. Woodward and the tools of twentieth-century organic chemistry. Studies in History and Philosophy of Science Part A, 33(1), 1–33. https://doi.org/10.1016/S0039-3681(01)00024-3
-
Solsona, N. R., Izquierdo, M., & De Jong, O. (2003). Exploring the development of students’ conceptual profiles of chemical change. International Journal of Science Education, 25(1), 3–12. https://doi.org/10.1080/09500690010006536
-
**Stains, M., & Talanquer, V. (2007). A2: Element or compound?. Journal of Chemical Education, 84(5), 880-883. https://doi.org/10.1021/ed084p880
-
**Stavy, R. (1991). Children’s ideas about matter. School Science and Mathematics, 91(6), 240– 244. https://doi.org/10.1111/j.1949-8594.1991.tb12090.x
-
*Stock, J. T. (1977). Short course on the history of analytical chemistry and the related sciences. Journal of Chemical Education, 54(10), 635-637. https://doi.org/10.1021/ed054p635
-
*Strathern, P. (2000). Mendeleyev’s dream: The quest for the elements. London: Hamish Hamilton.
-
*Štulík, K., & Zýka, J. (1992). Analytical chemistry — Today’s definition and interpretation. Fresenius’ Journal of Analytical Chemistry, 343, 832–833. https://doi.org/10.1007/BF00328569
-
*Szabadváry, F., & Robinson, A. (1980). The history of analytical chemistry. In G. Svehla (Ed.), Comprehensive Analytical Chemistry (Vol. 10, pp. 61–282). Amsterdam: Elsevier. https://doi.org/10.1016/B978-0-444-41859-3.50008-6
-
**Taber, K. S. (2019). Alternative conceptions and the learning of chemistry. Israel Journal of Chemistry, 59(6-7), 450-469. https://doi.org/10.1002/ijch.201800046
-
**Talanquer, V. (2006). Commonsense chemistry: A model for understanding students’ alternative conceptions. Journal of Chemical Education, 83(5), 811-816. https://doi.org/10.1021/ed083p811
-
**Talanquer, V. (2008). Students’ predictions about the sensory properties of chemical compounds: Additive versus emergent frameworks. Science Education, 92(1), 96-114. https://doi.org/10.1002/sce.20235
-
Talanquer, V. (2013). How do students reason about chemical substances and reactions?. In G. Tsaparlis & H. Sevian (Eds.), Concepts of matter in science education (pp. 331–346). Springer. https://doi.org/10.1007/978-94-007-5914-5_16
-
**Talanquer, V. (2017). Progressions in reasoning about structure–property relationships. Chemistry Education Research and Practice, 19(4), 998-1009. https://doi.org/10.1039/C7RP00187H
-
Talanquer, V. (2019). Assessing for chemical thinking. In M. Schultz, S. Schmid & G. Lawrie (Eds.), Research and practice in chemistry education (pp. 123–133). Springer. https://doi.org/10.1007/978-981-13-6998-8_8
-
Tan, K. C. D., Goh, N. K., Chia, L. S., & Treagust, D. F. (2002). Development and application of a two-tier multiple choice diagnostic instrument to assess high school students’ understanding of inorganic chemistry qualitative analysis. Journal of Research in Science Teaching, 39(4), 283–301. https://doi.org/10.1002/tea.10023
-
**Tan, K. C. D., Goh, N. K., Chia, L. S., & Treagust, D. F. (2001). Secondary students’ perceptions about learning qualitative analysis in inorganic chemistry. Research in Science & Technological Education, 19(2), 223–234. https://doi.org/10.1080/02635140120087740
-
*Valcarcel, M. (1992). Analytical chemistry — Today’s definition and interpretation. Fresenius’ Journal of Analytical Chemistry, 343, 814–816. https://doi.org/10.1007/BF00328561
-
*Vershinin, V. I., & Zolotov, Y. A. (2009). Periodization of the history of chemical analysis and analytical chemistry as a branch of science. Journal of Analytical Chemistry, 64(8), 859–867. https://doi.org/10.1134/S1061934809080152
-
**Wang, Y. (2014). Nanogeochemistry: Nanostructures, emergent properties and their control on geochemical reactions and mass transfers. Chemical Geology, 378-379, 1-23. https://doi.org/10.1016/j.chemgeo.2014.04.007
-
*Weisberg, M., Needham, P., & Hendry, R., (2019). Philosophy of chemistry. In E. N. Zalta (Ed.), The Stanford encyclopedia of philosophy (Spring 2019 ed.). Stanford University, Metaphysics Research Lab. Retrieved February 12, 2024, from https://plato.stanford.edu/archives/spr2019/entries/chemistry/
-
Yan, F., & Talanquer, V. (2015). Students’ ideas about how and why chemical reactions happen: Mapping the conceptual landscape. International Journal of Science Education, 37(18), 3066–3092. https://doi.org/10.1080/09500693.2015.1121414
-
Zhou, S., Han, J., Koenig, K., Raplinger, A., Pi, Y., Li, D., … & Bao, L. (2016). Assessment of scientific reasoning: The effects of task context, data, and design on student reasoning in control of variables. Thinking Skills and Creativity, 19, 175–187. https://doi.org/10.1016/j.tsc.2015.11.004
-
*Zolotov, Y. A. (2020). Evolution of chemical analysis methods. Herald of the Russian Academy of Sciences, 90(1), 56-62. https://doi.org/10.1134/S1019331620010220
-
*Zuckerman, A. M. (1992). Analytical chemistry – Today’s definition and interpretation. Fresenius’ Journal of Analytical Chemistry, 343, 817-818. https://doi.org/10.1007/BF00328562
Funding
Open access funding provided by HEAL-Link Greece. This work has been funded by a pre-doctoral fellowship (assigned to M.M.) administered by the Hellenic Foundation for Research & Innovation (H.F.R.I.–ELIDEK).
Ethics declarations
Ethics
Not applicable.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Reprints and permissions
About this article
Cite this article
Mavridi, M., Salta, K. & Koulougliotis, D. Conceptual Profile of Chemical Analysis. Sci & Educ (2025). https://doi.org/10.1007/s11191-025-00663-y
- Received
- Revised
- Accepted
- Published
- DOI https://doi.org/10.1007/s11191-025-00663-y
Keywords
- Chemical analysis
- Chemical thinking
- Conceptual profile
- Alternative conceptions
- Commitments
