Article Content
Introduction
In Japan, the national health insurance system partially covers patients’ medical expenses through public funding [1]. However, with advances in medical care and an aging population, medical expenses are increasing annually [2]. Notably, drug expenses alone exceed JPY 9 trillion. Consequently, measures to reduce drug costs can effectively lower overall medical expenses.
Various injectable drugs require dosage adjustments based on the patient’s body weight or body surface area. Because anticancer drugs are administered near the maximum tolerated dose, the therapeutic range—the margin between the effective antitumor dose and the dose causing adverse events—is significantly narrower compared to conventional drugs. Therefore, the appropriate dosage is carefully calculated based on the patient’s body weight or body surface area [3]. This practice often leads to the drug residue remaining in the vial after preparation. Even for expensive anticancer drugs, some of the drug in the vial is used, whereas the remainder is discarded. As billing is permitted for the entire vial even if not fully used, the patient and insurer must cover the cost of the discarded drug, leading to economic loss [4, 5].
Although drug vial optimization––which allows the distribution of drug vials among multiple patients––has been approved, it remains uncommon in hospitals due to limited economic benefits [4, 5]. Additionally, drugs are marketed in various vial sizes, and changing vial sizes can help reduce drug expenses [6, 7]. However, these regulations have not been reviewed. Pharmaceutical companies that manufacture these drugs incur costs for vials, rubber stoppers, and other materials. Given the development and manufacturing costs, small standard vials may not be feasible due to potential losses in sales. Thus, changing vial standards requires support from pharmaceutical companies, as medical institutions alone cannot effect this change.
Recently, there has been a shift towards not producing drug residues rather than merely reducing them. When the planned dosage of an anticancer drug is slightly higher than the product specification unit, the dosage is adjusted to fit within the product specification unit by slightly reducing the dosage. In 2018, the Hematology/Oncology Pharmacy Association (HOPA) and the American Society of Pharmacists issued a statement [8] aiming to reduce medical costs and waste by adjusting the dosage within a range of 10% and determined the dosage per product unit. Several studies have been conducted on the relationship between dose adjustment and therapeutic efficacy, indicating that adjustments in the range of 10% for antibody products and 5% for cytotoxic anticancer drugs do not alter efficacy [8–12]. ‘Many monoclonal antibodies, in contrast to cytotoxic chemotherapeutic agents, never reach a maximum tolerated dose (MTD), and there may be a “just enough” dose needed to saturate the receptors. For example, in the dose range of bevacizumab in colon cancer, it was initially used at 10 mg/kg, but similar efficacy has been achieved with a dose of 5 mg/kg.’
However, no such assessment has been conducted in Japan. Various medical information databases exist in Japan [13–15], but they are based on receipt or documentary evidence, making it challenging to obtain accurate dosage data as it is often mixed with vial-based billing units. Therefore, collecting accurate data from medical records is essential to determine exact dosages in each facility.
Moreover, the top-selling drugs are immune checkpoint inhibitors, which are administered at fixed doses and do not require adjustments. Among injectable drugs that require dose adjustments, bevacizumab is the most commonly sold. Therefore, this study investigated the impact of dose adjustment on medical costs for bevacizumab. This study was conducted in Japan because body size, a factor that influences dosage, varies by race, and the system of medical costs differs by country.
Materials and methods
Investigation of usage results using NDB open data
We collected annual usage data for bevacizumab from the National Database of Health (NDB) Open Data. The NDB Open Data provides statistical information on medical care in Japan and results from specific health checkups, as provided by the Ministry of Health, Labor, and Welfare since fiscal year (FY) 2014. Drug data were categorized into outpatient (in-hospital), outpatient (out-of-hospital), and inpatient treatment, with dosage forms including oral, topical, and injection. Additionally, the actual consumption of the top 30 most frequently used drugs was summarized using drug effects categories. The data were published as prescription quantities (including the number of tablets and capsules). For this study, we used drug data from FY 2014 to FY 2021, which are publicly available through NDB Open Data. Annual drug prices were used to calculate expenses. This dataset was used in the study of trends in bevacizumab use in Japan in Fig. 1. As all personal data were excluded from the NDB Open Data, ethics committee approval was not required for this analysis.
