Article Content
Abstract
Aims
To evaluate the safety profile of imatinib using the FDA Adverse Event Reporting System (FAERS) database, identifying both documented and undocumented adverse events.
Methods
The FAERS data is widely used in drug safety surveillance studies, helping to identify potential safety issues. The FAERS data, spanning from the first quarter of 2014 to the fourth quarter of 2024, were subjected to a comprehensive analysis utilizing various disproportionality analysis methods. These methods included the reporting odds ratio (ROR), proportional reporting ratio (PRR), Bayesian confidence propagation neural network (BCPNN), and empirical Bayes geometric mean (EBGM). The primary objective of this analysis was to quantify the associations between specific drugs and their corresponding adverse events (AEs). By employing these advanced statistical techniques, we aimed to identify and evaluate potential safety signals within the vast dataset, thereby providing valuable insights into the pharmacovigilance landscape over the specified decade.
Results
A total of 56,364 reports (170,659 AE occurrences) were included. Imatinib exhibited expected AEs (e.g., nausea, diarrhea) consistent with summaries of product characteristics (SPCs), alongside potential novel signals such as pubertal failure, large intestine fibroma, ototoxicity, and pregnancy complications. Severe outcomes comprised 84.24% of reports (34.44% death), with 38.08% of AEs occurring > 360 days post-treatment. Malignant neoplasm progression showed a strong association.
Conclusions
This study has revealed the safety issues of imatinib, particularly in terms of gastrointestinal reactions. It emphasizes the need for careful monitoring and further research in clinical applications to understand the mechanism, improve treatment efficacy, and minimize adverse events.
Explore related subjects
Discover the latest articles and news from researchers in related subjects, suggested using machine learning.
- Checkpoint Signalling
- Chronic myeloid leukaemia
- Growth Factor Signalling
- Statistical Software
- Drug Safety and Pharmacovigilance
Introduction
In 2001, imatinib, the first kinase inhibitor, was approved by the U.S. Food and Drug Administration (FDA) [1]. Imatinib inhibits the Abelson (ABL) tyrosine kinase, which is expressed as a dysregulated fusion protein, BCR-ABL, in nearly all chronic myeloid leukemia (CML) cases [2]. This oncogenic fusion protein arises from a chromosomal rearrangement that juxtaposes the breakpoint cluster region protein (BCR) gene with the ABL gene, forming the Philadelphia chromosome.
The phase 3 International Randomized Study of Interferon and STI571 (IRIS) demonstrated superior efficacy and tolerability of imatinib (400 mg once daily) compared to interferon alfa plus cytarabine in newly diagnosed chronic-phase CML. In the treatment of chronic myeloid leukemia (CML) and certain gastrointestinal stromal tumors (GIST), imatinib, as the first-generation tyrosine kinase inhibitor (TKI), ushered in a new era of targeted therapy. Although the second- and third-generation TKIs (such as dasatinib, nilotinib, and ponatinib) have made significant progress in overcoming drug resistance and improving treatment efficacy, imatinib, due to its lower toxicity, good tolerance, and outstanding efficacy in first-line treatment, remains the preferred drug for many patients. At the 18-month follow-up, the imatinib arm achieved a significantly higher complete cytogenetic response rate (0% Philadelphia chromosome-positive metaphases: 76.2% (95% CI, 72.5–79.9) vs. 14.5% (95% CI, 10.5–18.5); P < 0.001) and a greater probability of freedom from disease progression to accelerated phase or blast crisis (96.7% vs. 91.5%; P < 0.001). These results established imatinib as a first-line therapy with enhanced therapeutic activity and reduced toxicity in early CML management [3].
Imatinib has fundamentally transformed the treatment of CML, resulting in significant improvements in patient prognosis [4]. In the USA, the age-adjusted annual mortality rate for CML patients decreased from 0.9 per 100,000 individuals in 1996 to 0.4 per 100,000 in 2006 [5]. Similar trends of reduced mortality have been observed in CML patients across other global regions [6, 7].
