Article Content
Introduction
Polycystic ovary syndrome (PCOS) is a multidimensional endocrine and metabolic disorder impacting 11–13% of women in reproductive years [1]. Hyperandrogenism is one of the critical features of PCOS and induces steroid secretion defects, obesity, hirsutism, acne, androgenetic alopecia, and thus great psychological stress [2].
In addition to hormonal imbalance and metabolic disorders, the role of sleep should be highlighted in PCOS. Women with PCOS are at elevated risk of sleep problems and related clinical comorbidities [3]. Bedtime, as a symbol of the congruence of circadian rhythm and sleep, is significantly related to a range of metabolic abnormalities [4]. Staying up late (SUL) is characterized as an occurrence in which the average time to fall asleep occurs after 23:00 throughout the 6 months before admission [5]. Previous studies showed that SUL was more prevalent in women with PCOS than in control groups [6]. Since the endocrine system activity is crucial in maintaining sleep-wake cycles, and metabolic disturbances are prominent in PCOS, it is conceivable that PCOS disrupts sleep or that there are more intricate interrelationships.
Though excess adipose tissue has primarily been emphasized, the quantity of lean mass (LM) is a critical factor in the regulation of insulin-dependent glucose uptake and may be linked to cardiometabolic dysregulation (e.g., insulin resistance [IR] and type 2 diabetes) in PCOS [7]. LM represents a clinically significant body composition parameter in PCOS. Current evidence has yielded contradictory results regarding the status of LM in PCOS versus controls [8,9,10]. Studies have elucidated that sleep-related factors are associated with variations in sex hormones and skeletal muscle mass in the general population [11,12,13]. However, to our knowledge, there are no epidemiological studies that have specifically examined the relationship between sleep and LM in women with PCOS.
In this cross-sectional study, we examined sleep behaviors assessed by the Pittsburg Sleep Quality Index (PSQI) and specific bedtime characteristics while investigating the compromised effects of various bedtimes on the level of androgenic hormones and variability of LM in women with PCOS to better delineate any need for targeted interventions that effectively prevent or postpone the onset of hyperandrogenism and muscle loss in this clinical population.
Methods
Participants and study design
This cross-sectional study was conducted on 899 women with PCOS who were admitted to the PCOS subspecialty clinic at Shanghai Tenth People’s Hospital. The diagnosis of PCOS was based on the 2003 Rotterdam criteria [14], and two senior consultant-level physicians reviewed all patients to ensure they met the Rotterdam criteria. We excluded participants who were < 18 or > 45 years old (n = 47); those without information about bedtime data (n = 102); those without dual-energy X-ray absorptiometry (DEXA) data (n = 75); and those without covariate data (n = 37). Finally, 636 participants with PCOS were included. The data presented includes participants from our observational study (NCT05063383). Upon approval of the study protocol by the Ethics Committee of Shanghai Tenth People’s Hospital (approval number: SHSY-IEC-4.1/21–227/01), all participants in this study signed the informed consent form.
Clinical and biochemical assessment
A well-trained researcher collected information on anthropometric measurements. Face-to-face interviews were conducted for each subject to gather socio-demographic and sleep information. The Ferriman-Gallwey score and menstrual condition were recorded. Participants underwent measurements of weight, height, waist circumference (WC), and hip circumference (HC) by using a calibrated scale after removing their shoes and heavy clothing. Body mass index (BMI) was calculated as the weight (kg) divided by height2 (m2). The waist-to-hip ratio (WHR) was calculated as WC divided by HC. Systolic blood pressure (SBP) and diastolic blood pressure (DBP) were measured after resting for at least 15 minutes. Liver stiffness measurement (LSM) and controlled attenuation parameter (CAP) were assessed by trained operators using transient elastography (FibroScan 502, Echosens, Paris, France). Following the manufacturer’s protocols, all participants underwent at least 10 valid measurements after a minimun of 3 hours of fasting.
