Article Content
1 Introduction
Water is a source of life and an indispensable part of the ecosystem. Rapid growth of global industrialization has resulted in the production of pollutants which contaminate water. Organic pollutants (dyes and other industrial wastes) are widely used in many industries and are discharged into water bodies, and have been found in potable and non-potable waters, severely affecting the ecosystem, particularly, humans and aquatic wildlife [1,2,3,4,5,6,7,8]. Among the organic pollutants, methylene blue (C16H18N3SCl) is particularly dangerous since it is highly poisonous, carcinogenic, non-biodegradable, and causes serious health problems for people. Several water treatment approaches, such as electrochemical oxidation, coagulation adsorption, membrane filtration, chemical precipitation etc., have been developed to remove organic pollutants and other industrial wastes from wastewater [9,10,11,12,13]. However, these water treatment approaches result in a low pollutant removal efficiency, a high selectivity for a certain dye pollutants, and need a high operating cost [14].
Recently, photocatalysis has been widely considered to be a promising approach, given the direct use of inexhaustible sunlight with minimal/no secondary adverse effects on the environment and economy from its by-products. In the photocatalytic process, the catalyst converts absorb light followed by production of reactive oxygen species (ROS), which further decomposes the contaminants (e.g. organic pollutants) in water to harmless by-products such as water (H2O) and carbon dioxide (CO2) [1, 3, 15, 16]. Metal oxide semiconductors (e.g. TiO2, ZnO, CuO, ZrO2, Cu2O, V2O5, Cr2O3, NiO etc.,) mediated photocatalysis of contaminants or organic pollutants have attained excessive technological significance in environmental remediation due to their superior light absorption abilities and favorable charge transport properties [17,18,19,20]. However, metal oxide semiconductors are mainly characterized by a large band gap energy (Eg) and thus have intrinsically low absorption efficiency, as their light absorption is only a small portion in the Ultraviolet region which is less than 5% [21,22,23].
The Noble metals such as silver (Ag), gold (Au) or platinum (Pt) strongly absorb visible light due to their localized surface plasmon resonance (LSPR) and promotes light-matter interactions, which contribute to the extended absorption range of visible light [22,23,24,25,26,27]. Surface plasmon resonance (SPR) arises from oscillation of free electrons in plasmonic structure and is characterized by strong absorption and/or scattering of light, as well as generation of intense electric fields. When plasmonic metals are incorporated with metal oxide semiconductors, the wide bandgap of the metal oxide catalyst shifts to lower bandgap, as a result bandgap reduction greatly facilitates the photo-generated carrier’s (electron-hole pair) separation, ultimately increasing degradation rate of organic pollutants under visible light irradiation. Moreover, the unique noble metal-semiconductor interfaces facilitate the formation of Schottky barriers and thus boosts the separation of photoexcited pairs for higher photodegradation rate [24, 25]. Recently, silver(Ag) metal integrated plasmonic photocatalysts such as Ag@TiO2 [28], Ag@Cu2O [29], Ag@AgCl [30], and Ag@ZnO [31] were reported. In each photocatalyst, the absorption edge has shifted to the visible region and performance has been enhanced by Ag NPs. Copper oxide (CuO) is an important p-type semiconductor with a narrow band gap (1.2–2.4 eV) and has been envisioned as a promising candidate in water remediation due to its fascinating properties of high photosensitivity, versatility, non-toxicity, and physical and chemical stabilities [32,33,34]. However, the limited absorption wavelength range and fast recombination rate of the photogenerated electron-holes (e−-h+) pairs in CuO result in low photocatalytic efficiency. A feasible method is incorporation of noble metals like Ag NPs with CuO matrix, to shift the absorption from the UV to visible range and suppress the recombination of charge carriers, resulting in improved photocatalytic efficiency [1, 35,36,37].
In the context of Ag doped CuO synthesis, green synthesis or biogenic approaches have emerged as a sustainable, reliable, and eco-friendly approach compared with conventional physical, and chemical approaches. In biogenic synthesis, water is mostly used as the solvent along with eco-friendly materials or extracts as reducing and stabilizing agents. The phytochemicals extracted from plants are active compounds that function as reducing, capping and stabilizing agents for synthesis of nanomaterial [38,39,40,41]. Cactus pears (Opuntia ficus indica) belong to the family of dicotyledonous angiosperm Cactaceae and contains various phytochemicals, including flavonoids, phenolic compounds, amino acids, proteins, ascorbic acid, ferulic acid and contents. These compounds, while chemically complex, are eco-friendly, and can act as reducing, capping and stabilizing agents in nanoparticle synthesis [40,41,42,43]. In this work, we reported for the first time green-synthesized heterogeneous Ag-doped CuO nanocomposites using cactus pears (Opuntia ficus indica) extract as a reducing, formation and stabilizing agent for photocatalytic degradation of methylene blue (MB) in water. A reduction and capping agent using plant extracts is aligned with the principles of green chemistry and offers many benefits as well as paving the way for industrial applications.
2 Experimental
2.1 Materials and Chemicals
The following reagents were employed; Cupric nitrate trihydrate (Cu(NO3)23H2O, 99.99%, Sigma-Aldrich Co., Ltd), Sliver nitrate (AgNO3, 99.99%, Sigma-Aldrich Co., Ltd). Methylene blue (MB, C16H18ClN3S, Sigma-Aldrich Co., Ltd) was additionally acquired for this work. The Cactus pears were obtained from Tigray, Ethiopia. All reagents/chemicals were used as received without further purification.