Although imatinib has undoubtedly provided substantial benefits for many cancer patients, these agents are not curative. Most merely delay tumor progression, as advanced malignancies develop escape pathways to evade targeted inhibition, leading to adverse effects and drug resistance. Thus, investigating and analyzing the real-world safety profile of imatinib remain critically important.
The FAERS, a prototypical public spontaneous reporting system, collates post-marketing safety reports and clinical study data related to FDA-approved drugs and therapeutic biologics, both within and outside the USA. It has been extensively employed for pharmacovigilance potential signal detection [8]. Our study leveraged comprehensive disproportionality analyses of imatinib-associated AEs extracted from FAERS data. By rigorously evaluating potential signal strengths within real-world evidence, this approach provides a robust framework for assessing the efficacy and safety of imatinib therapy, facilitating effective AE detection and management. Through this analysis, our research aims to elucidate imatinib’s safety profile and contribute evidence on its adverse events. This endeavor not only advances our understanding of imatinib-related risks but also underscores the necessity of continuous post-marketing drug safety surveillance. Ultimately, this pharmacovigilance study seeks to inform clinical practice, guide therapeutic decision-making, and enhance patient safety.
Methodology
Study design and data sources
This study analyzed AEs associated with imatinib using data from the FAERS. FAERS is selected for its unparalleled scale of real-world post-marketing safety data, enabling global surveillance of adverse events across drugs and medical products. As FDA’s primary pharmacovigilance database, it serves as the gold standard for detecting emerging safety signals and supporting regulatory decisions. The raw data spanned 84 quarters (the first quarter of 2014 to the four quarter of 2024), encompassing 18,613,992 background reports (55,357,463 AE occurrences), with 56,364 reports (170,659 AE occurrences) specifically linked to imatinib. To address inherent limitations of spontaneous reporting systems, such as duplicate or withdrawn reports, rigorous data cleaning was performed following FDA guidelines (https://www.fda.gov/drugsatfda). Duplicate reports were removed by sorting the DEMO table fields (PRIMARYID, CASEID, FDA_DT) and retaining the entry with the highest FDA_DT for identical CASEIDs; if CASEID and FDA_DT matched, the entry with the largest PRIMARYID was retained. Post-2019 quarterly deletion lists were further utilized to exclude invalid reports. Statistical methods were applied to identify adverse drug reactions (ADRs). AEs related to imatinib were categorized into preferred terms (PTs) and system organ classes (SOCs) based on the hierarchical structure of the Medical Dictionary for Regulatory Activities (MedDRA20.0). The data screening workflow is illustrated in Fig. 1.
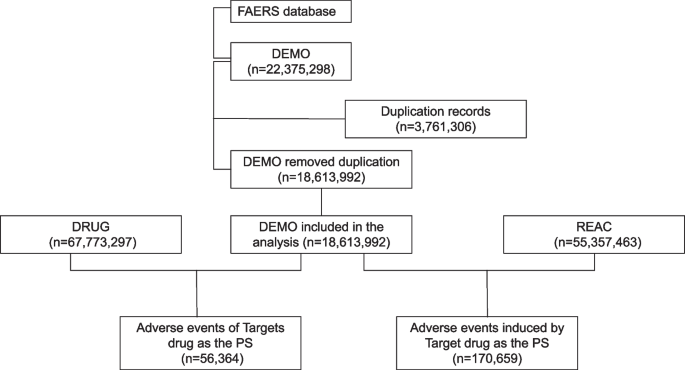
Multistep process of data extraction, processing, and analysis from the FAERS database
Statistical analysis
A disproportionality analysis was conducted to identify potential drug-AE associations in pharmacovigilance. Four algorithms were applied: the reporting odds ratio (ROR), proportional reporting ratio (PRR), Bayesian confidence propagation neural network (BCPNN) [9, 10], and multi-item gamma Poisson shrinker (MGPS). These methods compare the proportional reporting rates of AEs between imatinib and all other drugs [11]. Four algorithms were employed to quantify imatinib-associated AE potential signals, with their equations and decision criteria detailed in Supplementary Table 1. Signal detection rules were applied at both the system organ class (SOC) and preferred term (PT) levels. A signal was flagged at the SOC level if at least one of the four indices met the predefined criteria, while PT-level signals required concurrent satisfaction of all four criteria [12, 13]. Higher scores across these parameters generally indicated stronger disproportionality [14]. Statistical analyses were performed using SAS software (version 9.4), as recommended by the FDA. Raw ASCII data downloaded from the FDA website were imported into SAS, deduplicated following FDA-recommended protocols, and subsequently analyzed.
