Article Content
1 Introduction
Recently, BaTiO3 perovskite nanoparticles, as environmentally friendly material, have much interest due to significant properties such as high dielectric, optical, ferroelectric, and piezoelectric [1,2,3,4]. Additionally, it has widespread applications of electronic, and optoelectronic such as chemical sensors, nonlinear optics, nonvolatile memories, capacitors, transducers, and actuators [5,6,7]. BaTiO3 perovskite is one of the foremost lead-free ferroelectric materials and has several polymorphs like cubic, tetragonal, orthorhombic, and rhombohedral phase [8, 9]. Further, BaTiO3 perovskite undergoes many phase transitions with increasing the temperature such as the transition from tetragonal to cubic phase at Curie temperatures (Tc). Below Tc, the perovskite structure of BaTiO3 is tetragonal phase with spontaneous polarization along the c axis while above Tc, the perovskite structure of BaTiO3 is a cubic paraelectric phase [9, 10]. Modern experiments have reported the presence of both ferroelectric and ferromagnetism occurring at the surface of BaTiO3 perovskite nanoparticles which make it useful for magneto-electric applications [8, 9]. Mangalam et al. [8] reported that the oxygen vacancies at surface of BaTiO3 nanoparticles induced ferromagnetism while the ferroelectric arising from the core. Over the last decade, the compositional modifications of BaTiO3 perovskite are used to improve its physical properties such as optical, magnetic, dielectric, ferroelectric and piezoelectric properties [11, 12]. Ali et al. [13] investigated the ferroelectric, and piezoelectric properties of (1 − x) BaTiO3 − x (BaNi0.5Nb0.5TiO3) (where 0 ≤ x ≤ 0.05) perovskite ceramics. They found that the ferroelectric parameters of BaTiO3 depend on x value and the piezoelectric properties of BaTiO3 are enhanced with increasing x. Karoblis et al. [5] investigated the structural, morphological and magnetic properties of (1 − x) BaTiO3 − x(BiMnO3) (where x = 0, 0.1, 0.2, 0.3, 0.4, 0.5, and 0.6) solid solutions. They found that the magnetization of BaTiO3 increased with increasing BiMnO3 content in the solid solutions. Therefore, the enhanced properties and applications of the BaTiO3 nanoparticles were achieved by using the optimized preparation method and the introducing of the magnetic material into BaTiO3 host [1, 7]. Among magnetic materials, strontium ferrite (SrFeO3-δ) perovskite has much great interest due to its unique structural, optical and magnetic properties which depend on the values of the oxygen deficient (δ) and the oxidation states of Fe cations [14, 15]. Further, SrFeO3-δ perovskite exhibits antiferromagnetic order at Neel temperature (TN) of 134 K [14]. Based on these properties, SrFeO3-δ materials have many applications such as fuel cells, oxygen sensors, and memory devices [14, 15]. Additionally, SrFeO3-δ perovskite materials have various phases which depend on the values of δ, like cubic, tetragonal, orthorhombic structure at δ = 0, 0.125, and 0.25, respectively [15,16,17]. Besides the benefits of SrFeO3-δ perovskite, it is suitable for integration into other materials as a solid solution [11, 18]. Manimuthu et al. [17] reported that the oxygen stoichiometry in SrFeO3-δ materials plays an important role in determining the thermal, dielectric, and ferroelectric properties. They found for the first time the presence of ferroelectric behaviour in SrFeO2.91 through the dielectric, thermal, and polarization measurements. Abd El-Naser et al. [14] investigated the thermal, dielectric, magnetic properties of SrFeO3-δ perovskite nanoparticles. They found the presence of ferroelectric phase transition (Tc) and the magnetic state is mainly antiferromagnetic with small amount of weak ferromagnetic. Hung et al. [11] reported that the SrFeO3-δ materials are suitable for integration into Bi0.5Na0.5TiO3 materials as a solid solution and they found that the Sr and Fe cations were distributed randomly into the lattice of the Bi0.5Na0.5TiO3 host materials. Further, their findings showed that the magnetization and the optical band gap of host materials enhanced and reduced with increasing SrFeO3-δ content, respectively [11]. Among the synthesis methods, the solid solution is a promising method for modifying the properties of the functional materials [5]. In view of the aforementioned aspects, it is interested to explore the influence of SrFeO3-δ doping on structural, thermal, optical, magnetic, dielectric, ferroelectric and piezoelectric properties of the BaTiO3-δ perovskite nanoparticles.