2.2 Plant Preparation and Extraction
According to our previous study [42], the fresh Cactus pears were collected and washed several times using deionized water to remove dust and contaminants before being cut into small pieces and dried in an oven at 60 oC for 12 h. The dried sample was ground with an electric blender. Typically, 5 g of cactus pear was mixed with 200 mL of distilled water in a 250 ml beaker and heated at 80 oC for an hour with continual magnetic stirring. The solution was cooled and filtered using Whatman No.1 filtered paper; The pH of the extract was 6.8.
2.3 Synthesis of CuO NPs and Ag@CuO Nanocomposites
In a typical procedure, 100 mL of 5 mM Cu(NO3)23H2O solution was mixed with 50 mL of extracts and was allowed the reaction mixture to heat at 100 oC for 7 h with constant magnetic stirring until a brownish-black precipitate was obtained. The obtained product was centrifuged for 15 min at 4000 rpm and washed several times with deionized water and ethanol. The sample was dried at 100 oC for 1 h in an oven and then finally annealed at 420 oC in a furnace to give CuO NPs.
In synthesis of Ag@CuO NCs, 5 mL of cactus pears extracts was mixed with 20 ml of 1 mM silver nitrate (AgNO3) solution (pH = 6.5) and stirred for 6–8 min until a dark brown was observed, which indicated the formation Ag NPs or reduction of Ag+ to Ag metal in the reaction mixture [44]. Subsequently, CuO NPs (0.5 gm) in 2 mL dispersion solution were slowly added, the pH of the mixed solution was 7.8 and then the mixture kept at room temperature with continual magnetic stirring for 2 h. The suspension solution was centrifuged for 15 min at 4000 rpm and then washed repeatedly using deionized water and ethanol to remove impurities. Finally, the obtained sample was dried at 60 oC for 2 h in a vacuum oven to give Ag@CuO nanocomposites. Figure 1 illustrates the green synthesis of Ag@CuO NCs using cactus pears extracts.
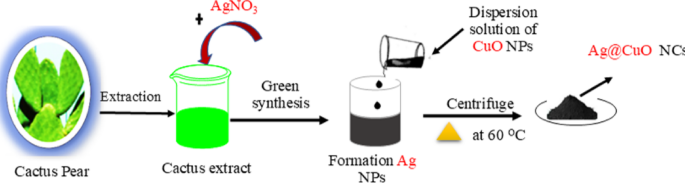
Schematic representation of biosynthesis of Ag@CuO nanocomposites
2.4 High-Pressure Liquid Chromatography
The reversed-Phase high performance liquid chromatography (RP-HPLC) was performed for screening contents presence in cactus pears extracts. The RP-HPLC system consists of a syringe-loading injector valve and a UV–Vis spectrophotometric detector (model K-2600; 275 nm). The analysis was performed using an ODS column (Shim-pack VP-ODS). The RP-HPLC solvent system was set using H2O/MeOH/MeCN with formic acid in ratios of 80:10:10 and 0:5:95 for separation. Detection was carried out at UV wavelengths of 230 nm/340 nm with a 50 µL injection volume. All measurements were performed at room temperature with a flow rate of 1 mL min−1.
2.5 Characterizations
The morphology features, crystal structure, and elemental compositions of Ag@CuO nanocomposites were analyzed using scanning electron microscopy (SEM, Leo-Stero Scan 440), X-ray diffraction (XRD, Bruker with 1.5406 A˙ radiation), and Energy dispersive X-ray spectroscopy (EDX), respectively. Rutherford backscattering spectrometry (RBS) characterizations were performed using 2 MeV He+ ion with a detector at backscattering angle of 165o to analyze elemental contents of the obtained samples. The specific surface areas and the pore size nature of the samples were obtained from Nitrogen (N2) adsorption/desorption isotherms using micromeritics ASAP 2020 instrument at 77 K. The photodegradation process of the methylene blue (MB) was detected using Ultraviolet-visible spectrophotometer (Shimadzu UV-2550) spectra with a scan range of 400–800 nm. The Perkin Elmer Lambda 950 UV–Vis NIR was used to record solid state ultraviolet-visible (UV–Vis) spectra of the obtained samples.
2.6 Photocatalytic Activities
The photocatalytic activity of Ag@CuO NCs photocatalyst was evaluated by photo-degradation of methylene blue (MB) upon visible light irradiation. A 300 W Xe lamp with a 420 nm cutoff filter was used as a light source. The experiment was carried out at room temperature as follows; 50 mg of photocatalyst was dispersed in 100 mL 20 µM of MB solution. Prior to irradiation, the suspension was stirred in the dark for 30 min to establish absorption/desorption equilibrium between the Ag@CuO NCs photocatalyst and MB solution and then allowed to react under visible light irradiation with continual magnetic stirring. At each sampling time (5 min), the irradiation was switched off and about 4 mL suspensions were sampled and centrifuged to remove the photocatalyst particles. The concentration of MB solution was monitored using UV-vis spectroscopy (Shimadzu UV-2550) by recording the variations of the absorption band maximum (663 nm).
The photo-degradation efficiency (DE %) of the resultant samples were determined by Eq. (1) below [45,46,47].
Where Co is initial concentration of before irradiation and Ct is the concentration at any sampling time.