Fasting blood samples were collected by trained nurses following standardized protocols, with subsequent biochemical analyses performed by certified laboratory technicians using automated analyzers under strict quality control measures. Venous blood was drawn in the morning following at least 10 hours of overnight fasting. Fasting plasma glucose (FPG), fasting insulin (FINS), glycosylated hemoglobin A1c (HbA1c), total cholesterol (TC), triglycerides (TG), low-density lipoprotein cholesterol (LDL-c), high-density lipoprotein cholesterol (HDL-c), creatinine (CR), serum uric acid (SUA), blood urea nitrogen (BUN), alanine aminotransferase (ALT), and aspartate aminotransferase (AST) were measured. Postprandial plasma glucose (PPG) levels were assessed by using a 75-g oral glucose tolerance test, with evaluations conducted at 2 hours post-ingestion. Sex and reproductive hormones, including luteinizing hormone (LH), follicle-stimulating hormone (FSH), total testosterone (TT), free testosterone (FT), androstenedione (AD), dehydroepiandrosterone sulfate (DHEA), sex hormone-binding globulin (SHBG), and anti-Müllerian hormone (AMH), were also measured. Homeostasis model assessment of insulin resistance (HOMA-IR) was computed using the following equation: [FPG (mmol/L) × FINS (mU/L)]/22.5 [15].
Renal (CR, BUN, SUA) and hepatic (ALT, AST, CAP, LSM) parameters were included to evaluate metabolic comorbidities in PCOS, such as renal dysfunction, hyperuricemia, liver injury, and liver fibrosis progression, all of which may be relevant to bedtime or muscle metabolism.
Subjective sleep information
Sleep behaviors were determined using the PSQI. The PSQI is a self-reported questionnaire that assesses seven components of sleep, including subjective sleep quality, sleep latency, sleep duration, habitual sleep efficiency, sleep disturbances, use of sleeping medication, and daytime dysfunction. A global PSQI score greater than 5 indicates poor sleep quality [16, 17]. Bedtime refers specifically to the self-reported time of falling asleep each night and it was calculated by using the questions “During the past month, what time have you usually gone to bed at night?” and “During the past month, how long (in minutes) has it usually take you to fall asleep each night?”, which were adapted from the PSQI [16], and has been validated in previous studies [18,19,20,21]. As SUL is defined as a consistent bedtime after 23:00 for at least 6 months [5], and combined with the average time to fall asleep in our study, we divided bedtime into three levels: ≤23:00, > 23:00 to 24:00, > 24:00 and they are marked as early bedtime, intermediate bedtime, and late bedtime, respectively.
DEXA analysis
Body fat and lean-related composition were determined using DEXA (APEX 4.5.0.2, Hologic, USA). The fat measures used in this study were total body fat percentage, total fat mass (FM), trunk FM, and appendicular FM. The LM measurement used in this analysis encompassed the following: total LM, trunk LM, muscle mass index (LM/height2), and appendicular muscle mass index (appLM/height2) [22]. Appendicular LM was defined as the sum of the LM of the bilateral upper and lower limbs. The latter were indices based on the total LM or appendicular LM, not on the body weight, and were standardized by height that can be calculated with the software provided by the manufacturer.
Statistical analysis
The results were displayed for each normally distributed continuous variable as the mean with the standard deviation. Continuous variables in three-group comparisons were performed by using analysis of covariance, with age as the covariate, followed by post hoc pairwise comparisons using the Bonferroni method. The multiple linear regression analysis of the association between bedtime and reproductive hormones or regional body LM was performed by using three adjustment models: model 1 adjusted for age and BMI; model 2 additionally adjusted for HOMA-IR, ALT and TG; model 3 further adjusted for SUA and TT. We included these potential covariates with p < 0.20 in univariate analysis [23], as well as clinically plausible factors, but excluded similar variables to avoid multicollinearity. All statistical analysis was conducted with SPSS version 25.0 software (SPSS Inc., Chicago, IL, USA). A p-value of less than 0.05 was considered statistically significant.
Results
Anthropometric and metabolic characteristics of patients with PCOS stratified by the bedtime category
In the study, 636 subjects with PCOS were eligible for the inclusion criteria with available bedtime and DEXA data (Fig. 1). Approximately 24.4% (155/636) of women with PCOS had early bedtime (≤ 23:00), 36.8% (234/636) had intermediate bedtime (> 23:00 to 24:00), and 38.8% (247/636) had late bedtime (> 24:00). Anthropometric and metabolic parameters of patients with PCOS categorized by the bedtime were given in Table 1. Women in the late bedtime group had fewer annual menstrual cycles than those in the early bedtime group. There were no significant differences in WHR, Ferriman-Gallwey score, blood pressure, glucose and insulin metabolism, lipid profiles, and hepatic and renal characteristics after adjustment for age among the three groups.