2 Experimental
2.1 Synthesis of (1-x) BaTiO3-δ − xSrFeO3-δ (where x = 0 and 0.05) perovskite samples
BaTiO3-δ perovskite sample was prepared by sol–gel method. The grade chemicals of barium acetate Ba(C2H3CO2)2 (Alpha Aesarr 99%), titanium (IV) isopropoxide C12H28O4Ti (Alpha Chemika ≥ 99%), citric acid monohydrate (C6H8O7.H2O) (Chempur 99.9%), and ethylene glycol C2H6O2 (Alpha Chemika 99.5%) were used as starting materials. The experimental flowchart of the sol–gel method for the synthesis of the BaTiO3-δ perovskite is shown in Fig. 1. The molar ratio of the C6H8O7.H2O, Ba(C2H3CO2)2, and C12H28O4Ti are 3:1:1. The C6H8O7.H2O was dissolved in 20 ml of distill water on a hot plate at 90 °C with stirring. C12H28O4Ti was added to the above solution with stirring at the same temperature. The PH of the solution was adjusted by adding nitric acid (HNO3, 55%) to prevent the formation of any precipitates. After that, Ba(C2H3CO2)2 was added to the above solution which dissolved rapidly. Then C2H6O2 was added to the above solution at 90 °C with stirring for 1.5 h to obtain a homogeneous solution. The obtained solution was concentrated by the evaporation of solvent at 250 °C to get viscous gel. The formed gel was dried in the furnace at 250 °C and the crushed powder was calcined in air at a temperature of 750 °C for 3 h. Finally, the calcined powders were pressed into pellets and sintered at a temperature of 1150 °C for 9 h in air and then cooled to room temperature (RT) to obtain the required BaTiO3-δ perovskite. The sample with the composition 0.95BaTiO3-δ − 0.05SrFeO3-δ was prepared by conventional solid state reaction method. The appropriate amounts of SrFeO3-δ (prepared in ref 14) and BaTiO3-δ (prepared above) were mixed together by planetary ball milling technology with absolute ethanol for 4.5 h at 500 rpm. Subsequently, the powder mixture obtained by ball milling was dried for 2 h at 170 °C in a drying oven and then pressed into pellets. To obtain the required sample, the pellets were sintered in air at 1050 °C for 12 h and cooled to RT.
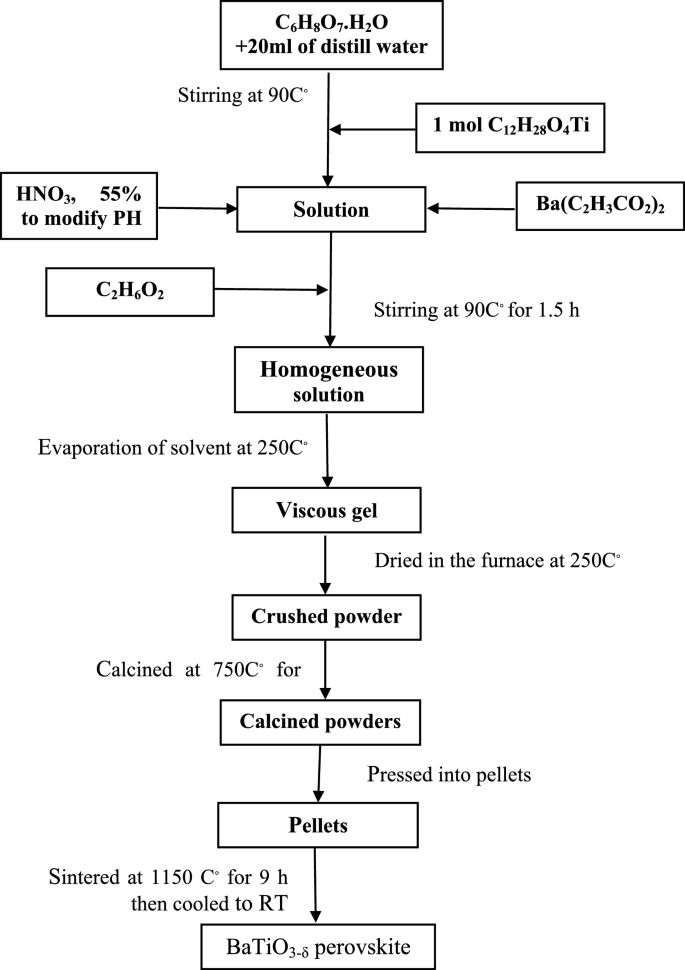
Flowchart of the sol–gel method for the synthesis of the BaTiO3-δ perovskite
2.2 Characterization of the present samples
