Article Content
1 Introduction
There has been an increasing level of interest in using mixed oxides having the perovskite structure as supports for metal catalysts. The concept, first reported by researchers at Daihatsu [1,2,3], was that catalysts which had undergone deactivation due to metal sintering could be regenerated by sequential oxidation and reduction at high temperatures. The reason this could reactivate the catalyst is that, with some metal-perovskite combinations [4], metal cations can enter the perovskite lattice under oxidizing conditions and then be exsolved as small metal particles upon reduction. Later studies showed that the exsolved metals can have unique catalytic properties. For example, unlike Ni particles on traditional supports, Ni exsolved from La0.46Sr0.34Ni0.03Ti0.97O3 does not form carbon whiskers when heated in dry methane [5, 6].
Unfortunately, several factors have limited the commercial application of perovskite-supported metals [7]. First, most of the perovskites of interest have low surface areas compared to conventional supports, such as γ-Al2O3 or SiO2. More importantly, a number of studies have shown that a significant fraction of the exsolved metal often remains embedded in the bulk of the perovskite and unavailable for catalyzing reactions [8,9,10]. For at least some metal-perovskite pairs, reduced-metal particles first form in the bulk of the mixed oxide and are only then “squeezed” to the surface in a separate step that is often very slow [9, 11, 12].
To circumvent both problems with perovskite supports, our groups have been preparing thin films of mixed oxides on more stable supports, such as Ca-doped γ-Al2O3 [11, 13] and MgAl2O4 [12, 14, 15], using Atomic Layer Deposition (ALD). With ALD, a wide range of oxides can be deposited uniformly, with well-defined coverages. By depositing alternating cycles of the two oxides that make up the perovskite, we have prepared thin films in which Ca-doped γ-Al2O3 or MgAl2O4 are completely covered by perovskite films between 0.5 and 2-nm in thickness. Metals deposited on these thin films show many of the expected characteristics of exsolution catalysts. For example, Ni catalysts oxidized at 1073 K and reduced at 773 K were inactive for steam reforming; but the activity of these catalysts could be restored by reduction at 1073 K [6, 16]. Also, Ni showed extreme tolerance against carbon whisker formation when supported on CaTiO3 and LaMnO3, even when exposed to dry methane at 1073 K [6, 16].
Two perovskites that have received relatively little attention for application as exsolution supports are BaCeO3 and BaZrO3. These perovskites are of interest because incorporation of catalytic metals into the lattices of these two perovskites should be facilitated by the large size of the A- and B-site cations [17,18,19,20]. Some representative studies have shown that these two materials could have interesting properties. For example, catalysts prepared by Pd exsolution from BaCeO3 were reported to be highly active for CO oxidation and were unusual in that Pd appeared to be extruded as “nanowires” [18]. In another study, high-temperature reduction of Ba(Ce0.9Y0.1)0.8Ni0.2O3−δ gave rise to carbon-resistant anodes for Solid-Oxide Fuel Cells (SOFC) [21]. That BaZrO3 could also serve as an exsolution support was demonstrated by early work showing that Rh can be inserted into the BaZrO3 lattice [4].
Our interest in these materials stems from the fact that both BaCeO3 and BaZrO3, when doped with a + 3 ion like yttria, can be proton conductors in the presence of water vapor due to migration of hydroxyls within the bulk [22,23,24]. It is possible that the mobility of hydrogen in the bulk of these materials may allow them to promote metal-catalyzed reactions involving steam, such as methane-steam reforming (MSR). To avoid any ambiguity associated with CeO2-assisted reactions [25], we have restricted our focus to BaZrO3, which has no easily reducible cations.
In this study, we have prepared a BaZrO3-supported Ni catalyst and compared its MSR performance to that of other perovskite-supported catalysts. The BaZrO3 was again prepared as a thin film on a more stable oxide to avoid having exsolved metal remain embedded in the bulk of the perovskite [8,9,10]. We will show that the BaZrO3-supported, Ni catalysts exhibit higher rates for the MSR reaction compared to other perovskite-supported, Ni catalysts, together with excellent tolerance against coking.