Flow diagram. PCOS, polycystic ovary syndrome; DEXA, dual-energy X-ray absorptiometry
Sex and reproductive hormonal characteristics of patients with PCOS stratified by the bedtime category
The results of sex and reproductive hormonal characteristics among the three groups were shown in Fig. 2, using the violin plot. Compared with participants in the early bedtime category, those in the late bedtime category showed significantly higher levels of TT, while individuals with intermediate bedtime had greater values for AMH. Meanwhile, compared with women in the early bedtime and intermediate bedtime groups, women in the late bedtime group had higher levels of AD. Levels of LH, FSH, FT, DHEA, and SHBG were comparable after adjustment for age among the three groups.
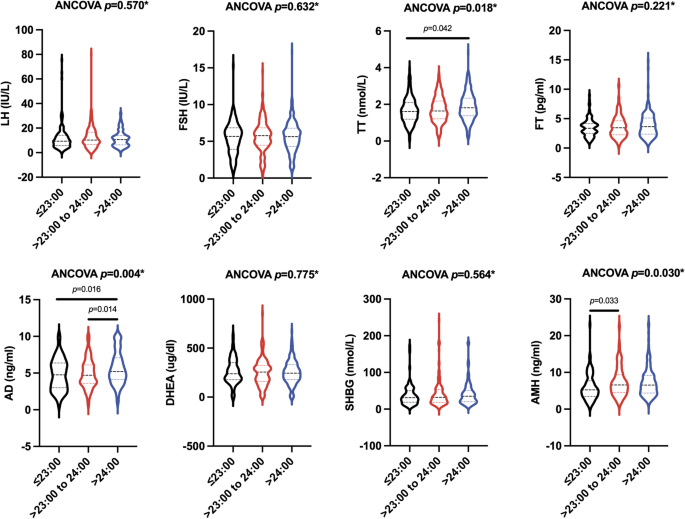
Sex and reproductive hormonal characteristics of the patients with PCOS stratified by the bedtime category. *p-values represented the results after adjustment for age. ANCOVA, analysis of covariance; LH, luteinizing hormone; FSH, follicle-stimulating hormone; TT, total testosterone; FT, free testosterone; AD, androstenedione; DHEA, dehydroepiandrosterone sulfate; SHBG, sex hormone-binding globulin; AMH, anti-Müllerian hormone
Body composition of patients with PCOS stratified by the bedtime category
As shown in Fig. 3, concerning body fat distribution, after adjusting for age, the percentage of total body fat, total FM, trunk FM, and appendicular FM were similar among the three groups. With respect to body lean distribution, after adjusting for age, no significant differences were observed in the levels of total LM, trunk LM, LM/height2, and appLM/height2 among all three bedtime groups.