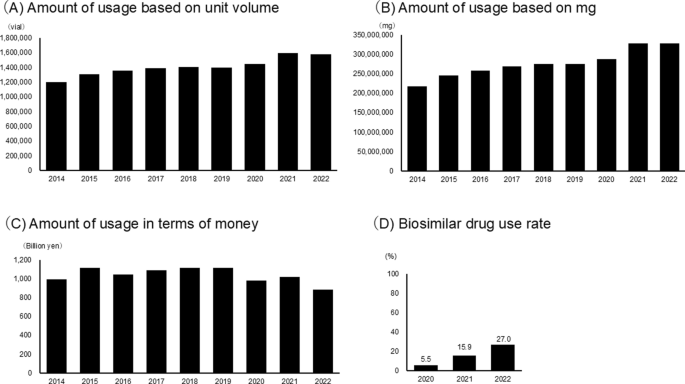
Trends in bevacizumab use in Japan. Annual bevacizumab use in Japan from 2014 to 2021, based on the National Database of Health (NDB) Open Data. a Number of vials used per year. b Total annual cost (in JPY 100 million). c Total mg administered per year. d Percentage of biosimilar drugs used
Assessment based on medical record data from university hospitals
This multicentre, retrospective study was conducted at four national university hospitals in Tokushima, Okayama, Kagawa, and Asahikawa. It included cancer patients treated with bevacizumab between January 2011 and December 2023. We recorded the year of bevacizumab administration and dosage. Personal data, such as individual identity (ID), age, sex, and disease name, were excluded from this analysis.
The results for surveys other than those shown in Fig. 1 are the result of analysing the data obtained here.
We assessed changes in drug expenses when doses were adjusted. Three percentages were selected: 10%, the dose considered to have no effect on therapeutic efficacy with antibody preparations [8]; 5%, the dose considered to have no effect on therapeutic efficacy with cytotoxic anticancer agents; and 2%, about half of the dose with antibody preparations, which is low. Drug costs were calculated based on drug prices in March 2024. The analysis summary is shown in Fig. 2.
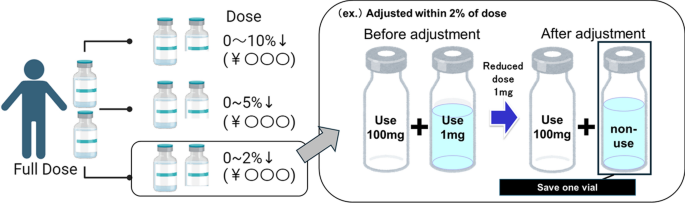
Dose adjustment simulation. An overview of the method for calculating drug costs when the dose is reduced by each adjustment rate relative to the maximum dose is presented
When the 10% range is set as the maximum value, we assume that if we reduce the dose by 10% from the actual dose obtained, we reduce the dose if the number of vials required for dosing is reduced. The percentage reduction from the actual dose is then determined when the dose reduction was stopped to the maximum amount filled in the vial.
For example, for a person receiving a 310 mg dose, a 10% dose reduction would be 279 mg. However, since bevacizumab is in 100 mg vials, the dose was calculated to be 300 mg, the maximum dose that would not produce residual drug.
For national-scale applications, simulations were performed using NDB data. The average value for each year was used as facility data, and NDB data collected from 2014 to 2021 were used to determine percentages. The latest percentage (27.0% in FY 2022) of biosimilar use was applied. In addition, we have also simulated the case of 60%, 80%100% market penetration to take into account the possibility of biosimilar penetration in the future.
Results
Trends in bevacizumab use in Japan
Bevacizumab use was analysed using NDB Open Data (Fig. 1). A total of 12.7 million vials, or 2.49 billion mg, were used during the collection period, with a total drug cost amounting to JPY 935 billion. The usage rates for each year were as follows: 2014: 1.2 million vials, JPY 99.2 billion, and 220 million mg; 2015: 1.31 million vials, JPY 111.8 billion, and 250 million mg; 2016: 1.36 million vials, JPY 104.7 billion, and 260 million mg; 2017: 1.39 million vials, JPY 109 billion, and 270 million mg; 2018: 1.41 million vials, JPY 111.4 billion, and 280 million mg; 2019: 1.4 million vials, JPY 111.2 billion, and 280 million mg; 2020: 1.45 million vials, JPY 98 billion, and 290 million mg; 2021: 1.59 million vials, JPY 101.6 billion, and 330 million mg and 2022: 1.58 million vials, JPY 881.4 billion, and 330 million mg
Analysis of bevacizumab use based on facility data
We analysed facility medical record data (Fig. 3). Figure 3a illustrates the annual trends in usage. The total number of patients and doses were as follows: 2011: 1,919 patients, 973,301 mg; 2012: 2,302 patients, 1,172,938 mg; 2013: 2,609 patients, 1,329,621 mg; 2014: 3,112 patients, 1,693,761 mg; 2015: 3,298 patients, 1,772,712 mg; 2016: 3,692 patients, 1,953,136 mg; 2017: 3,666 patients, 1,925,776 mg; 2018: 3,888 patients, 2,134,213 mg; 2019: 4,330 patients, 2,346,194 mg; 2020: 4,732 patients, 2,631,974 mg; 2021: 5,109 patients, 3,071,122 mg; 2022: 5,557 patients, 3,380,365 mg; 2023: 4,868 patients, 2,805,390 mg. Figure 3b illustrates the distribution of doses. The highest frequency was for 201–300 mg in 9,639 patients, followed by 301–400 mg in 7,546 patients.