2 Experimental Methods
2.1 Sample Preparation
The catalysts were all supported on MgAl2O4 (MAO) that was prepared in our laboratory and is described in more detail elsewhere [6]. This support material had a surface area of 120 m2/g, and its XRD pattern showed only the characteristic peaks for the spinel structure. The ALD precursors were bis(2,2,6,6-tetramethyl-3,5-heptanedionato) barium hydrate (Ba(TMHD)2·xH2O, Strem, USA), Tetrakis(2,2,6,6-tetramethyl-3,5-heptanedionato)zirconium(IV), (Zr(TMHD)4, Strem, USA), and bis(2,2,6,6-tetramethyl-3,5-heptanedionato) nickel(II) (Ni(TMHD)2, Strem, USA).
Details on the ALD equipment and catalyst-synthesis procedures have been discussed elsewhere [6]. In these experiments, the MAO and precursor powders, separated by glass wool, were placed in a stainless-steel tube that could be evacuated and sealed. The temperature of the sealed tube was then raised to 543 K (for Zr and Ni) or 573 K (for Ba) to vaporize the precursor, after which excess vapor was removed by evacuation. The MAO that had been exposed to precursor vapor was then placed in a muffle furnace at 823 K. This procedure was repeated for as many cycles as needed to obtain the desired film thickness. Growth rates were determined by measuring the sample weight after each cycle and were found to be 3.3 × 1017 Ba/m2-cycle [26] and 8.1 × 1017 Zr/m2-cycle [27], in agreement with previous measurements. To obtain the correct stoichiometry for BaZrO3, we repeatedly deposited 2 cycles of ZrO2, followed by 5 cycles of BaO, to form one supercycle of BaZrO3. Ni was incorporated onto oxide supports using 1 ALD cycle with Ni(TMHD)2.
X-ray diffraction (XRD) patterns were recorded on a Rigaku MiniFlex diffractometer equipped with a Cu Kα source (λ = 0.15416 nm). Rietveld analysis of the XRD patterns was accomplished using Profex 5.4.1. Scanning Transmission Electron Microscopy (STEM) measurements were performed on powder specimens that had been diluted in isopropanol and then deposited onto carbon support films on copper grids (Electron Microscopy Sciences). Imaging and elemental mapping via Energy Dispersive X-ray Spectroscopy (EDS) were performed with a JEOL NEOARM operated at 200 kV. Surface areas were measured using BET isotherms for N2 in a homemade apparatus.
Rates for Methane Steam Reforming (MSR) were determined with differential conversions in a 0.25-inch, quartz, tubular-flow reactor at atmospheric pressure, using 100 mg of catalyst. The total gas flow rate was maintained at 120 mL min− 1, with partial pressures of 18 torr H2O and 18 Torr CH4, the balance being He. The H2O partial pressure was maintained by flowing the He through a temperature-controlled bubbler. Products were analyzed using a gas chromatograph (SRI 8610 C) equipped with a TCD detector. Conversions were less than 10% for all rate measurements, ensuring that the measurements were performed under differential conditions. To investigate the effect of catalyst pretreatments, the samples were either oxidized in flowing 10% O2:He mixtures at 1073 K for 1 h or reduced in 10% H2:He mixtures at the same temperature.
3 Results and Discussion
To achieve the targeted loadings of 27- and 43-wt% BaZrO3, 4 or 8 supercycles (8 or 16 cycles of ZrO2 and 20 or 40 cycles of BaO) were deposited onto the MAO. Assuming the films had the same density as the bulk perovskite and were deposited uniformly over the entire MAO surface, these weight loadings correspond to film thicknesses of 0.5 and 1.0 nm. These samples will be referred to as 0.5 BaZrO3/MAO and 1.0 BaZrO3/MAO, respectively, and are listed in Table 1 along with some of their physical properties. After deposition, both BaZrO3/MAO samples were subjected to five cycles of calcining in air for 2 h at 1073 K followed by reduction in a 10% H2-He mixture for 2 h, to promote formation of the perovskite phase. Following the redox treatments, the BET surface areas were 120 m2/g for MAO, 76 m2/g for 0.5 BaZrO3/MAO, and 44 m2/g for 1.0 BaZrO3/MAO samples. The decreased surface areas for the BaZrO3-containing samples were primarily due to the increase in mass associated with the films.