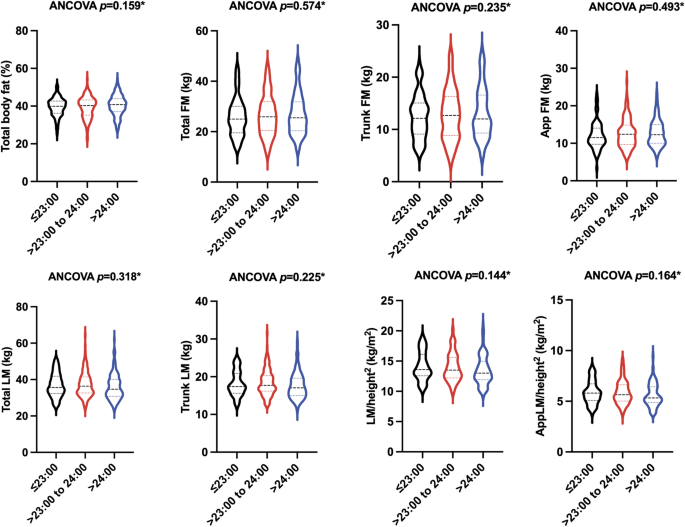
Body composition of the patients with PCOS stratified by the bedtime category. *p-values represented the results after adjustment for age. ANCOVA, analysis of covariance; FM, fat mass; App FM, appendicular fat mass; LM, lean mass; LM/height2, Lean mass index; AppLM/height2, appendicular lean mass index
PSQI components of patients with PCOS stratified by the bedtime category
Our analysis of sleep quality patterns in women with PCOS revealed clinically significant variations across the bedtime category. Using the PSQI global score > 5 cutoff, 68% of participants exhibited poor sleep quality (data not shown). The comparative analysis demonstrated that participants with late bedtime (> 24:00) showed consistently impaired sleep profiles relative to those with early bedtime (≤ 23:00) and intermediate bedtime (> 23:00 to 24:00), manifesting in: (1) markedly worse subjective sleep quality (1.45 ± 0.97 vs. 1.05 ± 0.79, 1.45 ± 0.97 vs. 1.10 ± 0.67, respectively; p < 0.001), (2) significantly prolonged sleep latency (1.37 ± 1.03 vs. 0.95 ± 0.89, 1.37 ± 1.03 vs. 1.09 ± 0.89, respectively; p < 0.001), (3) significantly short sleep duration (0.71 ± 0.72 vs. 0.42 ± 0.68, 0.71 ± 0.72 vs. 0.51 ± 0.62, respectively; p < 0.001), (4) more pronounced sleep disturbances (1.33 ± 1.45 vs. 1.08 ± 0.56, p = 0.038), and (5) more pronounced daytime dysfunction (1.91 ± 0.96 vs. 1.49 ± 0.99, 1.91 ± 0.96 vs. 1.65 ± 0.90, respectively; p < 0.001). Notably, the late bedtime (> 24:00) group displayed the highest mean PSQI score (7.18 ± 3.52), substantially exceeding the threshold for poor sleep quality. While habitual sleep efficiency (p = 0.880) and use of sleep medication (p = 0.245) showed no significant difference, all other PSQI components exhibited statistically significant variations among the groups (all p < 0.05), indicating that bedtime may systematically influence multiple dimensions of sleep health in the PCOS population (Table 2).
Anthropometric, metabolic, sex and reproductive hormonal characteristics, body composition, and sleep quality components of patients with PCOS stratified by BMI
According to the Asian BMI classification [24], underweight was < 18.5 kg/m2, normal weight was 18.5 to < 23 kg/m2, overweight was 23 to < 25 kg/m2, and obesity was ≥ 25 kg/m2. The data were presented as subgroups regarding the underweight group (BMI < 18.5 kg/m2), the normal weight group (18.5 ≤ BMI < 23 kg/m2), and the overweight/ obese group (BMI ≥ 23 kg/m2) in the Supplementary Table 1. After adjusting for age, participants in the overweight/obese group had lower levels of menstrual cycles, LH, SHBG, and higher levels of SBP, DBP, FPG, PPG, FINS, HbA1c, HOMA-IR, TG, LDL-c, SUA, ALT, CAP, FT than those in the underweight and normal weight group. The overweight/obese group showed significantly lower levels of HDL-c and AMH, and higher levels of AST, LSM, and TT than the normal weight group. No significant differences in the mean Ferriman-Gallwey score, TC, CR, BUN, AD, and DHEA were observed among the three groups. The characteristics of body composition were significantly different in different BMI subgroups. There were no significant differences in the score of PSQI components (except for use of sleeping medication) among the three groups.