The crystal structure of the (1-x) BaTiO3-δ − xSrFeO3-δ (where x = 0 and 0.05) samples was investigated by X-ray diffraction (XRD: PANalytical B.V. X-ray diffractometer) with Cu Kα radiation (λ = 1.5406 Å). The XRD data of the samples were recorded in 2θ ranging from 20° to 80° and scanning rate of 0.013°. Rietveld refinement of the crystal structure was performed by the FULLPROF software suite. The surface morphology of these samples was investigated using scanning electron microscopy equipped. The morphology of these samples was investigated using transmission electron microscopy (TEM: model JEM-2100 PLUS (Japan) with LaB6 source at an accelerating voltage of 200 kV). The energy dispersive X-ray spectroscopy (EDS) elemental mapping analysis of these samples was investigated using a JEM-2100 F (URP) instrument operating at 200 kV. The chemical states of these samples were investigated by K-Alpha™ X-ray photoelectron spectrometer (Thermo Scientific™ (USA)) with a source of AL-Kα exciting radiation within an energy range up to 4 keV. Thermal properties of these samples were investigated by employing differential scanning calorimetry (DSC) (TA Instrument STA model 650) with an accuracy of ± 1 °C. The optical properties of these samples were investigated by UV–Vis. absorption spectroscopy which performed using a Jasco-V-570 spectrophotometer. The errors in the values of the absorption edges and Eopt are ± 1 nm and ± 0.01 eV, respectively. Magnetic hysteresis (M-H) loops of these samples were investigated using vibrating sample magnetometer (VSM of Lakeshore 7410a instruments) at RT. The dielectric constant (εr) of these samples as a function of temperature at different frequencies in 0.120–100 kHz range was performed using electronic RLC bridge type SR 720. Polarization–electric field (P-E) loops of these samples were investigated using ferroelectric tester (Precision premier II, Radiant technologies Inc., USA) at various electric fields and frequency of 10 Hz. Bipolar strain- electric field (S-E) loops of these samples were investigated using bulk ceramic piezoelectric test bundle (B-PTB)) under electric field of 25 kV/cm and frequency of 10 Hz. To study the pulse-echo response, the 0.95 BaTiO3-δ − 0.05 SrFeO3-δ sample was polarized. After that, the sample was connected to the flaw detector (USN60 GE inspection Technologies) to observe the pulse-echo waveform. The flaw detector was calibrated according to standard method (ASTEM E317-21). The measurements were carried out utilizing VI standard reference block with thickness 25 mm. Finally, the nominal frequency of the piezoceramic sample will be estimated.