3 Result and Discussion
3.1 Characterization of the Ag@CuO Nanocomposites
3.1.1 HPLC Analysis and Possible Reaction Mechanism
HPLC was applied to determine the content of the cactus pears extracts according to retention times (RT) and UV spectra. Figure 2 depicts the HPLC chromatogram of the cactus pears extracts, and seven dominant peaks are observed at RT of 3.73, 3.57, 3.97, 7.82, 9.00, 10.78, and 11.80 min in the extract. The first three peaks at RT of 3.73, 3.57, and 3.97 min indicated the presence of polar secondary metabolites, while the last peaks at 10.78, and 11.80 min likely corresponded to less polar compounds in the extract. The cactus pears extract possesses various biomolecules such as flavonoid and phenolic compounds, enzymes, amino acids, proteins, sugars and other traces of metals. However, flavonoid (quercetin and catechin), phenolic (gallic acid), ascorbic acid, and ferulic acid are the major polar phytochemicals present in cactus pear extracts [48,49,50,51]. These secondary metabolites are strongly involved in the bio-reduction process and play crucial roles in the formation and stabilization of nanoparticles [52]. In this possible reaction mechanism, we postulated a biosynthesis of silver (Ag) NPs using flavonoid (i.e. quercetin) as displayed in scheme-1. Silver nitrate (AgNO3) salt is decomposed into Ag+ and NO−3 ions in aqueous solution. Upon the release of protons from quercetin compound, Ag+ ions can form a complex structure with quercetin compounds resulting in the preparation of the Ago NPs [53, 54].
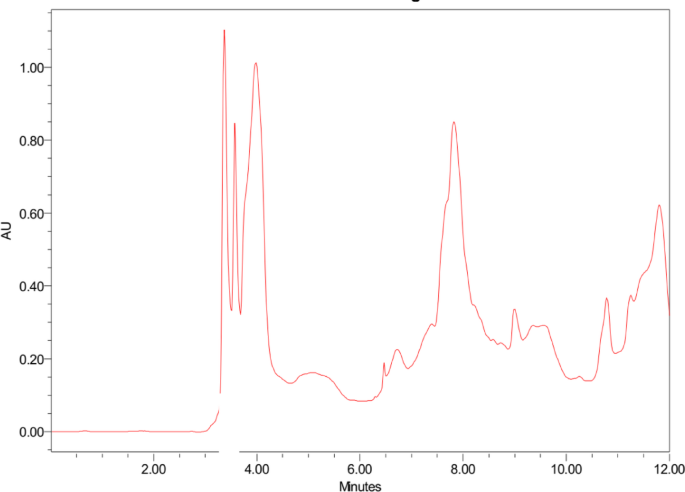
HPLC chromatogram of Cactus pears extract analyzed at 230 nm/340 nm
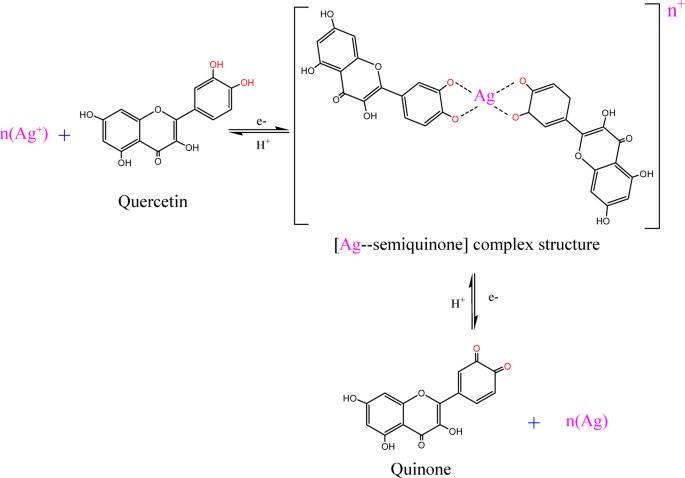
The bio-reduction of silver (Ag+) ions with quercetin for preparation of Ag NPs
3.1.2 Surface Morphology
The morphology features of CuO NPs and Ag@CuO nanocomposites (NCs) were characterized using scanning electron microscopy (SEM). Figure 3a shows the SEM image of CuO NPs and exhibits a nanorod-like surface with uniform particles distribution. Figure 3b reveals the SEM image of Ag@CuO NCs and confirms that bright white Ag NPs with a spherical shape and well dispersed on CuO surface. The size of Ag NPs on CuO surface ranged from 8 to 10 nm. The existence of these Ag NPs changed the boundary of CuO, and thus irregular nanorod-like structures are observed, compared with CuO, as shown in Fig. 3b, this may be assigned to high agglomeration, which came from lack crystallinity of Ag@CuO NCs. Due to the difficulties in nucleation, Ag clusters tend to agglomerate for further growth [55].

SEM images of a CuO NPs and b Ag@CuO nanocomposites
3.1.3 Structural, and Compositional Analysis
The crystallinity and phase purity of CuO NPs and Ag@CuO NCs were confirmed by X-ray diffraction (XRD) analysis. As revealed in Fig. 4a, the crystallographic orientations originated at a diffraction angle of 32.45o, 35.51o, 38.62o, 48.73o, 53.41o, 58.30o, 61.52o, 66.33o and 68.16o corresponding to (110), (002), (200), (20 − 2), (020), (202), (11 − 3), (31 − 1) and (220), crystal planes, respectively, which confirm the formation of a monoclinic phase of CuO crystals, and it is well-matched with standard JCPDS number 48–1548 [56]. Moreover, the XRD patterns at 2θ values of 38.14o, 44.27o and 64.2o corresponding to (111), (200) and (220) crystal planes, respectively, which are assigned to the face-centered cubic (fcc) phase of Ag metal (JCPDS number 89–3722) [56]. These results confirm the successful embedment of Ag NPs to form Ag@CuO NCs. When Ag NPs are incorporated into CuO, the peak intensity of CuO decreases, this may be due to the reduction of crystallinity of the obtained Ag@CuO NCs as well as the scattering factor difference of Cu and Ag atoms [57]. However, there are no changes in the position of peaks; thus, the integrated Ag NPs do not affect the monoclinic phase structure of CuO.
