Article Content
Introduction
Plaque psoriasis is a chronic, immune-mediated skin disease that affects approximately 2–3% of the global population. Among its several clinical variants, plaque psoriasis (psoriasis vulgaris) is the most common form, accounting for about 80–90% of all psoriasis cases. It is characterized by well-demarcated, erythematous plaques with silvery-white scales, typically appearing on the scalp, elbows, knees, and lower back (Raharja et al. 2021; Gisondi et al. 2020). The pathophysiology is still unknown, but it is thought to be a T cell-mediated immunological reaction. Lymphocytes induce specific cytokines such as IL-12, IL-17, and IL-23, which trigger immunological responses to produce more keratinocytes (Raharja et al. 2021; Cherubin et al. 2023; Rendon and Schäkel 2019).
Furthermore, it is thought that many different genetic and environmental factors may contribute to this condition. For example, obesity, smoking, and alcohol drinking are some of the risk factors for psoriasis (Raharja et al. 2021; Salimi et al. 2020). It not only impairs quality of life due to discomfort and cosmetic concerns but is also frequently linked with systemic comorbidities, including psoriatic arthritis, cardiovascular disease, metabolic syndrome, and other related conditions, complicating its overall management.
Moreover, the chronic nature of the disease and the high costs associated with long-term treatment, particularly with biologics, place a considerable financial burden on patients and healthcare systems (Raharja et al. 2021).
Management of psoriasis requires a tailored, multidisciplinary approach. While mild cases may respond to topical agents such as corticosteroids, vitamin D analogs, and retinoids, moderate to severe cases often necessitate systemic therapies. These include conventional agents like methotrexate and cyclosporine, small-molecule inhibitors such as apremilast, and biologic therapies that target specific immune pathways, most notably the IL-17 and IL-23 axes (Sbidian et al. 2020; Chat et al. 2020).
Among the newer biologics, mirikizumab (Miri) is a humanized IgG4 monoclonal antibody used to target immunological reactions. It is composed of two subunits: p19 and p40, with the p19 subunit being unique to IL-23 and critical for its biological activity. IL-23 promotes the expansion and survival of Th17 cells, a subset of T-helper cells that produce pro-inflammatory cytokines such as IL-17A, IL-17F, and IL-22. These cytokines play a central role in driving keratinocyte hyperproliferation, sustained skin inflammation, and the characteristic psoriatic plaques. Overexpression of IL-23, particularly the p19 subunit, has been detected in psoriatic skin lesions, and its signaling is strongly associated with disease severity. Miri has shown incredible results in immune-mediated inflammatory diseases, such as ulcerative colitis, which makes it vulnerable to being tried in other conditions. Also, it was associated with fewer adverse effects and better efficacy. A few clinical trials evaluating patients with varying degrees of plaque psoriasis found rapid and long-lasting clinical improvement, along with a decrease in Psoriasis Area and Severity Index (PASI) scores (Blauvelt et al. 2022 Dec 1; Reich et al. 2019 Jul 1; Papp et al. 2023; Magro et al. 2024; Kobayashi et al. 2024).
This study aimed to assess the effectiveness of mirikizumab in individuals with plaque psoriasis.
Methods
This study followed the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines for systematic review and meta-analysis, ensuring transparency and thoroughness in reporting the research. The protocol was registered on PROSPERO (Moher et al. 2009), available from https://www.crd.york.ac.uk/PROSPERO/view/CRD42024578351
Search strategy
We investigated several databases, including PubMed, Cochrane CENTRAL, Scopus, and Web of Science (WOS), for relevant studies using the following search strategy: “((LY3074828 OR miri OR mirikizumab) AND (psoriasis OR Palmoplantaris Pustulosis OR Pustulosis of Palms and Soles))”.
Eligibility criteria
- Language: English studies.
- Population: Patients who presented with demarcated erythematous plaques with silvery-white scales, commonly located on the scalp, elbows, knees, and lower back, and a diagnosis of plaque-psoriasis was confirmed by a dermatologist.
- Intervention: Use of Mirikizumab.
- Comparator: Placebo.