3 Results and discussion
3.1 XRD, SEM, TEM, SAED, and HRTEM studies
Figure 2 shows the Rietveld refinements of XRD patterns of the (1-x) BaTiO3-δ − xSrFeO3-δ (where x = 0 and 0.05) samples. These patterns exhibit single perovskite phase with small diffraction peaks at 2θ ≈ 31o and 33.89 o for SrFeO3-δ phase at x = 0.05 which indicate that all the Sr and Fe cations not diffuse into the host lattice of BaTiO3-δ [11]. The Rietveld refinement of these patterns indicates that the samples crystallized in the tetragonal structure with space group of P4mm, consistent with previous results [19, 20]. Further, the splitting of diffraction peak at 45° into 002 and 200 indicates that these samples are tetragonal ferroelectric [21]. The refined structural parameters of these samples such as the lattice parameters, unit cell volume, atoms, Wyckoff position, lattice coordinate, and reliability factors are listed in Table 1. The ratio c/a at x = 0 and 0.05 is 1.0073 and 1.0053, respectively, which indicates that these samples were in the tetragonal phase, consistent with previous results [21,22,23]. The lattice parameter (a) and the unit cell volume increase while the lattice parameter (c) decreases with introducing SrFeO3-δ (x = 0.05). The increase of the unit cell volume can be due to the difference in the ionic radii of Sr2+ (1.44 Å) and Ba2+ (1.17 Å) [11]. This behavior is similar to the solid solution of SrFeO3-δ in Bi0.5Na0.5TiO3 materials [11]. Based on the refined atomic positions, the tetragonal structure of these samples was drawn using VESTA software in three dimensional as shown in Fig. 3. This figure indicates that the Ti/Fe (B-site) cations are located at the center of octahedron of oxygen (TiO6/FeO6) and the oxygen atoms are located in two nonequivalent sites (two OI and four OII) at the octahedron vertices [24, 25]. These octahedra are shared together by corners to form three dimensional frameworks with the neighboring octahedra. Further, the Ba/Sr (A-site) cations in 12-fold coordinated with the oxygen atoms and occupy the corners of the unit cell [22, 25]. The tetragonal phase at x = 0 has higher ratio of c/a than that at x = 0.05 which indicated more distorted local structure at x = 0 [26].
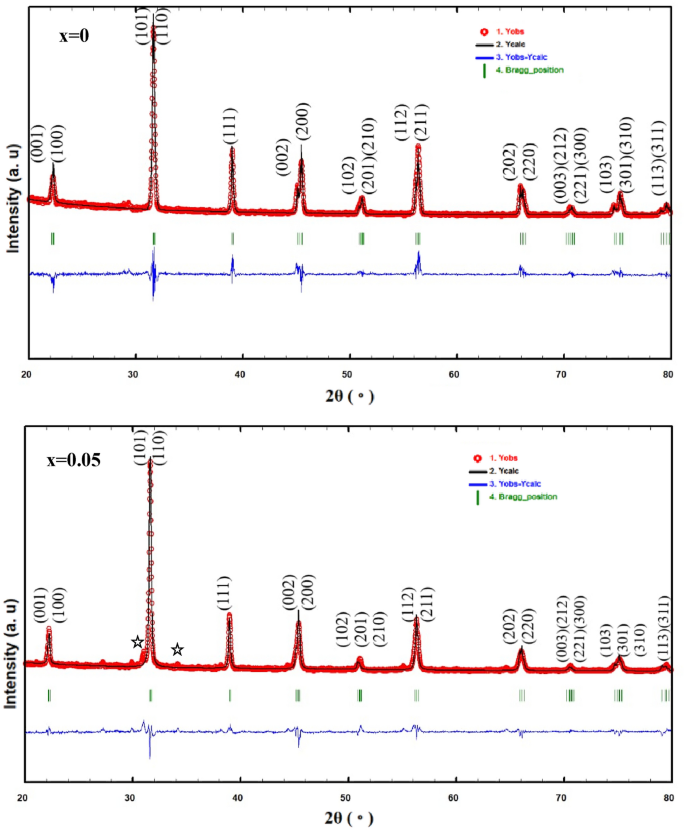
Rietveld refinement XRD patterns of the (1-x) BaTiO3-δ − xSrFeO3-δ samples with observed data (1. red dots), calculated curves (2. black line), the profile (3. blue line), and the expected Bragg’s positions (4. green vertical lines). The diffraction peaks with stars are attributed to the SrFeO3-δ
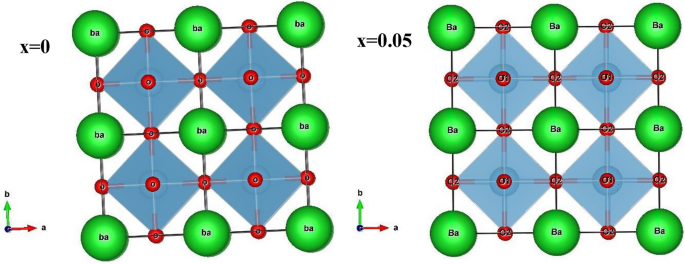
The crystal structure of the tetragonal of the (1-x) BaTiO3-δ − xSrFeO3-δ samples
Figure 4 shows the SEM images of the (1-x) BaTiO3-δ − xSrFeO3-δ (where x = 0 and 0.05) samples. This figure displays irregular grains of varying sizes with some agglomerates and porous [2, 3]. The agglomeration in these samples may be attributed to the chemical nature of the used solvent as polymerization agent [27]. The existence of porous structure and irregular shape of grains in these samples can be attributed to the sintering temperature that affected the densification and the formation