Results
General characteristics
In the FAERS database, 56,364 reports were identified with 170,659 AEs attributed to imatinib, averaging 3.02 AEs per individual. Females accounted for 24,179 cases (42.9%), males 27,808 (49.34%), and 4377 (7.77%) had unspecified gender. Age data were available for 28,962 reports (mean age = 59 ± 14 years). Reports aged < 18 years (n = 1180, 2.09%), 18–44 years (n = 5721, 10.15%), 45–64 years (n = 10,954, 19.43%), and ≥ 65 years (n = 11,107, 19.71%) comprised the cohorts. The highest reporting year was 2014 (n = 5131, 9.10%). Most reports originated from consumers (39.98%), with the USA contributing the majority (n = 16,962, 30.09%). The primary indication was chronic myeloid leukemia (n = 25,664, 45.53%). Severe outcomes constituted 84.24% of reports, predominantly death (n = 19,409, 34.44%) and other serious outcomes (n = 24,861, 44.11%) (Table 1). AE occurrence time (excluding unspecified medication dates) peaked at 0–30 days (24.22%) and > 360 days (38.08%) (Fig. 2).
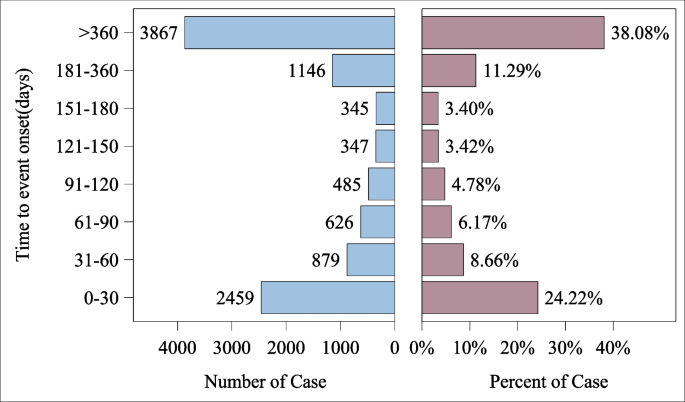
Time to event report distribution of AE reports
Imatinib-associated AEs affected multiple organ systems
The top four involved categories were general disorders and administration site conditions (40,355, 23.65%), gastrointestinal disorders (17,125, 10.03%), neoplasms benign/malignant/unspecified (15,433, 9.04%), and investigations (14,428, 8.45%) (Table 2).