Moreover, energy dispersive X-ray spectroscopy (EDX) was employed to characterize the elemental compositions of Ag@CuO NCs. The EDX spectrum clearly highlighted the appearance of highly intense peaks of Cu and O atoms. In addition to Cu, and O, peaks from Ag metal are observed, as shown in Fig. 4b, which confirms that the Ag metal is well embedded into CuO matrix to form Ag@CuO NCs.
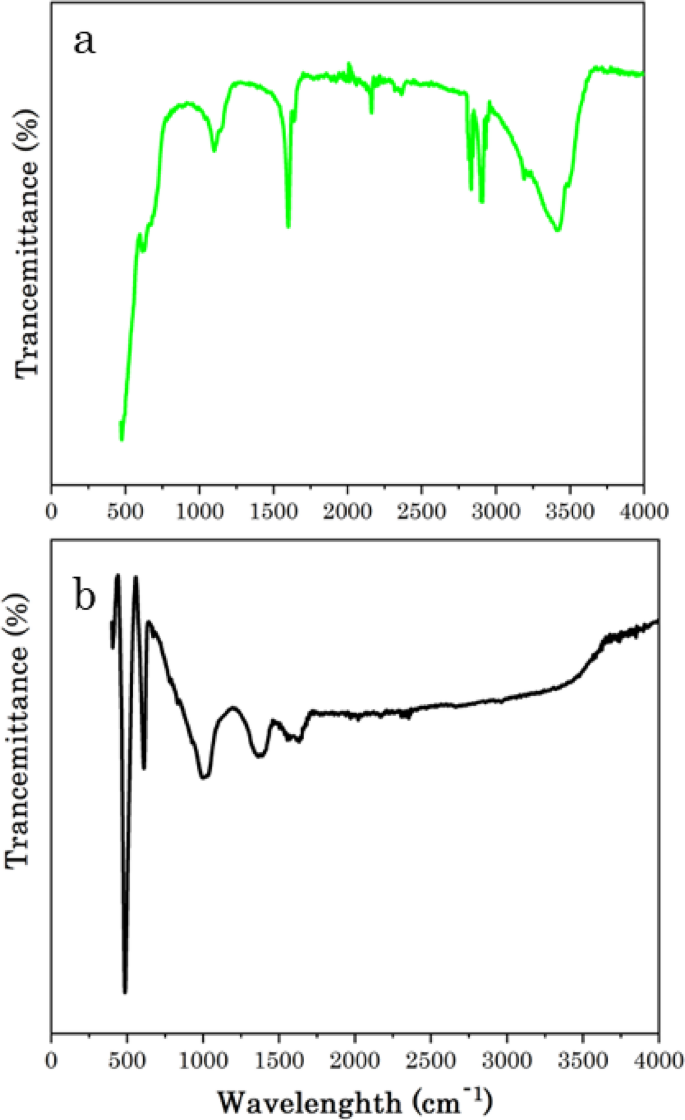
FTIR spectrum of a Cactus pears extract and b Ag@CuO NCs
3.1.4 FTIR Analysis
Fourier transform infrared spectroscopy (FTIR) was employed to investigate the vibration bonds present in Cactus pears (Opuntia ficus indica) plant material and Ag@CuO NCs, as shown in Fig. 5a, b, respectively. Figure 5a depicts the FTIR spectra of Cactus pears, revealing strong absorption peaks attributed to functional groups within the plant material. These peaks confirm that secondary metabolites are potential sources for the synthesis of the catalyst. The peak around 3445.4 cm−1 is corresponding to the stretching vibration of hydroxyl (–OH) functional groups from phenols, which is similar to the band obtained using Mimosa pudica extracts [58]. The bands 2917.5 and 2849.7 cm−1 are accredited to the C–H asymmetric and symmetric stretching, respectively, and the peaks at 2365.7 and 2146.8 cm−1 ascribed to–C≡N and–C≡C– stretching vibration, respectively. A strong absorption peak related to the carbonyl (C=O) functional group appeared at 1610.3 cm−1. The peak at 1100.6 cm−1 shows C-H bending [58, 59]. Figure 5b shows the FTIR spectra of the green synthesized Ag@CuO NCs. The bands at 1619.2, and 1367.8 cm−1 could be related to the presence C=O, and C=C stretching vibrations, respectively, and the band at 1011.4 cm−1 is corresponding to C–H bending vibration from the plant material on the Ag@CuO NCs catalyst. The slight shift in the band positions of Ag@CuO compared with the plant extracts confirms that the biosynthesized Ag NPs are well-impeded on the CuO matrix [53]. Peaks under 1000 cm−1 confirm the presence of metal–oxygen (M–O) interaction. The sharp and intense peaks at and 613.7 and 502.6 cm−1 revealing the characteristic stretching vibrations of Cu–O bonds in CuO NPs [39, 60, 61].
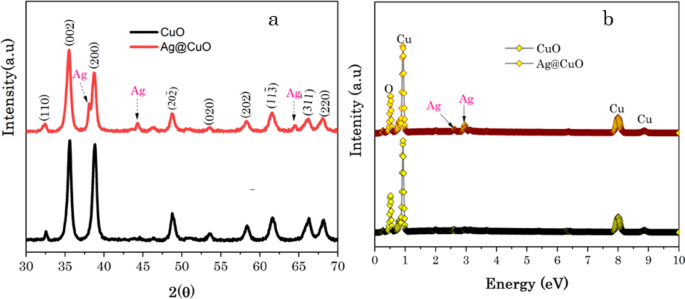
a XRD patterns and b EDX of CuO NPs and Ag@CuO NCs
3.1.5 Rutherford Backscattering Spectrometry
Further elemental compositions and contents of Ag@CuO NCs. were analysed using Rutherford backscattering spectrometry (RBS). RBS is the simplest ion beam analysis (IBA) method for elemental surface analysis and depth profiling, as gives information on the thickness, content, and absolute concentration of elements [25, 62]. Figure 6a, b shows the RBS spectra of CuO NPs and Ag@CuO NCs. RBS simulations using SIMNRA 7.02 software was used to fitting the spectra and to determine the elemental compositions of the biogenic Ag@CuO NCs. In Fig. 6a two peaks of alpha particles backscattered from Cu and O atoms at channels 2424, and 1112 are observed with elemental contents of 54.6, and 45.4 at %, respectively. In Ag@CuO NCs, the plasmonic Ag atom appeared at channel 2605, in addition to peaks of Cu, and O atoms, as shown Fig. 6b and the plasmonic Ag content in Ag@CuO NCs is found to be 4.6 at %.