of grains [3]. With introducing SrFeO3-δ (x = 0.05), the pores between the grains increase and become less dense. Further, the grain size slightly increases with introducing SrFeO3-δ (x = 0.05) which is due to the decrease in the ratio of c/a [23, 24]. Figure 5 shows the EDS elemental mapping images of the (1-x) BaTiO3-δ − xSrFeO3-δ (where x = 0 and 0.05) samples. At x = 0, the EDS elemental mapping reveals the existence of Ba (red), Ti (green), and O (blue) elements, which constitute the BaTiO3-δ sample. Further, all these elements were homogeneously distributed [28]. At x = 0.05, the EDS elemental mapping reveals the existence of Ba (green), Sr (purple), Ti (red), Fe (yellow), and O (blue) elements, which constitute the 0.95 BaTiO3-δ − 0.05 SrFeO3-δ sample. Further, all these elements were homogeneously distributed with a little loss of the uniform distribution for Fe which may be attributed to the low percentage of Fe in this sample. Figure 6 shows the TEM images of (1-x) BaTiO3-δ − xSrFeO3-δ (where x = 0 and 0.05) samples. This figure displays nearly spherical particles with aggregated nanoparticles [29]. The aggregation in these samples may be due to the dipole–dipole magnetic attraction of nanoparticles [30]. The inset of Fig. 6 shows the particle size distribution histogram of (1-x) BaTiO3-δ − xSrFeO3-δ (where x = 0 and 0.05) samples, fitted using a Gaussian function. These histograms exhibit a narrow particle size distribution, indicating uniformity in the particle size distribution. The estimated average particle sizes of these samples are 52.32, and 53.19 nm for x = 0, and 0.05, respectively. The average particle size slightly increases with introducing SrFeO3-δ (x = 0.05) which can be attributed to the decrease in the ratio of c/a [23, 31]. This result is consistent with decreasing the magnitude of off-center distortion of Ti ions [8]. Figure 7 shows the HR-TEM images of (1-x) BaTiO3-δ − xSrFeO3-δ (where x = 0 and 0.05) samples. These images display the lattice fringes, indicating the existence of nanostructure character and the high degree of crystallinity in these samples. Further, these images display different directions of lattice fringes as green open ovals, indicating the polycrystalline nature of these samples with clear crystal boundaries [30]. The interplanar distance (d) between two lattice planes is found to be 0.28 nm for these samples. The value of d may be corresponding to (1 1 0) lattice plane of these samples with tetragonal structure [32]. This result agrees with the d-values (0.274 and 0.279 nm) which estimated from the XRD analysis for the (1 1 0) plane. The inset of Fig. 7 shows the SAED patterns of the (100-x) BaTiO3-δ − xSrFeO3-δ (where x = 0 and 0.05) samples. These patterns exhibit spotty rings, indicating the polycrystalline nature of these samples [32]. Additionally, the existence of relatively sharp spots indicates the presence of different planes in these samples. These diffraction rings are agreement with the previous results for tetragonal BaTiO3 phase [32]. The change in the electron diffraction patterns with introducing SrFeO3-δ (x = 0.05) may be due to the variation in crystal parameters.