- Outcome Measures: PASI, body surface area (BSA), Dermatology Life Quality Index (DLQI), static Physician’s Global Assessment (PGA), and any reported adverse effects.
- Study design: All randomized clinical trials and observational research, whether case–control or cohort studies.
We excluded reviews, case studies, editorial letters, conference abstracts, study protocols, animal studies, and studies with insufficient data from our analysis.
Study selection
After abstract and title screening, we performed full-text screening. Two independent authors carried out the screening process by applying exclusion and inclusion criteria. Two authors extracted data from three included trials, which consisted of information on the study ID, type of study, number of patients recruited, duration, criteria for inclusion, average ages, age at diagnosis, results, and interventions used. Any conflicts were resolved through a group discussion.
Outcome assessment
The primary outcomes were PASI response, DLQI, BSA, and sPGA. The secondary outcomes included the overall count of adverse events (AEs), infections, malignancies, upper respiratory tract infections, medication discontinuation, and injection site reactions.
Quality assessment
We evaluated the quality of randomized controlled trials (RCTs) utilizing the Cochrane Handbook (ROB-2), taking into account the following criteria for study quality: randomization, allocation concealment, blinding, missing data, selective outcome reporting, and other biases.
Statistical analysis
We did our meta-analysis using the Review Manager (RevMan) software version 5.2. We used risk ratios for dichotomous data and mean differences for continuous data with 95% confidence intervals. We used the chi-square test to calculate I2 and assess heterogeneity. Subgrouping analysis was done based on drug dosage.
Results
Literature search
A total of 217 articles were identified through searching different databases. By looking at the titles and abstracts of 166 records, 111 records were duplicates. The full-text screening was done for 13 eligible studies. Finally, three papers met our inclusion criteria (Blauvelt et al. 2022 Dec 1; Reich et al. 2019 Jul 1; Papp et al. 2023). The PRISMA flowchart is shown in Fig. 1.
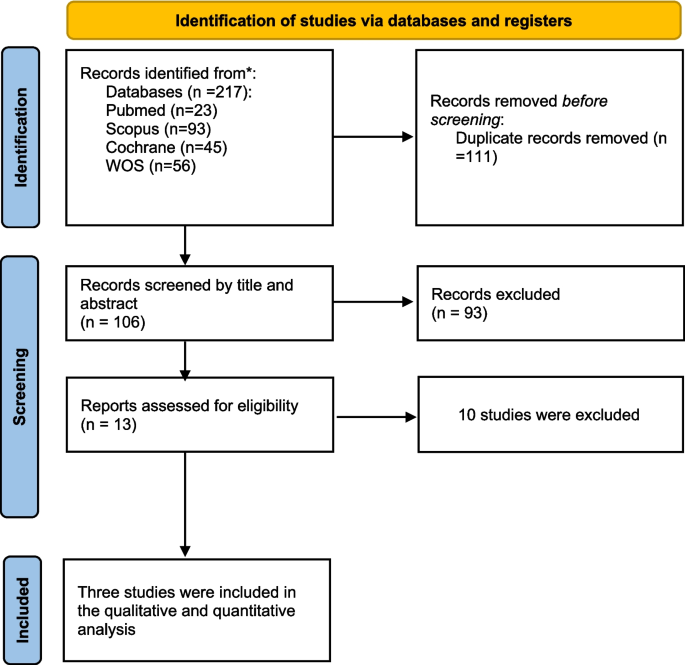
Prisma Flow Diagram
Characteristics of the included studies
Three RCTs involved participants with plaque psoriasis over the age of 40. Table 1 shows the general characteristics of the included studies, while Table 2 outlines the extracted data of the participants’baseline characteristics.
Quality assessment
ROB-2 evaluation of RCTs found that the three included studies demonstrated high quality, as shown in Fig. 2 and Fig. 3.