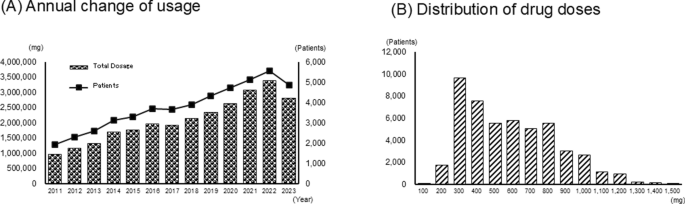
Bevacizumab usage analysis using data from medical records. a Annual changes in the number of patients treated with bevacizumab and annual trends in total dose (mg) at four national university hospitals. b Dose distribution per patient
Reducing medical expenses through dosage adjustments
There were 49,082 eligible patients with a total dose of 27,190,502 mg, with an average dose of 554 mg per patient (Table 1). The cost per dose was JPY 171,234 for branded drugs and JPY 63,118 for biosimilar drugs. A total of 1,681,124 mg of the drug was discarded, averaging 34 mg per administration.
Change in dose after adjustment
With dose adjustments of 10%, 5%, and 2%, the percentage of patients who underwent dose reduction was 36%, 14%, and 3%, respectively (Table 2). The average dose reduction was 6%, 3%, and 1%, respectively. The average reduction in drug expenses was JPY 10,163, JPY 3,910, and JPY 924 per patient, respectively, when converted to branded drugs. This resulted in total reductions of approximately JPY 499, JPY 192, and JPY 45 million, respectively. Figure 4 illustrates the distribution of percentage reductions.
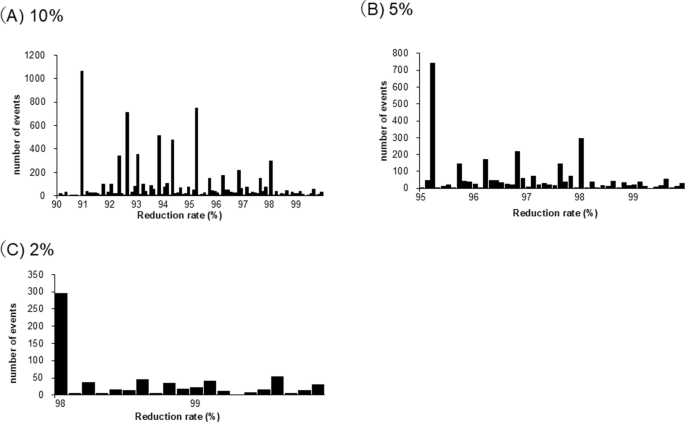
Reducing medical expenses through dosage adjustments. Effects of dose adjustment on bevacizumab healthcare costs. Average cost savings per patient for brand-name drugs at dose adjustment levels of 10%, 5%, and 2%
Estimation of bevacizumab dosage adjustment for national use
In this assessment, the average amount of bevacizumab administered during 2014–2021 was 0.83% of the national dose (Table 3). To estimate national reductions in drug expenses, we assumed that bevacizumab usage nationwide mirrored that of the four centres. Using the latest data on biosimilar use (27.0% in 2022), the drug expense savings from dose adjustments of 10%, 5%, and 2% were estimated to be JPY 3,833,109,219, JPY 1,474,725,764, and JPY 348,444,279, respectively. In addition, we examined the cost reductions with increasing rates of biosimilar use. Although the amount of cost reduction decreased with an increase in the use of biosimilars, even when 100% biosimilars were used, the drug expense savings from dose adjustments of 10%, 5%, and 2% were estimated to be JPY 1,706,401,336, JPY 656,277,993, and JPY 154,933,146, respectively.