Disproportionality analysis of imatinib AEs
Using the ROR method, the top 50 PTs by potential signal frequency and strength were ranked (Fig. 3). Frequent potential events signals included death (n = 15,667; ROR (95% CI) = 7.34 (7.22–7.46)), nausea (n = 3000; ROR = 1.39 (1.34–1.44)), diarrhea (n = 2532; ROR = 1.46 (1.40–1.52)), malignant neoplasm progression (n = 2177; ROR = 8.33 (7.98–8.69)), and vomiting (n = 2000; ROR = 1.57 (1.50–1.64)). The strongest potential signals (highest ROR) were blast crisis in myelogenous leukemia (n = 762; ROR = 612.66 (542.99–691.27)), pubertal failure (n = 7; ROR = 452.74 (143.69–1426.52)), large intestine fibroma (n = 4; ROR = 431.18 (96.50–1926.58)), slit-lamp tests abnormal (n = 5; ROR = 404.23 (108.55–1505.38)), and chromosome analysis abnormal (n = 124; ROR = 326.24 (254.21–418.68)). A four-algorithm integrated analysis (Table 3) ranked the frequent potential events signals as [1] death (n = 15,667; ROR = 7.34 (7.22–7.46); PRR = 6.76; IC = 2.73; EBGM = 6.64), [2] nausea (n = 3000; ROR = 1.39 (1.34–1.44); PRR = 1.38; IC = 0.47; EBGM = 1.38), [3] diarrhea (n = 2532; ROR = 1.46 (1.40–1.52); PRR = 1.45; IC = 0.54; EBGM = 1.45), [4] drug ineffective (n = 2436; ROR = 0.67 (0.64–0.69); PRR = 0.67; IC = − 0.57; EBGM = 0.67), and [5] malignant neoplasm progression (n = 1519; ROR = 8.33 (7.98–8.69); PRR = 8.23; IC = 3.01; EBGM = 8.05). By potential signal strength (ROR lower limit, Table 4), the top potential signals were [1] blast crisis in myelogenous leukemia (ROR = 612.66 (542.99–691.27); PRR = 609.93; IC = 7.73; EBGM = 211.98), [2] pubertal failure (ROR = 452.74 (143.69–1426.52); PRR = 452.72; IC = 7.56; EBGM = 189.22), [3] large intestine fibroma (ROR = 431.18 (96.50–1926.58); PRR = 431.17; IC = 7.53; EBGM = 185.36), [4] slit-lamp tests abnormal (ROR = 404.23 (108.55–1505.38); PRR = 404.22; IC = 7.49; EBGM = 180.21), and [5] chromosome analysis abnormal (ROR = 326.24 (254.21–418.68); PRR = 326.00; IC = 7.35; EBGM = 162.84).
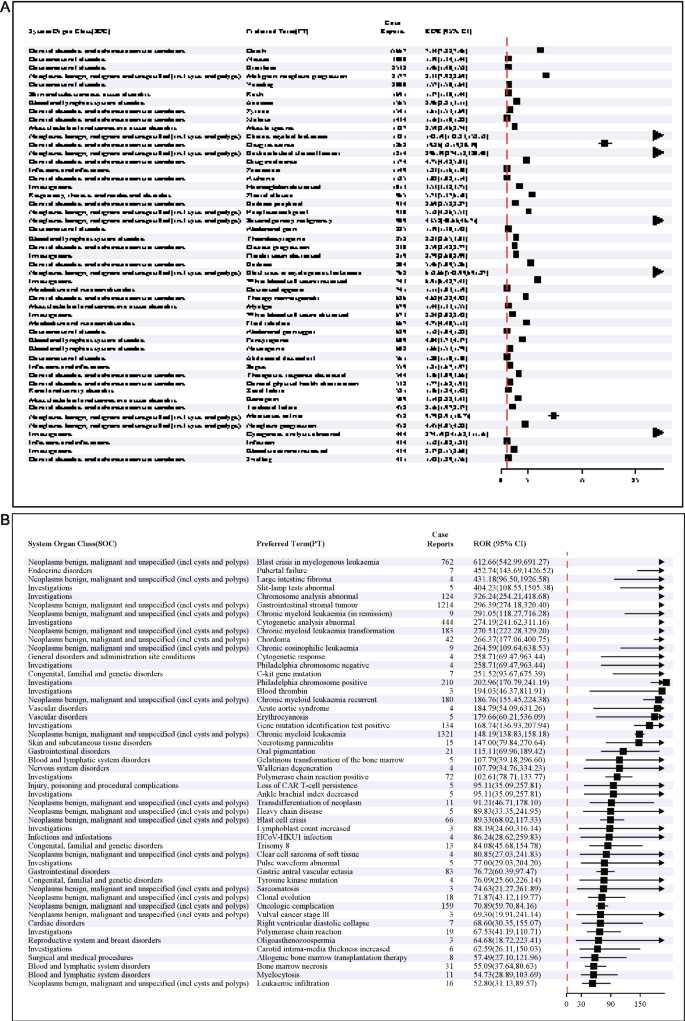
Signal strength of AEs at the PT level in ROR. A Ranked by frequency of positive signals. B Ranked by intensity of positive signals
Discussion
This study is the first comprehensive and systematic pharmacovigilance investigation, using the FDA Adverse Event Reporting System (FAERS) database to analyze the related adverse events of imatinib after its market launch. Among them, signal detection is the most important part of the study and also the first step.