Association between bedtime and reproductive hormones or regional LM in PCOS
To investigate the influence of bedtime on reproductive hormones or regional LM variability in PCOS, we performed the multiple linear regression analysis with adjustments for different potential confounding factors (Table 3). With adjustment for age and BMI (model 1), late bedtime was significantly associated with higher TT (β = 0.206, 95% confidence interval [CI]: 0.052 to 0.359, p = 0.009) and AD (β = 0.807, 95% CI: 0.242 to 1.372, p = 0.005) when compared with early bedtime, and both intermediate and late bedtime was significantly associated with higher AMH (β = 1.180, 95% CI: 0.133 to 2.227, p = 0.027; β = 1.043, 95% CI: 0.032 to 2.054, p = 0.043, respectively) when compared with early bedtime. Immediately after, in model 3 with adjustment for age, BMI, HOMA-IR, ALT, TG, and SUA, late bedtime was independently associated with higher TT (β = 0.197, 95% CI: 0.019 to 0.375, p = 0.030) and AD (β = 0.908, 95% CI: 0.263 to 1.552, p = 0.006), and intermediate bedtime was independently associated with higher AMH (β = 1.380, 95% CI: 0.243 to 2.518, p = 0.018) when compared with early bedtime. Concerning regional LM, in model 1, late bedtime was significantly correlated to reduced LM/height2 and appLM/height2 as compared with early bedtime (β=-0.634, 95% CI: -1.051 to -0.218, p = 0.003; β=-0.186, 95% CI: -0.358 to -0.014, p = 0.034, respectively). After adjustment for all potential covariates (age, BMI, HOMA-IR, ALT, TG, SUA, and TT) (model 3), these robust correlations were still independently existent (β=-0.535, 95% CI: -0.885 to -0.186, p = 0.003, for LM/height2; β=-0.214, 95% CI: -0.399 to -0.029, p = 0.024, for appLM/height2). However, the relationships between all bedtimes and total LM / trunk LM were not statistically significant in models 1–3.
Discussion
This study examined the association between bedtime, androgen metabolites, and body LM detected by DEXA in the PCOS population. Our data demonstrated that late bedtime (> 24:00) was significantly associated with elevated serum concentrations of TT and AD, as well as reduced levels of LM/height2 and appLM/height2 in women with PCOS. There have been studies reported that androgen status is independently associated with sleep disorder risks in PCOS [25] and sleep debt will favor the loss of muscle mass [26], but no literature to date has reported the link between sleep and body LM in women with PCOS, highlighting a significant gap in current research. To the best of our knowledge, this is the first study to clarify that late bedtime (> 24:00) has a significant adverse impact on LM in PCOS.
Sleep issues are an important global public health concern and have a substantial impact on cardiovascular diseases, type 2 diabetes, anxiety, and depression [27,28,29]. Women with PCOS have been found to experience circadian rhythm and sleep disturbances due to some pathophysiological and psychosocial implications [6, 30, 31]. We herein focus on bedtime, which could increase adverse long-term cardiometabolic health risks in PCOS [32], and for its minimal financial investment and technical proficiency while simultaneously providing opportunities for immediate clinical intervention. Compared to 94.25% reported in other literature [32], we found that 75.6% of the participants with PCOS had the habit of SUL. Simon et al. showed that poor sleep behaviors were related to metabolic disturbances and more metabolic syndrome symptoms in adolescents with PCOS and obesity [33]. This differs from the findings presented here that there were no differences observed in metabolic dysfunctions, including blood pressure, glucose and insulin metabolism, lipid profiles, and hepatic and renal characteristics among the early, intermediate, and late bedtime groups. A possible explanation for this might be age and racial differences.
Women with PCOS commonly suffer from menstrual disorders. Disrupted circadian rhythmicity may impair the secretion of reproductive hormones, thereby leading to menstrual irregularity [34]. Our findings were in accordance with recent studies [35, 36] indicating that SUL resulted in decreased annual menstrual cycles. It was suggested that biological clock disruption contributed to androgen excess in PCOS [37]. Specifically, our results showed that women with PCOS with late bedtime (> 24:00) had higher TT and AD concentration when compared with those with early bedtime (≤ 23:00). Additionally, late bedtime was independently associated with increased TT and AD concentration. Although the effects of bedtime on androgen levels in PCOS or other populations have not been reported, our findings were similar to those of a previous study by Nagata et al., who demonstrated that undesirable sleep-related factors were associated with higher levels of TT in premenopausal women [13]. Simon et al. theorized that adolescent girls with PCOS exhibited modified melatonin secretion and circadian dysregulation, which were positively linked with serum testosterone concentration [38]. Furthermore, our data observed that the intermediate bedtime exhibited significantly higher AMH than the early bedtime, and the intermediate bedtime was independently related to higher AMH. This finding was contrary to a previous study, which indicated that poor sleep quality, as evaluated by the PSQI, was associated with decreased levels of AMH in general reproductive women [18]. The exact reason is unclear, but it may be related to the fact that all participants had PCOS status in our study, and further studies with more focus on the relationship between bedtime and AMH in PCOS are therefore needed. In contrast to earlier findings [18, 25, 39], no differences in LH, FSH, FT, DHEA, and SHBG were detected among all bedtime groups. Taken together, our findings were generally in line with earlier research indicating that sleep problems were increasingly recognized as risk factors for women’s reproductive health [40,41,42] and the underly mechanisms may be genetics, dysregulation of the hypothalamic-pituitary-adrenal axis, oxidative stress, systemic inflammation, and insulin resistance [43].