Discussion
Rounding of anticancer drug doses, which may be effective in reducing medical costs, has not been verified in Japan. Thus, in this study, we examined the impact of dose adjustment on medical costs for bevacizumab, one of the most used drugs by volume.
A major consideration in dose adjustment is its effect on therapeutic efficacy. For various anticancer agents, dose reductions of over 20–30% can affect therapeutic efficacy, whereas adjustments of 5–10% or less generally have no negative effects on therapeutic efficacy [8]. The degree of dose adjustment has been demonstrated at various cutoff values, such as 5% and 10% [8–12]. Therefore, we conducted simulations on the degree of alterations in drug expenses in Japan using three different cutoff values: 5%, 10%, and 2%, which are specified in the dispensing guidelines as the upper limit of the margin of error for dispensed drugs.
Initially, we used NDB Open Data to confirm trends in the nationwide use of bevacizumab, the drug under study. Due to periodic NHI price revisions and the introduction of biosimilar drugs, the annual change in the value base shows an increase or decrease, but the usage volume increases from year to year. Additionally, medical record survey data indicated that bevacizumab use increased approximately 1.6-fold in 2021 compared to 2014, a trend similar to the rate of increase observed between the first and last years of NDB Open Data.
Bevacizumab is approved for various cancer types [16] and is administered in doses of 5–15 mg/kg. The average weight of Japanese adults varies depending on age and sex; however, the typical range is 48–73 kg [17]. Consequently, bevacizumab doses are expected to range between 250 and 1,100 mg, which is consistent with the results from medical record survey data. Based on these findings, the dosages used in this assessment are considered to reflect the actual use in Japan.
We conducted a simulation using survey data from medical records to evaluate potential cost savings through dose adjustments. The reduction in drug expenses averaged JPY 9.6, JPY 3.7 million, and JPY 0.87 million per facility per year for branded drugs at adjustment ranges of 10%, 5%, and 2%, respectively.
Additionally, biosimilar drugs have been approved globally. In Japan, the national target is to replace at least 80% of drug ingredients with biosimilars, aiming to achieve a minimum of 60% of the total ingredient count in biosimilar drugs by the end of FY 2029 [18]. Therefore, the use of biosimilar formulations is anticipated to increase. We estimated the potential reduction in drug expenses if all bevacizumab formulations were replaced with biosimilar formulations. Based on our analysis, we expect reductions of JPY 3.5 million, JPY 1.4 million, and JPY 0.32 million per facility per year with adjustment margins of 10%, 5%, and 2%, respectively.
The NDB Open Data were calculated for each vial individually and exceeded the actual amount used. Consequently, the actual reduction in drug usage may be greater, potentially resulting in a greater reduction in drug expenses.
Focusing on the degree of dose reduction across subjects, the average decrease was 6%, 3%, and 1% for adjustment ranges of 10%, 5%, and 2%, respectively. These reductions are approximately half of the maximum dose reduction and may not significantly impact the treatment efficacy in the population. However, certain patients experienced a maximum dose reduction of 10%. The optimal adjustment range and potential fluctuations in treatment effects should be carefully considered before implementing similar measures in clinical practice. Additionally, it is necessary to evaluate the measures to prevent medical accidents. Even with dosage adjustments of ≤2%, which correspond to the error range specified in the dispensing guidelines, a nationwide reduction in drug costs exceeding JPY 100 million can be expected, indicating substantial potential savings.
The data used in this study represent approximately 1% of Japan’s total usage of bevacizumab and are based on university hospitals. Therefore, due to regional differences, facility size, and other factors, these data may not accurately reflect actual usage. For a more accurate assessment of the current situation, an evaluation with more study sites is needed. Evaluation should also be conducted with drugs other than bevacizumab. Additionally, in Japan, patients who receive reduced doses often have underlying factors that necessitate these adjustments. Consequently, it is challenging to determine from previous patients how much dose reduction would not impact therapeutic efficacy. This issue must be assessed alongside medical economic evaluations. Clarifying the dose at which therapeutic efficacy is maintained even with a reduction is valuable, as it can also contribute to reducing the occurrence of side effects.
Similar to the NDB Open Data, medical record data show that bevacizumab use has increased annually, with usage expected to triple by 2022 compared to 2011. Bevacizumab is approved for combination therapy with immune checkpoint inhibitors [19], and numerous clinical trials have since been conducted for additional indications [20, 21]. As a result, the use of bevacizumab in cancer chemotherapy is increasing, and the reduction in drug expenses may exceed the estimated costs. Expanding similar efforts to other anticancer drugs could result in further cost reductions.