This study revealed that AEs associated with imatinib occurred slightly more frequently in males (49.34%) than females (42.9%), aligning with epidemiological trends [15]. The majority of affected reports (39.14%) were aged ≥ 45 years, reflecting the predominant demographic of CML and GIST populations [16]. Consumers submitted 39.98% of reports, consistent with the spontaneous reporting nature of the FAERS database [17]. Severe AEs accounted for 84.24% of imatinib-related reports, likely attributable to its mechanism as a multi-target Abl kinase inhibitor, which modulates critical biological pathways [18, 19]. The high mortality rate (34.44% of outcomes) may stem from life-threatening complications such as tumor progression or organ failure. Notably, 38.08% of AEs occurred > 360 days post-treatment, underscoring the necessity for long-term safety monitoring. At the SOC level, the frequent potential events categories included general disorders and administration site conditions, gastrointestinal disorders, and neoplasms benign, consistent with SPCs’ safety profiles. Common PTs such as nausea, diarrhea, vomiting, and malignant neoplasm progression were frequently reported, corroborating clinical trial findings [20, 21].
While most AEs in our analysis were largely consistent with safety data from drug SPCs and clinical trials, we identified previously unreported significant AE potential signals not explicitly documented in regulatory trials. These included new-onset benign tumors (e.g., lipomas, fibromas) or neoplasms of uncertain behavior. The FDA SPCs for imatinib primarily focus on its therapeutic efficacy against existing malignancies (e.g., GIST, CML) and known complications (e.g., tumor hemorrhage) but do not explicitly address the potential induction of or association with new benign or unclassified neoplasms during treatment [22]. The insidious toxicities associated with long-term medication require particular attention, such as sciatica, joint ankylosis, and even ischemic necrosis of the femoral head observed in the musculoskeletal system, while the FDA SPCs merely mentions “musculoskeletal pain” without specifying severity or chronic consequences [23]. Severe dermatological reactions like exfoliative dermatitis and acute generalized exanthematous pustulosis are inadequately characterized in product SPCs despite documented progression risks in clinical cases [24, 25]. We advocate for enhanced long-term monitoring, particularly targeting musculoskeletal toxicity, metabolic syndrome, and other insidious risks, with recommendations including regular bone density assessments and systematic skin reaction evaluations. This call emphasizes the necessity to bridge SPCs-practice discrepancies through proactive surveillance mechanisms.
Under the SOC “Investigations,” PTs encompassed non-specific enzymatic elevations (e.g., alkaline phosphatase (ALP), lactate dehydrogenase [26]) or structural organ changes identified via imaging. While the FDA SPCs lists abnormal liver function as a monitoring parameter, it does not classify “investigation abnormalities” as standalone adverse reactions, potentially underestimating their impact on clinical decision-making [27, 28].