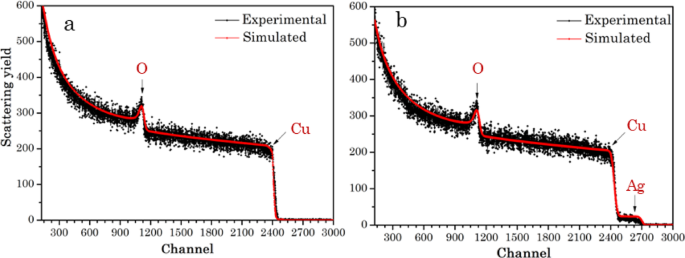
RBS spectra of a CuO NPs, and b Ag@CuO NCs
3.1.6 N2 Adsorption-Desorption Isotherms
The surface area of a photocatalyst is significantly influences the performance and stability of photocatalytic systems [63]. N2 adsorption and desorption isotherm measurements were employed to investigate the role of surface area and porosity nature of the biogenic CuO NPs and Ag@CuO NCs, as demonstrated in Fig. 7. The surface area and pore size of CuO NPs and Ag@CuO nanocomposites were achieved using Brunauer–Emmett–Teller (BET) method from N2 adsorption and desorption isotherms. According to the classification of the IUPAC, N2 sorption plots (P/P0 0 to 1.0) of CuO and Ag@CuO have distinct type-IV hysteresis loops with mesoporous nature. The CuO NPs possess a lower BET specific surface area of 8.67 m2 g−1, while that of Ag@CuO NCs (15.14 m2 g−1) is much larger with the pore volumes are 3.46 × 10−3 cm3 g−1, and 2.607 × 10−2 cm3 g−1, respectively. As can be seen from the pore size distribution (inset in Fig. 7), CuO and Ag@CuO have a mesoporous nature with a range centered at 26.5 and 21.4 nm, respectively. The higher surface area affords more active catalytic sites for photochemical reactions, which contribute to the transportation and diffusion of organic or dye pollutants and higher photodegradation efficiency [64].
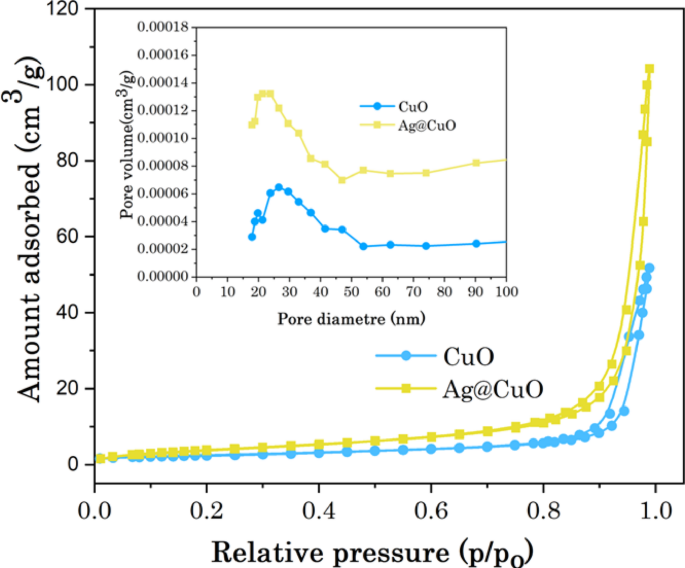
N2 adsorption/desorption isotherms of CuO and Ag@CuO and the inset are the corresponding pore size distributions
3.1.7 UV-Visible Spectroscopy and Tauc Plot
The optical properties of CuO NPs before and after embedding of Ag NPs were studied using UV–Vis-NIR spectroscopy (Perkin Elmer Lambda 950). The absorbance spectrum and bandgap energy (Eg) of the biogenic CuO NPs and Ag@CuO NCs are demonstrated in Fig. 8a, b. As can be seen in Fig. 8a, these two samples showed a wide absorption range for visible light. Moreover, the surface plasmon resonance (SPR) effect of Ag NPs intensified the absorption of Ag@CuO NCs in the visible light region. SPR enhanced the local electromagnetic field when Ag NPs are subjected to incident light, and this causes to increase the density of electrons at the CuO’s surface and generates more electron trapping sites which are crucial to encourage redox reactions in photocatalytic process. The synergistic interaction between the catalysis and SPR-induced electron effects exemplifies the intricate and diverse nature of the observed processes [56,57,58,59,60,61,62,63,64,65,66].
The corresponding bandgap energy (Eg) can be determined from the UV − Vis absorption spectrum using the Tauc plot according to the direct semiconductor feature by Eq. (2) [35].