ROB summary
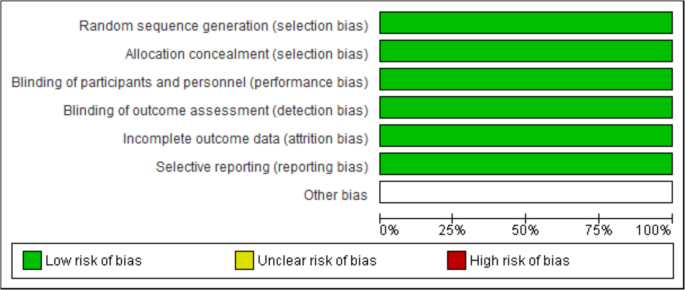
ROB Graph
Outcomes
Psoriasis area and severity index (PASI 75, responders) at week 16
Mirikizumab treatment significantly increased the risk of obtaining PASI by 75% compared to placebo (RR = 10.47, 95% CI (6.94, 15.78), P < 0.00001). The pooled analysis was homogeneous (I2 = 0%). Figure 4
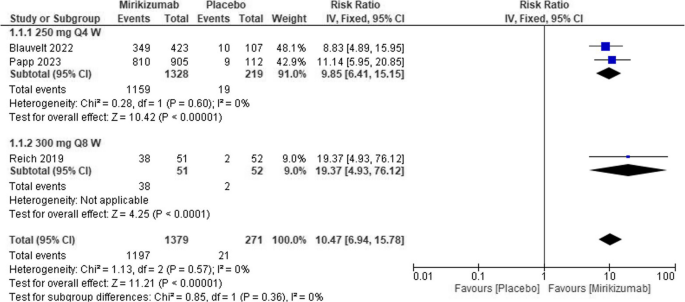
Forest plot of PASI 75 in the mirikizumab group vs. placebo
Psoriasis area and severity index (PASI 90, responders) at week 16
The overall effect showed that patients treated with Miri had a markedly higher likelihood of achieving PASI 90% compared to placebo [RR = 11.5, 95% CI (6.97, 18.96), P < 0.00001]. The analysis was homogeneous (I2 = 0%; P for Cochran Q = 0.40). Figure 5
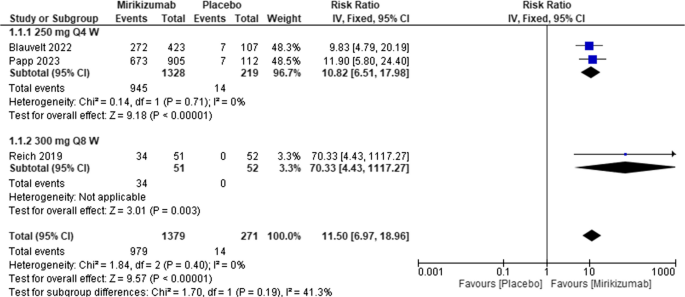
Forest plot of PASI 90 in the mirikizumab group vs. placebo
Psoriasis area and severity index (PASI 100, responders) at week 16
Miri produced substantially more significant improvements in PASI 100 compared to placebo [RR = 25.94, 95% CI (9.14, 73.66), P < 0.00001]. There was no heterogeneity among the included studies (I2 = 0%; P for Cochran Q = 0.90). Figure 6
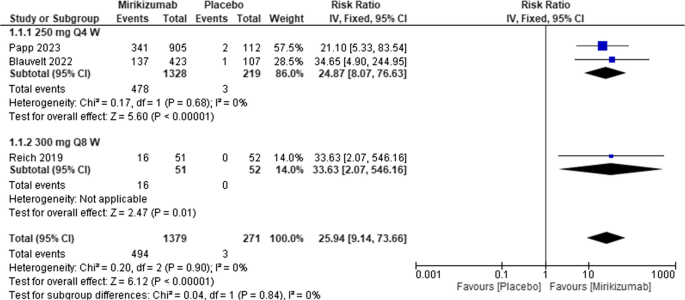
Forest plot of PASI 100 in the mirikizumab group vs. placebo
Dermatology life quality index DLQI (0,1)
DLQI was reported by two studies showing that mirikizumab was associated with higher DLQI compared to placebo [RR = 10.94, 95% CI (5.12, 22.03), P < 0.00001]. There was no heterogeneity among the included studies (I2 = 0%; P = 0.81). (S1 in Supplements).