For the SOC “Pregnancy, Puerperium, and Perinatal Conditions,” cases accounted for 0.41% of reports, yet the ROR method detected 13 potential positive signals (1.07%). Specific PTs included fetal developmental anomalies and gestational complications (e.g., miscarriage, preterm delivery). The FDA currently warns only of “embryo-fetal toxicity” and recommends contraception but lacks explicit risk data on imatinib use during pregnancy [29, 30]. This underscores the need for enhanced monitoring of pregnancy-exposed reports and research into imatinib’s potential effects on placental function or fetal organogenesis. Within the psychiatric disorders (SOC), “psychiatric symptoms” accounted for 1.58% of reports, with specific PTs including depression, anxiety, and cognitive impairment. Notably, the FDA-approved SPCs for imatinib do not list psychiatric symptoms as common or severe adverse reactions, only mentioning that impaired driving ability may be associated with fatigue [31, 32]. Further investigation is warranted to determine whether imatinib crosses the blood–brain barrier to directly alter neurotransmitter activity or synergizes with the psychological burden inherent to chronic illness.
The FDA-approved SPCs for imatinib do not currently acknowledge ototoxicity-related risks. However, our study identified potential signals in the Ear and Labyrinth Disorders System Organ Class (SOC), including preferred terms (PTs) such as tinnitus, hearing impairment, and vestibular dysfunction [33]. This finding underscores the need for proactive monitoring of vestibular function in reports undergoing long-term imatinib therapy, particularly among elderly individuals or those with comorbid renal impairment.
This study leverages the inherent advantages of large-scale real-world investigations and sophisticated data mining techniques. However, several limitations necessitate careful consideration. The FAERS database, as a spontaneous reporting system, may introduce analytical biases due to incomplete or inaccurate data collection across countries and healthcare professionals, including reporting bias and indication bias, with challenges in distinguishing adverse events caused by the drug versus disease progression. Additionally, adverse events with extremely low incidence rates associated with imatinib might lack statistical significance in disproportionality analyses, potentially leaving undetected safety potential signals. The specificity of attributing adverse events to imatinib is further limited by confounding from concomitant medications. Finally, while disproportionality analysis identifies statistical significance based on potential signal strength, it cannot fully eliminate confounding effects from polypharmacy.
Conclusion
This study conducted a real-world pharmacovigilance analysis of imatinib using the FDA Adverse Event Reporting System (FAERS) database. Disproportionality analysis (ROR, PRR, BCPNN, EBGM) was applied to evaluate adverse events (AEs) from 2004 to 2024. Results confirmed expected AEs (e.g., nausea, diarrhea) aligned with drug SPCs, while identifying undocumented potential signals such as pubertal failure (ROR = 452.74), large intestine fibroma (ROR = 431.18), ototoxicity, and pregnancy complications. Severe outcomes accounted for 84.24% of reports (34.44% death), with 38.08% of AEs occurring > 360 days post-medication. The study underscores the need for vigilant monitoring of long-term toxicities (e.g., bone metabolism disorders, psychiatric effects) and rare AEs to enhance patient safety.
These findings highlight the necessity of conducting rigorous monitoring of the long-term toxicity and rare AEs of imatinib, particularly in terms of potential long-term effects such as adolescent developmental disorders, colonic fibromas, ototoxicity, and pregnancy. The results of this study not only provide important references for clinicians, alerting them to pay more attention to the potential risks of long-term medication use when using imatinib, but also provide a basis for optimizing drug vigilance strategies. We suggest that in future drug vigilance work, enhanced monitoring and research of rare AEs should be conducted, and further confirmation of the authenticity and relevance of these potential signals should be achieved through multi-source data verification. Additionally, these findings may also prompt regulatory agencies to re-evaluate the safety of imatinib and consider updating relevant information in the drug instructions and regulatory policies to better ensure patient safety.
Data availability
No datasets were generated or analysed during the current study.