STEM-EDS measurements were performed on the 1.0 BaZrO3/MAO sample, with results shown in Fig. 1. The STEM image is essentially identical to that observed for MAO without a BaZrO3 film. Despite the relatively high BaZrO3 loading, there were no obvious features that could be associated with the perovskite. The diffraction lines in the image are difficult to distinguish from those associated with the MAO structure. The EDS maps clearly show that both Ba and Zr are evenly distributed over the MAO surface, with no evidence for segregation on the 2-nm length scale.

STEM image with EDS mappings of 1.0 BaZrO3/MAO after 5 redox cycles at 1073 K. The red box indicates the EDS region
To characterize the structure of the films, XRD measurements were performed, with patterns for the various samples reported in Fig. 2, together with reference patterns. The pattern for the 0.5 BaZrO3/MAO sample is identical to that reported for unmodified MAO, implying that the film is either x-ray amorphous or that the crystallite length scale for the perovskite phase is so small that the peaks are broadened into the baseline. The XRD pattern for the 1.0 BaZrO3/MAO sample is also dominated by the diffraction lines for MgAl2O4; but additional features, especially a large feature at ~ 28° 2θ, are also present that can be assigned to a BaZrO3 perovskite phase. Note that XRD measurements on the as-deposited BaZrO3/MAO sample show an amorphous phase with no distinct peaks attributable to BaZrO3.
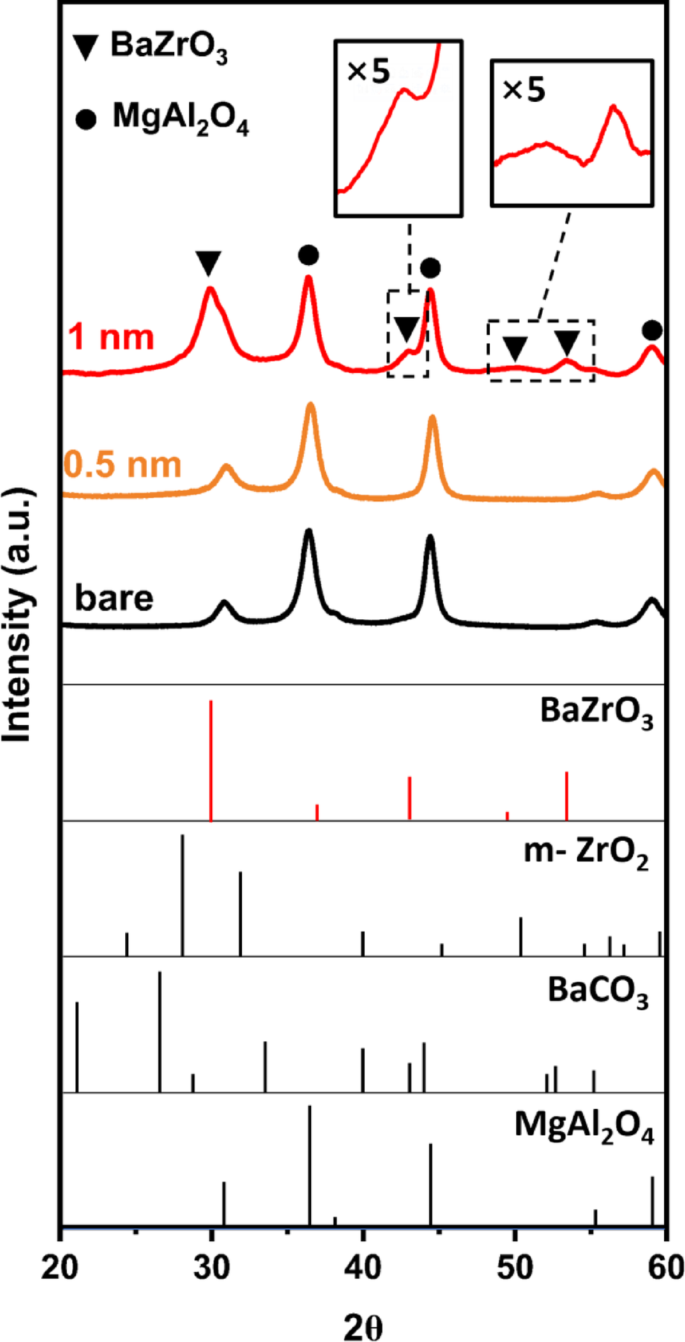
XRD of the 1.0 BaZrO3/MAO, 0.5 BaZrO3/MAO, and MAO samples, after 5 redox cycle at 1073 K. Standard reference patterns are shown for BaZrO3, monoclinic ZrO2, BaCO3, and MgAl2O4
Rietveld refinement was used to extract additional information from the XRD pattern for the 1-nm film, with results shown in Fig. 3; Table 2. The analysis demonstrates that one can fit the pattern to a physical mixture of roughly 85-wt% MgAl2O4 crystallites (13.5 nm in size) and 15-wt% BaZrO3 crystallites (7 nm in size). First, it is important to recognize that