Where α is absorption coefficient, h is Planck’s constant, v is the photon’s frequency, Eg is the band gap energy, A is absorption constant, and n is the nature of the electronic transition (direct transitions n = 1/2 or indirect transition n = 2, here ‘n’ is indirect transition). The band gap energy (Eg) is determined by the intercept of the straight line at linear part of (αhν)2 vs. hν curve and then the intercept of the tangent on the photon energy (hv) axis gives the bandgap (Eg) values, as shown in Fig. 8b. The estimated band-gap energy (Eg) values for CuO and Ag@CuO are found to be 2.1 and 1.95 eV, respectively. The narrow band gap energy (Eg) of Ag@CuO indicates that more light can be absorbed and utilized easily [67].
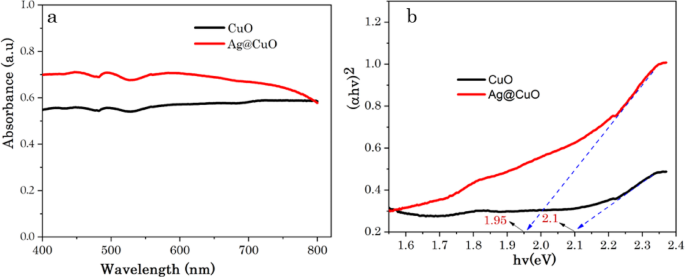
a UV−Visible spectrum and b the corresponding band gap energies of CuO and Ag@CuO NCs
3.2 Photocatalysis Performances
Photocatalysis is a green and eco-friendly approach to degrade pollutants released from industries into water bodies such as methylene blue (MB). The photodegradation performance of the green synthesized CuO NPs and Ag@CuO NCs for MB dye was evaluated under visible light irradiation, as shown in Fig. 9a, b. Figure 9a shows UV-vis absorption spectra of the photocatalytic MB on CuO NPs and Ag@CuO NCs surfaces under light irradiation after 5 min adsorption, and depicts degradation rates of 27 and 44%, respectively, the Ag NPs impacts are similar with V. H. Rathi et al. [56]. These results strongly imply that the photodegradation of MB under visible light irradiation is significantly improved by the integration of plasmonic (Ag) on CuO NPs, because when CuO was decorated with Ag NPs, charge transfer occurred from the CuO to the Ag NPs, suppressing the recombination of charge carriers, and thus leading to remarkably improved photocatalytic activities [25, 56]. Figure 9b demonstrates the degradation of MB solution using Ag@CuO NCs at different interval time. Prior to irradiation, the adsorption-desorption equilibrium for 30 min was carried out with Ag@CuO NCs catalyst and only less than 12% of MB was adsorbed or decomposed in 30 min. In the presence of light irradiation degradation was observed, and it is clear that the intensity absorption peak for MB (λmax ~ 663 nm) diminished gradually as the exposure time increased, as shown in Fig. 9b.
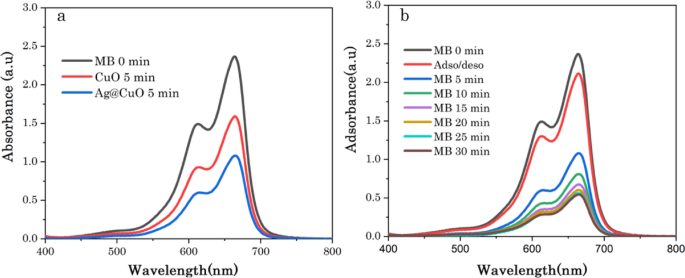
Photocatalytic of MB dye a using CuO and Ag@CuO at 5 min irradiation time and b Ag@CuO at different irradiation time
The photodegradation efficiency (DE%) of Ag@CuO NCs was evaluated using Eq. (1). Figure 10 shows the photodegradation efficiency of MB dye, CuO NPs and Ag@CuO NCs as function of irradiation time. In the blank test without CuO NPS or Ag@CuO NCs catalyst, the absorption of MB barely changed under visible light irradiation, indicates that MB is stable in visible light irradiation, as shown in Fig. 10. However, in the presence of Ag@CuO NCs catalyst, about 82.4% of MB is absorbed or degraded after 30 min light irradiation; in contrast, 67.2% of MB was degraded for pure CuO NPs, as demonstrated in Fig. 10. The enhanced degradation rate of Ag@CuO NCs is due to the embedded of surface plasmonic resonance (SPR) of Ag metal that created the electron shrinkage and decrease re-combination of the photogenerated pairs (e−-h+). Moreover, the integrated Ag NPs are responsible for enhancing the intrinsic absorption properties of CuO, because of the concentrated free plasmonic electrons whose particle sizes are comparable to or smaller than the incident light wavelength [56, 68].
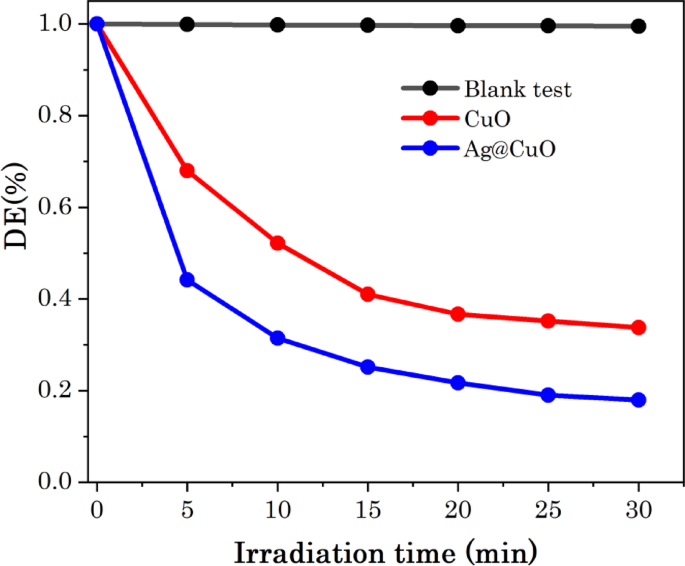
Photodegradation rate of MB as function of irradiation time
3.2.1 Photodegradation Kinetics Study
Kinetics are usually used to evaluate photocatalysis reactions, as a critical and significant photocatalytic parameter [69]. The kinetics of the photocatalytic degradation of MB dye using Ag@CuO NCs catalyst was investigated via a pseudo-first order model, as described in Eq. (3) [70,71,72].