Static physician’s global assessment sPGA score
Patients treated with mirikizumab had a higher sPGA score than patients managed with a placebo [RR = 12.48, 95% CI (7.63, 20.40), P < 0.00001]. There was no heterogeneity among the included studies (I2 = 0%; P for Cochran Q = 0.52). Figure 7
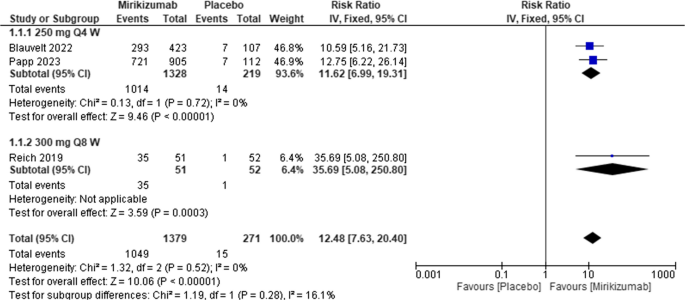
Forest plot of static Physician’s Global Assessment in the mirikizumab group vs. placebo
Body surface area (BSA)
The results found that patients receiving mirikizumab were much more likely to attain better BSA outcomes than those on placebo [RR = 34.53, 95% CI (13.03, 91.51), P < 0.00001]. The pooled data were homogenous (I2 = 0%; P = 0.89). (S2 in Supplements).
Overall adverse events
We found no significant differences in overall adverse events between the two groups [RR = 0.97, 95% CI (0.86, 1.1), P = 0.63]. No heterogeneity was found among the included studies (I2 = 0%; P for Cochran Q = 0.96). (S3 in Supplements).
Discontinuation from treatment due to AEs
The overall results showed that mirikizumab did not considerably lead to discontinuing treatment compared to placebo [RR = 0.68, 95% CI (0.14, 3.2), P = 0.63]. The analysis was consistent (I2 = 0%; P for Cochran Q = 0.90). (S4 in Supplements).
Infection
The risk of infection did not differ between the two regimens [RR = 1.11, 95% CI (0.89, 1.38), P = 0.36]. The included studies were homogenous (I2 = 0%; P for Cochran Q = 0.41). (S5 in Supplements).
Upper respiratory tract infection
Mirikizumab did not increase the risk of URTI compared to placebo [RR = 1.61, 95% CI (0.85, 3.04), P = 0.14]. The analysis was consistent (I2 = 0%; P for Cochran Q = 0.60). (S6 in Supplements).
Malignancies
Treating psoriatic patients with Mirikizumab did not reveal an increase in the risk of malignancy compared to placebo [RR = 0.45, 95% CI (0.06, 3.40), P = 0.44]. No heterogeneity was found (I2 = 0%; P for Cochran Q = 0.55). (S7 in Supplements).
Injection site reaction
The overall effect revealed that injection site reaction was similar between the two interventions [RR = 1.22, 95% CI (0.68, 2.19), P = 0.51]. The pooled analysis was homogeneous (I2 = 0%; P for Cochran Q = 0.67). (S8 in Supplements).
Discussion
Psoriasis is considered one of the emerging skin disorders that affects patients’ lives. Nowadays, biological therapies have shown significant results in managing psoriasis. Different types of biologics showed promising results in plaque psoriasis, besides arthritic psoriasis, such as guselkumab, risankizumab, and tildrakizumab, which target the same pathway (Raharja et al. 2021; Salimi et al. 2020; Liu et al. 2020; Menter et al. 2021).
Our results consistently demonstrate that Miri is highly effective in managing psoriasis at 16 weeks, with many patients achieving PASI 75, PASI 90, PASI 100, BSA, and DLQI than placebo.
Nevertheless, there is a high potential for developing malignancies and continuous infections due to its immunosuppression effect; our results revealed no difference between placebo and Miri in developing infections or malignancies.
Another systematic review and meta-analysis that evaluated the safety and efficacy of IL-17, IL-12/23, and IL-23 inhibitors in the management of moderate to severe plaque psoriasis produced similar findings (Bilal et al. 2018).
Previous studies on guselkumab and risankizumab also reported strong PASI 90 responses in moderate-to-severe psoriasis. Several studies on guselkumab demonstrated a significant proportion of patients attained PASI 90, which is comparable to the results seen with mirikizumab (Blauvelt et al. 2017; Reich et al. 2017).