In our study, body fat-based analysis showed no statistically significant differences in total body fat percentage, total FM, trunk FM, and appendicular FM among all bedtime groups. This outcome was contrary to that of Thomas et al., who found that later sleep timing was related to a higher body fat percentage in people with overweight/obesity [44], and Xu et al., who found that short sleep duration was independently related to the increased regional body fat in general adults [45]. Further research involving larger patient samples and biological underpinnings is required to shed light on the relationship between sleep and body fat distribution in PCOS.
Increasing evidence has shown that subjective sleep quality assessments are independently associated with changes in muscle mass in older adults [11, 46, 47]. Given that LM relates directly to IR [48] and testosterone [49] in PCOS, it is essential to examine the potential relationship between bedtime and LM within this population. Existing literature reports an inseparable association between sleep and muscle [50, 51]. Although similar levels of total LM, trunk LM, LM/height2, and appLM/height2 after adjusting for age in participants stratified by bedtime category were observed, we further demonstrated that late bedtime (> 24:00) was associated with reduced levels of LM/height2 and appLM/height2 by conducting the multiple linear regression. Despite no epidemiologic data on the association between bedtime and LM in PCOS or other populations so far, several studies have examined relationships between other sleep metrics and muscle in the general population. Song et al. discovered that appLM/height2 decreased as the sleep quality deteriorated [52]. In a study involving the 20-59-year-old population, it was found that poor sleep quality was associated with an increased risk of pre-sarcopenia [53], a condition characterized by low skeletal muscle mass but normal muscle function [54]. Furthermore, a meta-analysis has shown that abnormal sleep duration is associated with an increased risk of sarcopenia, featured by skeletal muscle mass loss and decline in muscular function [54, 55]. Sleep problems may contribute to diminished LM through potential mechanisms based on prior studies. People with late sleep timing are often accompanied by changed circadian rhythm and thus disruptions in melatonin secretion, which may generate reactive oxygen species and damage proteins, lipids, and deoxyribonucleic acid (DNA) in the human skeletal muscle, potentially contributing to muscle strength loss [56,57,58]. Furthermore, sleep problems may disrupt key hormonal pathways (e.g., insulin-like growth factor 1, insulin, growth hormone, testosterone, and cortisol) that modulate muscle fiber repair, strength, and function by suppressing anabolism while promoting catabolism in skeletal muscle [59].
The main advantages of this study are that the present study is the first to describe the association between bedtime, androgenic hormones, and LM in women with PCOS and uses a gold standard DEXA for measuring lean muscle mass. However, our study has several limitations. First, the cross-sectional design and reliance on self-reported bedtime data preclude the ability to make causal inferences. Further longitudinal studies incorporating objective sleep measurements are needed to confirm these associations and elucidate underlying mechanisms. Second, the current study only collected information on DEXA-based LM and did not deal with muscular function, indicating the need for additional research on this topic. Third, self-reported bedtime is prone to bias and may therefore yield inaccurate results. Finally, the absence of a non-PCOS group constrains the interpretation of the data from the PCOS cohort.
Conclusion
In conclusion, our cross-sectional study suggested that late bedtime (> 24:00) was independently associated with increased androgenic hormones and decreased lean muscle mass in the population with PCOS. These noteworthy findings corroborated the relationship between bedtime, androgen, and LM in reproductive-aged women with PCOS to underscore the significance of early sleep timing as a precaution against worsening hyperandrogenism and muscle mass loss.