the crystallite size for BaZrO3 obtained from Rietveld analysis is much greater than the average film thickness. Since the STEM results indicate that the BaZrO3 film covers the MAO uniformly, with an average thickness of only 1.0 nm, the crystallite size of the perovskite phase must correspond to the size of planar, two-dimensional crystallites. In previous studies of other perovskite films formed by ALD, it was also found that the surface was covered with two-dimensional crystallites in random crystallographic orientations [11, 28]. Second, the Rietveld analysis clearly underestimated the BaZrO3 loading. Although this may be due to poor crystallinity of the perovskite phase, it is important to recognize that Rietveld analysis does not take into account that the BaZrO3 phase is present as a two-dimensional film. Finally, the results for BaZrO3 are similar to what was reported in a previous study of LaFeO3 films on MAO [12]. That study also reported that the intensity of diffraction peaks associated with the perovskite phase increased in a nonlinear manner with LaFeO3 loading [12].
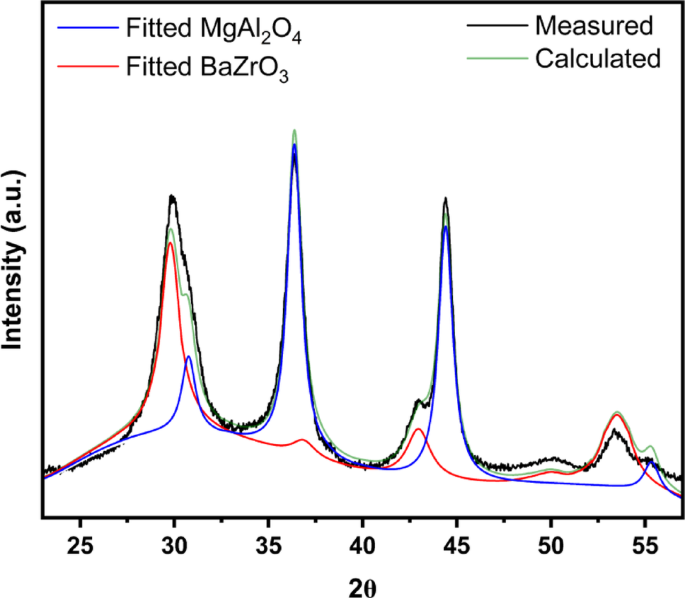
Rietveld analysis of the XRD pattern of the 1.0 BaZrO3/MAO
To understand how BaZrO3 affects the catalytic properties of Ni, we measured MSR rates on the 1.0 BaZrO3/MAO sample after depositing Ni using one ALD cycle (~ 1 wt%). Reaction conditions were chosen so that rates could be compared to previously published data obtained on 1-wt% Ni catalysts supported on unmodified MAO and MAO with a 1-nm film of CaTiO3 [6]. A 1:1 steam-to-methane ratio was used in the reactivity studies so the propensity for the catalyst to deactivate by coking could be explored (water is known to suppress coking at high steam to methane ratios). The Ni/MAO and Ni/CaTiO3/MAO catalysts were prepared using identical methods as those employed for the Ni/BaZrO3/MAO catalyst. A list of properties for all three catalysts is shown in Table 1. Rates were determined in a flow reactor, with conversions kept under 10%, using 2.4% CH4 and 2.4% H2O in He.