where C0 and Ct are the equilibrium concentration and concentrations of MB at the exposure time ‘t’ respectively and k is the apparent rate constant. Figure 11 presents the kinetic behaviors of MB degradation and exhibits a linear relationship between ln(C/C0) and irradiation time catalyzed over CuO NPs and Ag@CuO NCs, indicating that the MB photodegradation process follows pseudo-first-order kinetics. The rate constant (k, min−1) for MB is calculated to be 0.0362 min−1 for CuO and 0.0578 min−1 for Ag@CuO NCs under light irradiation, with the regression coefficient R2 are 0.9886 and 0.9560, respectively. The pseudo-frst-order kinetics or the kinetic constant (k) value of Ag@CuO NCs (0.0578 min−1) is higher than that of CuO catalyst, indicating the excellent photocatalytic activity of Ag@CuO NCs for MB dye under light irradiation. The photocatalytic behavior of semiconductor catalysts is mainly dependent on the separation of photo-generated electron-hole (e−-h+) pairs. In the Ag@CuO NCs system, the photo-induced electrons transfer to the Ag co-catalyst because it has higher electron attracting power than CuO, which reduces the re-combination of photo-generated electron-hole (e−-h+) pairs and improves the photocatalytic reactions [37, 61]. Thus, Ag@CuO NCs have superior photocatalytic performance compared to CuO NPs under similar experimental conditions.
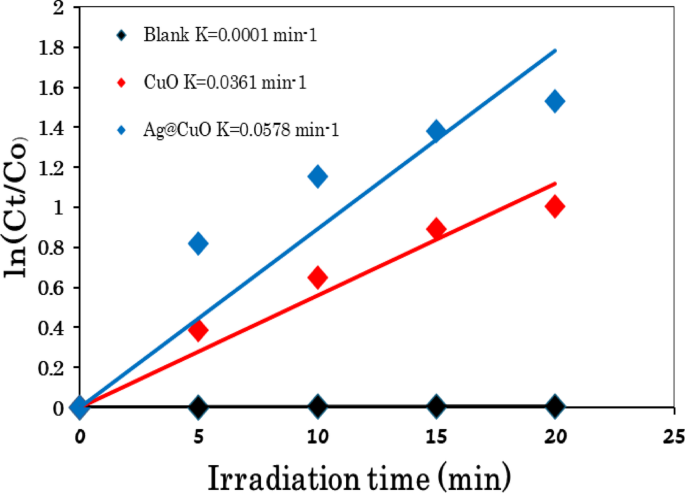
Kinetic linear fitting curves of MB with and without catalyst
In general, plasmonic-dopped metal oxide semiconductors have been found to significantly improve photocatalytic activities compared to other metals. Plasmonic metals, especially Ag embellishments, decrease the band gap energy (Eg) while improving the surface area and optical characteristics of the catalyst [56, 73, 74]. Table 1 shows superior photocatalytic activities of Ag dopped semiconductor catalysts compared to previous studies.
3.2.2 Mechanism of Photocatalysis
Development of visible-light-driven photocatalysts is considerably needed, as a green and eco-friendly approach for purification of organic pollutants [80]. It is well known that photocatalysis occurs on the surface of the catalyst via adsorption/desorption phenomena.
The photocatalytic reaction is initiated when a photoelectron is promoted to the empty conduction band (CB) from the filled valence band (VB) of a semiconductor photocatalyst as a result of irradiation. The energy difference between the conduction band (CB) and valence band (VB) of the semiconductor photocatalyst is acknowledged as the band gap energy (Eg) and governs light absorption of semiconductors [81,82,83]. Figure 12 illustrates the photocatalytic degradation mechanism of organic pollutants on semiconductor’s surface.
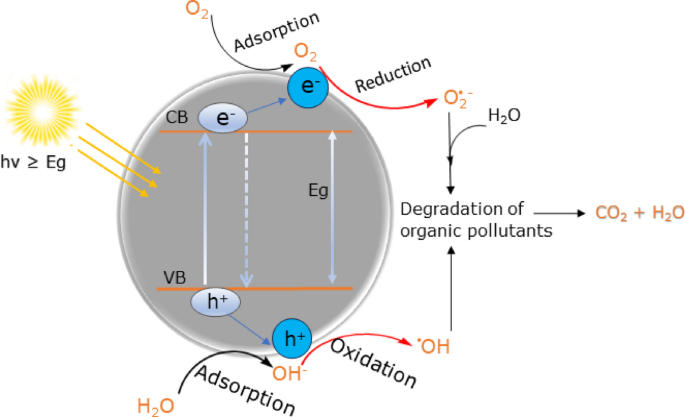
Schematic representation of degradation mechanism of organic pollutants on the semiconductors under visible light irradiation
Enhanced photocatalytic processes are dependent on (i) absorption of light and excitation of electrons, (ii) separation and migration of electrons-holes (e−-h+), (iii) recombination photo-excited e−/h+, and (iv) photo-induced charge carriers (e−/h+) undergo redox reactions [84, 85]. When a photocatalyst semiconductor (i.e CuO) is illuminated by a visible light, the photon energy (hv ≥ Eg) allows electrons from the valance band (VB) to become excited and migrate to the conduction band (CB), leaving electron vacancy hole (h+) in the VB and thus electron-hole (e−-h+) pairs are generated. This phase is said as ‘’photo-excitation’’ state of the photocatalyst [81,82,83,84,85,86]. The photocatalysis of organic pollutants and related chemical reactions on the surface of semiconductors upon light irradiation are postulated as follows with the help of Eqs. (4–8).