References
-
Cohen P (2002) Protein kinases – the major drug targets of the twenty-first century? Nat Rev Drug Discovery 1(4):309–315
-
Witte ON, Dasgupta A, Baltimore D (1980) Abelson murine leukaemia virus protein is phosphorylated in vitro to form phosphotyrosine. Nature 283(5750):826–831
-
O’Brien SG, Guilhot F, Larson RA, Gathmann I, Baccarani M, Cervantes F et al (2003) Imatinib compared with interferon and low-dose cytarabine for newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med 348(11):994–1004
-
Kantarjian H, O’Brien S, Jabbour E, Garcia-Manero G, Quintas-Cardama A, Shan J et al (2012) Improved survival in chronic myeloid leukemia since the introduction of imatinib therapy: a single-institution historical experience. Blood 119(9):1981–1987
-
Program S (2013) Fast Stats: an interactive tool for access to SEER cancer statistics
-
Chihara D, Ito H, Matsuda T, Katanoda K, Shibata A, Saika K et al (2012) Decreasing trend in mortality of chronic myelogenous leukemia patients after introduction of imatinib in Japan and the US. Oncologist 17(12):1547–1550
-
Bower H, Bjorkholm M, Dickman PW, Hoglund M, Lambert PC, Andersson TML (2016) Life expectancy of patients with chronic myeloid leukemia approaches the life expectancy of the general population. J Clin Oncol 34(24):2851-+
-
Su S, Wu LQ, Zhou GB, Peng LL, Zhao HZ, Wang X et al (2023) Indication and adverse event profiles of denosumab and zoledronic acid: based on US FDA adverse event reporting system (FAERS). Front Pharmacol 14
-
Sun L, Sun S, Wang TL, Li JY, Lin JS (2019) Parallel ADR detection based on spark and BCPNN. Tsinghua Sci Technol 24(2):195–206
-
Lindquist M, Ståhl M, Bate A, Edwards IR, Meyboom RHB (2000) A retrospective evaluation of a data mining approach to aid finding new adverse drug reaction signals in the WHO International Database. Drug Saf 23(6):533–542
-
Hauben M, Zou C, Bright S, Hung E (2021) More extreme duplication in the US FDA FAERS database and a suggested check point for disproportionality analysis. Pharmacoepidemiol Drug Saf 30(8):1140–1141
-
Hu YY, Gong J, Zhang LF, Li XL, Li XN, Zhao B et al (2020) Colitis following the use of immune checkpoint inhibitors: a real-world analysis of spontaneous reports submitted to the FDA adverse event reporting system. Int Immunopharmacol 84
-
Sakaeda T, Tamon A, Kadoyama K, Okuno Y (2013) Data mining of the public version of the FDA adverse event reporting system. Int J Med Sci 10(7):796–803
-
Guo ML, Shu YM, Chen GS, Li J, Li FE (2022) A real-world pharmacovigilance study of FDA adverse event reporting system (FAERS) events for niraparib. Sci Rep 12(1)
-
Berger U, Maywald O, Pfirrmann M, Lahaye T, Hochhaus A, Reiter A et al (2005) Gender aspects in chronic myeloid leukemia: long-term results from randomized studies. Leukemia 19(6):984–989
-
Senapati J, Jabbour E, Kantarjian H, Short NJ (2023) Pathogenesis and management of accelerated and blast phases of chronic myeloid leukemia. Leukemia 37(1):5–17
-
Li H, Sun XM, Sun D, Zhao J, Xu ZM, Zhao P et al (2021) Thromboembolic events associated with immune checkpoint inhibitors: a real-world study of data from the food and drug administration adverse event reporting system (FAERS) database. Int Immunopharmacol 98
-
Hoemberger M, Pitsawong W, Kern D (2020) Cumulative mechanism of several major imatinib-resistant mutations in Abl kinase. Proc Natl Acad Sci USA 117(32):19221–19227
-
Carlisle BG, Zheng T, Kimmelman J (2020) Imatinib and the long tail of targeted drug development. Nat Rev Clin Oncol 17(1):1–3
-
Lyseng-Williamson K, Jarvis B (2001) Imatinib Drugs 61(12):1765–1774