Differential reaction rates for 1.0 BaZrO3/MAO with 1-wt% Ni, 1Ni/BaZrO3/MAO, are shown in Fig. 4a) following various pretreatments. Similar to what was reported in studies with other perovskite-supported, Ni catalysts, rates were very low following oxidation of the catalyst at 1073 K [6]. Rates on this catalyst did not increase after reduction at 773 K, but increased dramatically following reduction at 1073 K. While it was not possible for us to measure rates at the same temperature on the oxidized and reduced catalysts, extrapolation of the Arrhenius plots indicates that the rates increased by more than four orders of magnitude upon reduction. Oxidation at 1073 K of the active, reduced catalyst returned the catalyst to its inactive form; and reduction of the inactive, oxidized catalyst returned it to the active form. There was no overall change in the catalyst following five oxidation/reduction cycles. These results are consistent with an exsolution process in which Ni is inserted into the BaZrO3 lattice following oxidation at 1073 K. Reduction at 773 K is insufficient to reduce the Ni and pull it out of the lattice, but Ni is released upon reduction at 1073 K. Catalytic activity is completely restored following the ingress/egress of the Ni. During rate measurements, each catalyst was tested for more than 12 h without showing any measurable deactivation. This observation indicates that the catalyst remains stable under the reaction conditions employed.
It is interesting to compare these MSR rates to previously measured rates on 1-wt% Ni/MAO and 1-wt% Ni/CaTiO3/MAO. Rates on each of these catalysts were measured in the same reactor, under similar conditions, and are summarized in Fig. 4b). Considering Ni/MAO first, even the rates on a freshly-prepared catalyst were significantly lower than those on reduced Ni/BaZrO3/MAO at the temperatures used in these measurements. The activation energy, 80 ± 10 kJ/mol, was also lower than that observed on Ni/BaZrO3/MAO, 180 ± 10 kJ/mol kJ/mol. Furthermore, reducing the Ni/MAO at 773 K was sufficient to activate the catalyst; and increasing the reduction temperature to 1073 K did not significantly affect reaction rates.
The Ni/BaZrO3/MAO and Ni/CaTiO3/MAO samples exhibited some similarities but also some major differences. First, similar to observations with Ni/BaZrO3/MAO, rates on Ni/CaTiO3/MAO oxidized at 1073 K were lower than rates on Ni/CaTiO3/MAO reduced at 1073 K. As with Ni/BaZrO3/MAO, reduction at 773 K was insufficient to activate an oxidized Ni/CaTiO3/MAO, and the catalytic activity of Ni/CaTiO3/MAO was maintained following repeated redox cycles at 1073 K. There were also several major differences between Ni/BaZrO3/MAO and Ni/CaTiO3/MAO. Rates on the reduced Ni/CaTiO3/MAO exhibited an activation energy more similar to that of Ni/MAO, and absolute rates were lower than what we observed with Ni/BaZrO3/MAO. More significantly, rates on the oxidized and reduced Ni/CaTiO3/MAO differed by only an order of magnitude, compared to the almost 4 orders of magnitude difference found with Ni/BaZrO3/MAO.