The rapid response with mirikizumab also parallels earlier findings from trials on risankizumab. Both drugs, through selective IL-23 inhibition, offer rapid reductions in psoriasis symptoms, with many patients experiencing significant improvement within the first 12 weeks. In the OASIS-1 study, results showed that mirikizumab effectively cleared skin plaques within 16 weeks and maintained this improvement for up to 52 weeks in psoriasis patients. Also, it is important to achieve patient satisfaction (Blauvelt et al. 2022 Dec 1; Gordon et al. 2018).
Mirikizumab is considered one of the broader treatments for plaque psoriasis and personalized therapies. Other biologics, like IL-17 and TNF inhibitors (adalimumab, infliximab, secukinumab, ixekizumab, etc.), revolutionized psoriasis treatment by offering more specific immune modulation compared to traditional systemic therapies like methotrexate or cyclosporine. However, the recent IL-23 inhibitors, including mirikizumab, offer even more precise targeting of the inflammatory pathways directly implicated pathophysiology of psoriasis, leading to improved efficacy and fewer adverse effects (Salimi et al. 2020; Papp et al. 2023; Bilal et al. 2018; Gordon et al. 2018).
Papp et al. compared Miri with secukinumab; they found that Miri was better than secukinumab at week 16, achieving a PASI 90 response in 74.4% and 72.8%, respectively (Papp et al. 2023).
Previous studies have shown that IL-17 inhibitors also achieve rapid skin clearance, but their mechanism targets the end effector of the IL-23/IL-17 pathway. IL-23 inhibitors, like Miri, block the upstream of cytokines, potentially offering more sustained efficacy with fewer immune-related side effects, which has led to increased interest in IL-23 inhibitors for long-term psoriasis management, with some evidence suggesting a lower relapse rate compared to IL-17 inhibitors (Liu et al. 2020; Menter et al. 2021).
Our meta-analysis has numerous strengths, and it is the first meta-analysis to assess the efficacy of Miri in plaque psoriasis. First, we included studies that were of high quality. Additionally, the consistency of results indicated by pooling data homogeneity strengthens the conclusions’ validity, making them more generalizable across populations. Besides, the study specifically addressed different outcomes, thus providing a detailed review regarding the drug.
However, despite these advantages, several limitations must be recognized. For example, the first limitation could be seen in having only three RCTs for analysis. Even though pooled analyses showed homogeneity, concerns were raised because only a few papers were considered in this analysis, which may raise some publication bias.
There were numerous abstracts discussing the efficacy of mirikizumab, but full texts have not been published yet.
In addition, our analysis focused mainly on short-term outcomes at 16 weeks, meaning we never took care of the long-term safety and efficacy of miri due to incomplete data and different dosages.
Subgroup analyses based on the dosage further supported its efficacy, with notable improvements observed in PASI scores favoring Miri over the placebo.
Regarding safety outcomes, it demonstrated no significant differences in adverse events, highlighting its favorable safety profile in comparison to the placebo.
Based on our findings, several recommendations can be made for future research and clinical practice. First, we recommend conducting large-scale RCTs with standardized protocols and more extended follow-up periods, which can provide more definitive insights into mirikizumab’s long-term efficacy and safety profile.
Additionally, exploring patient-specific factors, such as genetic markers or biomarkers, may help identify subpopulations that are more likely to benefit from the therapy, leading to personalized treatment approaches. Also, studies have been conducted that compare mirikizumab and IL-17/other IL-23 inhibitors.
Conclusion
This study showed that mirikizumab is safer and more effective in patients with plaque psoriasis than placebo. Adverse events were not of significant value with mirikizumab. However, it is still unclear if the drug will be safe in the long term. Finally, mirikizumab significantly improves the PASI score, body surface area, and DLQI at 16 weeks. We recommend further studies to demonstrate the safety of mirikizumab in psoriasis patients, focusing on determining the ideal dosage, exploring potential supplement combinations, and evaluating any potential side effects for a more thorough understanding of the treatment.