-
Joensuu H, Trent JC, Reichardt P (2011) Practical management of tyrosine kinase inhibitor-associated side effects in GIST. Cancer Treat Rev 37(1):75–88
-
Casali PG, Le Cesne A, Velasco AP, Kotasek D, Rutkowski P, Hohenberger P et al (2021) Final analysis of the randomized trial on imatinib as an adjuvant in localized gastrointestinal stromal tumors (GIST) from the EORTC Soft Tissue and Bone Sarcoma Group (STBSG), the Australasian Gastro-Intestinal Trials Group (AGITG), UNICANCER, French Sarcoma Group (FSG), Italian Sarcoma Group (AGITG), UNICANCER, French Sarcoma Group (FSG), Italian Sarcoma Group (ISG), and Spanish Group for Research on Sarcomas (GEIS). Ann Oncol 32(4):533–541
-
Kumar S, Bansal D, Prakash M, Sharma P (2014) Avascular necrosis of femoral head as the initial manifestation of CML. Pediatr Hematol Oncol 31(6):568–573
-
Raj A, Rai R, Rangarajan B (2011) Exfoliative dermatitis with leukemia cutis in a patient with chronic myeloid leukemia: a rare association. Indian J Dermatol Venereol Leprol 77(2):208-U10
-
Banka N, Aljurf M, Hamadah I (2003) Imatinib (STI-571)-induced exfoliative dermatitis in a Saudi patient with deck chair sign. Dermatology 207(3):329–30
-
Hilly O, Gil Z, Goldhaber D, Amit M, Biadsee A, Popovtzer A et al (2015) Elective neck dissection during salvage total laryngectomy – a beneficial prognostic effect in locally advanced recurrent tumours. Clin Otolaryngol 40(1):9–15
-
Treiber G, Wex T, Schleyer E, Troeger U, Hosius C, Malfertheiner P (2008) Imatinib for hepatocellular cancer – focus on pharmacokinetic/pharmacodynamic modelling and liver function. Cancer Lett 260(1–2):146–54
-
Judson IR (2008) Imatinib for patients with liver or kidney dysfunction: no need to modify the dose. J Clin Oncol 26(4):521–2
-
Pye SM, Cortes J, Ault P, Hatfield A, Kantarjian H, Pilot R et al (2008) The effects of imatinib on pregnancy outcome. Blood 111(12):5505–8
-
Robertson HF, Milojkovic D, Butt N, Byrne J, Claudiani S, Copland M et al (2024) Expectations and outcomes of varying treatment strategies for CML presenting during pregnancy. Br J Haematol 205(3):947–55
-
Tian XF, Wang CT, Yu BA, Fan YX, Zhang L, Zhang X (2023) 9.4 T static magnetic field ameliorates imatinib mesylate-induced toxicity and depression in mice. Eur J Nuclear Med Mol Imaging 50(2):314–27
-
Phillips KM, Pinilla-Ibarz J, Sotomayor E, Lee MR, Jim HSL, Small BJ et al (2013) Quality of life outcomes in patients with chronic myeloid leukemia treated with tyrosine kinase inhibitors: a controlled comparison. Support Care Cancer 21(4):1097–103
-
Lin HW, Roberts DS, Kay J, Stankovic KM (2012) Sensorineural hearing loss following imatinib (Gleevec) administration. Otolaryngol-Head Neck Surg 146(2):335–7
Ethics declarations
Ethics approval
There are no ethical issues involved in this study.
Competing interests
The authors declare no competing interests.
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors, and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (DOC 99 KB)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
Reprints and permissions
About this article
Cite this article
Xia, T., Shu, B., Peng, Y. et al. Post-marketing safety signals of imatinib: pharmacovigilance insights from the FDA Adverse Event Reporting System (FAERS) and implications for clinical practice. Eur J Clin Pharmacol (2025). https://doi.org/10.1007/s00228-025-03872-0
- Received
- Accepted
- Published
- DOIhttps://doi.org/10.1007/s00228-025-03872-0
Keywords
- Imatinib
- FAERS
- Disproportionality analysis
- Pharmacovigilance
- Safety profile