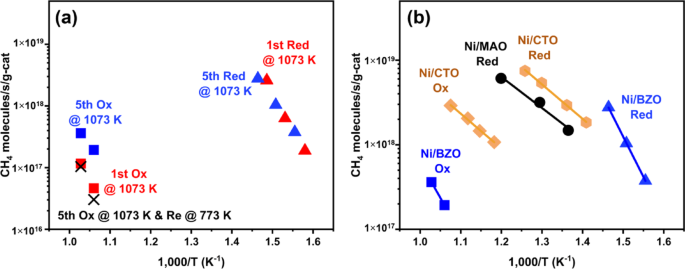
Steady-state, differential reaction rates for MSR with 18 Torr of CH4 and 18 Torr of H2O on a 1Ni-BaZrO3/MgAl2O4 reduced (triangles) and oxidized (squares) at 1073 K in the first redox cycle (red) and the fifth cycle (blue). The rates on the sample after 5th oxidation at 1073 K and then reduced at 773 K are also shown in the plot (black X), and b 1Ni/BaZrO3/MgAl2O4 reduced (blue triangles) and oxidized (blue squares) and 1Ni/CaTiO3/MgAl2O4 reduced (yellow hexagonals) and oxidized (yellow diamonds) at 1073 K, along with 1Ni/MgAl2O4 sample reduced at 773 K (black circles) as reference (for the 1Ni-BaZrO3/MgAl2O4, sample the rates on a per Ni site basis are presented in Fig. S1)
In earlier work with Ni/CaTiO3/MAO, it was also argued that oxidation treatments caused Ni to be inserted into the perovskite framework, while reduction brought the Ni particles back to the surface [6]. Higher reduction temperatures were required for Ni/CaTiO3/MAO compared to Ni/MAO because reduction of Ni2+ cations in the perovskite lattice is more difficult than reduction of NiO. Absolute MSR rates and activation energies on the reduced Ni/CaTiO3/MAO and Ni/MAO were similar. An interesting question here is why the Ni/BaZrO3/MAO catalyst appears to be so different. We suggest that the more dramatic deactivation observed upon oxidation of the Ni/BaZrO3 could be due to a more complete insertion of Ni cations into the perovskite lattice; however, the significant change in the activation energy for MSR on the reduced Ni/BaZrO3/MAO is more difficult to explain.
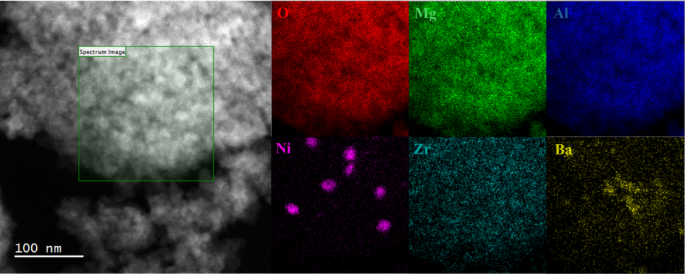
STEM image with EDS mappings of 1wt% Ni/BaZrO3/MAO after 5 redox cycling at 1073 K. The green box indicates the EDS region
One possible explanation for these observations could be a difference in Ni particle size. In the earlier study of the reduced Ni/CaTiO3/MAO, STEM images showed that the Ni particle-size distribution was uniform, with an average size of ~ 20 nm. The STEM/EDS results for Ni/BaZrO3/MAO after reduction at 1073 K, shown in Fig. 5, also show Ni particles ~ 20 nm in diameter. Since the Ni particle sizes are similar in the two samples, the differences in rates and activation energies cannot be related to this.
We instead propose that the enhanced rates on Ni/BaZrO3/MAO could be related to hydrogen transfer from the BaZrO3 to the Ni. As noted in the Introduction, BaZrO3 exhibits proton conductivity when doped with a + 3 ion, such as Y3+. Proton conduction is associated with mobile hydroxyls that can percolate the entire bulk structure. A bifunctional mechanism, involving reaction of protons from BaZrO3 with species on the Ni, could explain the higher rates Ni/BaZrO3/MAO catalyst. Since this is also a change in mechanism, a change in activation energy would not be surprising. Such a bifunctional mechanism has already been proposed for the water-gas-shift reaction over Ni supported on doped BaZrO3 [29]. The authors of that study also suggested that hydrogen from the perovskite phase could react with species on the Ni to promote the reaction.
Bifunctional mechanisms are well-known for metals on reducible supports, where oxygen from the support can react with adsorbates on the metal [30]. With ceria-supported metals, reactions at the metal-support interface can greatly increase rates for reactions, such as water-gas shift and steam reforming. That one might be able to promote reactions by proton transfer at the support interface is a relatively unexplored and potentially exciting concept. Clearly, more work is needed to determine where and how this might be used.
4 Conclusions
We have demonstrated that conformal films of BaZrO3 can be prepared on MgAl2O4 by ALD. When these are used as supports for Ni, the catalysts exhibit characteristics similar to that observed with Ni exsolution from bulk BaZrO3 in that high-temperature reduction is required to place them in an active state. Rates on Ni/BaZrO3 are enhanced relative to Ni on MgAl2O4, apparently due to proton transfer at the Ni-perovskite interface.