The negative electrons of the photocatalyst in the conduction band (eCB−) could be easily trapped by the dissolved oxygen molecules (O2) which results in the production of superoxide radicals (•O2−), concurrently, the positively photo-induced in the valance band (hVB+) reacts with adsorbed water (H2O) on the surface of CuO and splits the water (H2O) into hydrogen gas (H+) and highly energetic hydroxyl radicals (OH˙) as shown in Eqs. (5, 6). The establishment of these highly energetic reactive species (O2˙−, OH˙) helps to reduce the recombination rate of electron-hole (e−-h+) pairs and provide additional time to deal with MB dye [81,82,83,84,85,86]. The presence of surface plasmonic resonance (SPR) Ag NPs can cause the direct transfer of plasmonic energy into CuO, resulting in the acceleration and production of more electron-hole (e−-h+) pairs when the Ag NPs are exposed to light irradiation due to Ag metal has higher Fermi energy than CuO; as a result, suppressing the recombination of charge carriers(e−-h+) [56, 87]. Finally, the decomposition and mineralization of organic dye pollutants depends heavily on these generated active species (O2˙−, OH˙) and participate in the series redox reactions on the surface of the photocatalyst for mineralization MB dye as shown in Eqs. (7,8) [56, 81, 86, 88]. In conclusion, it is important to mention that the excellent photodegradation are attributed to high surface area, increased adsorption capacity, enhanced light absorption, and better charge separation in the photocatalyst [68, 82, 86, 89].
3.2.3 Stability and Recyclability
To reveal the photo-stability and reusability of the green synthesised Ag@CuO photocatalyst for the potential practical applications, the cyclic photocatalytic experiments MB dye was caried out. Figure 13 shows the recyclability or reusability of the mesoporous Ag@CuO NCs for MB dye in five runs of cycles. After each cyclic experiment, the photocatalyst was recovered by centrifugation and then washed with deionized water repeatedly, dried at 60 °C in oven and reused for photocatalytic of MB dye in the next cycle. The degradation rate of MB after three cycles is still above 90% as shown in Fig. 9. The photodegradation rates of MB are maintained at 96.7, 94.3 and 91.1% after the 1 st, 2nd and 3rd runs, respectively. The slight decrease in the effectiveness of the Ag@CuO catalyst may be due to adsorption of intermediate species onto active sites at the surface produced during the photodegradation of the dyes [90]. The small decline in the degradation potential confirming that high stability and recyclability of Ag@CuO catalyst in photocatalysis of MB dye.
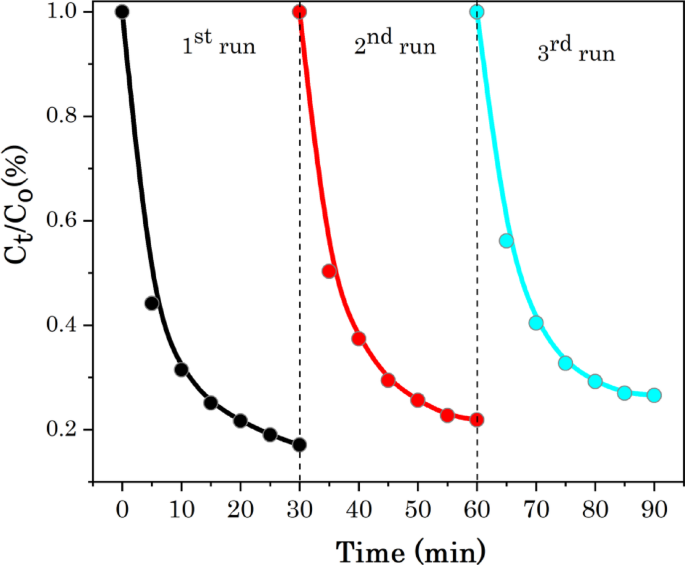
Stability studies of Ag@CuO NCs for degradation of MB dye under light irradiation
4 Conclusion
In the context of Ag doped CuO synthesis, the biogenic approach has emerged as sustainable and eco-friendly for photodegradation organic pollutants. Herein, a Low cost, non-toxic, and eco-friendly green synthesis of Ag@CuO nanocomposites using Cactus pears extracts for the decomposition of methylene blue (MB) is reported. The phytochemicals of aqueous Cactus pears extracts act as both reducing and capping agents to synthesise the nanoparticles. The morphological, structural, and elemental characteristics of the obtained samples were extensively analyzed. According to the XRD, the Ag@CuO nanocomposites displayed a monoclinic nature of CuO and face-cubic center (fcc) structure of Ag metal. The heterogeneous biogenic Ag@CuO nanocomposites (NCs) were evaluated for the photocatalytic of methylene blue (MB) upon light irradiation and the result revealed that about 82.4% of MB is decomposed after 30 min adsorption; in contrast, 67% for CuO NPs. The localized surface plasmon resonance (LSPR) of Ag NPs induces an activity of CuO under visible light due to the concentrated free electrons whose particle sizes are comparable to or smaller than the incident light wavelength. Furthermore, the recycling experiments confirmed good photostability and reusability of Ag@CuO catalyst in photocatalysis of MB. Overall, these findings suggest that the synthesized Ag metal-doped CuO using Cactus pears extracts can have photo-based applications in environmental remediation and other fields due to the absence of toxic chemicals.