Scanning electron microscopy (FE-SEM) images of the (1-x) BaTiO3-δ − xSrFeO3-δ samples

The energy dispersive X-ray spectroscopy (EDS) elemental mapping images of the (1-x) BaTiO3-δ − xSrFeO3-δ samples
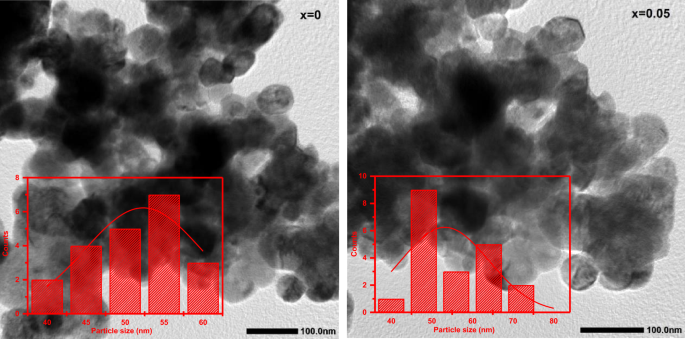
The transmission electron microscopy images of the (1-x) BaTiO3-δ − xSrFeO3-δ samples and the inset shows the particle size distribution histogram of these samples which fitted by using the Gaussian function
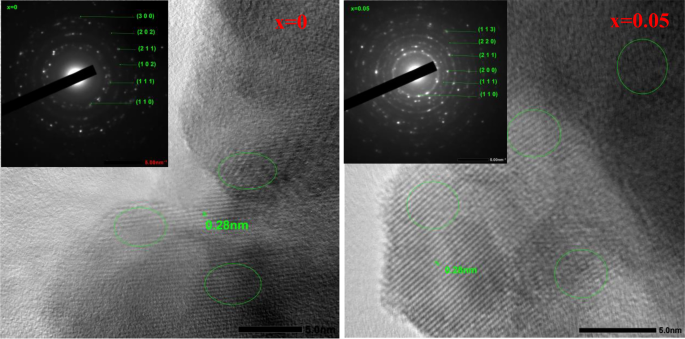
The high-resolution TEM (HR-TEM) images of the (1-x) BaTiO3-δ − xSrFeO3-δ samples and the inset shows the selected area electron diffraction (SAED) patterns of these samples. The different directions of lattice fringes have been marked green open ovals
3.2 XPS, thermal, optical and magnetic studies.
Figure 8(a, b, c, d and e), shows the XPS spectra of Ba 3d, Ti 2p, O 1 s, Sr 3d, and Fe 2p core levels of the (1-x) BaTiO3-δ − xSrFeO3-δ (where x = 0 and 0.05) samples, respectively. These spectra confirmed that the sample at x = 0 is composed of Ba, Ti, and O elements while the sample at x = 0.05 is composed of Ba, Sr, Ti, Fe, and O elements without any other impurity. As shown in Fig. 8(a), the XPS spectra of the Ba 3d are composed of two peaks at 780.38 and 795.78 eV for x = 0 as well as at 778.08 and 793.28 eV for x = 0.05. These peaks at binding energies of 780.38 and 795.78 eV can be attributed to the Ba 3d5/2 and Ba 3d3/2 spin states, respectively [33, 34]. This result indicates that the oxidation state of Ba ion is 2 + [33, 34]. The peaks of Ba 3d are shift to lower binding energies with introducing SrFeO3-δ (x = 0.05). As shown in Fig. 8 (b), the XPS spectra of Ti 2p were deconvoluted into four individual peaks at 458.09, 458.21, 463.62, and 463.91 eV for x = 0 as well as at 457.36, 457.68, 463.11 and 463.13 eV for x = 0.05. The peaks at binding energies of 458.09 and 463.62 eV can be attributed to the Ti 2p3/2 and Ti 2p1/2 spin states, respectively [35]. Additionally, the peaks at binding energies of 458.21 and 463.91 eV also due to Ti 2p3/2 and Ti 2p1/2 spin states, respectively [35]. This result is similar to that observed for BaTiO3-δ by Kumar et al., indicating the existence of different oxidation states of Ti ions [36]. The peaks at lower binding energies (458.09 and 463.62 eV) can be due to the existence of Ti3+ ions, while those at higher binding energies (458.21 and 463.91 eV) can be due to the existence of Ti4+ ions [29, 36]. From this observation, we can conclude that the existence of defects in the present samples, which lead to the reduction of Ti4+ to Ti3+ ions [29, 34]. The binding energies of Ti 2p are shifted to lower with introducing SrFeO3-δ (x = 0.05) which lead to the decrease of the coulombic interaction between the Ti ion core and the electron. Further, this shift leads to the transformation of some of the Ti4+ in BaTiO3-δ into Ti3+ ions [29, 34]. As shown in Fig. 8 (c), the XPS spectra of O 1 s were deconvoluted into two individual peaks at binding energies 531.93 and 534.31 eV for x = 0 as well as at 528.55 and 530.22 eV for x = 0.05. These peaks indicated that there are two different kinds of oxygen states on the surface of these samples [33, 34]. The first peak is attributed to the lattice oxygen (Olatt: O2−) of the perovskite samples [29]. The second peak with higher binding energy is attributed to the defect oxygen (Odef) and may be due to adsorbed oxygen (Oads: O−/O22−) which arises from oxygen vacancies [29]. The lattice oxygen can be due to Ba–O, Sr–O, Fe–O and Ti–O in the crystal lattice of these samples. These peaks in the samples are similar to that reported in the previous reports for BaTiO3-δ and SrFeO3-δ perovskite materials [14, 33]. From the area of Odef peak, the oxygen vacancies increase with introducing SrFeO3-δ (x = 0.05). Based on the results of XPS, we concluded the presence of oxygen vacancies and mixed valence states of Fe and Ti ions (3 + and 4 +) on the surface of the samples. The presence of oxygen vacancies in these samples can be attributed to the loss of oxygen during the sintering at high temperature [3]. As shown in Fig. 8 (d), the XPS spectrum of Sr 3d was deconvoluted to into two peaks at 132.18 and 134.00 eV for x = 0.05. The first peak at 132.18 can be attributed to the Sr (3d5/2) ions incorporated into the perovskite structure with the presence of oxygen deficient [14, 37]. The second peak at 134.00 eV can be attributed the Sr (3d3/2) ions into the perovskite structure which indicates that the oxidation state of Sr ion is 2 + [14, 37]. As shown in Fig. 8(e), the XPS spectrum of Fe 2p was deconvoluted into seven individual peaks of 2p3/2 and 2p1/2 electron levels and a weak satellite peak for x = 0.05. The two peaks at binding energies of 709.99 and 724.37 eV are due to 2p3/2 and 2p1/2 states, respectively, of Fe3+ ions in this sample [14, 15]. The two peaks at binding energies of 713.00 and 728.77 eV are assigned to 2p3/2 and 2p1/2 states, respectively, of Fe4+ ions in this sample [14, 15]. Furthermore, the intermediated two peaks at binding energies of 717.63 and 721.30 eV may be attributed to the intermediate oxidation state of Fe3.5+, consistent with Mössbauer result for SrFeO3-δ perovskite [14, 15]. The peak at high binding energy of 732.86 eV is assigned to Fe 2p1/2 and it is characteristic of satellite structure [15, 30]. In addition, the peak at 717.63 eV may be due to Fe 2p3/2 satellite peak of Fe3+ ions with oxygen vacancies on the surface of this sample [15, 30]. The peaks at higher binding energy in this sample may be due to the increase of the coulombic interaction between the Fe ion core and the electron [30]. Therefore, the Fe ions in this sample exist in mixed valence states.
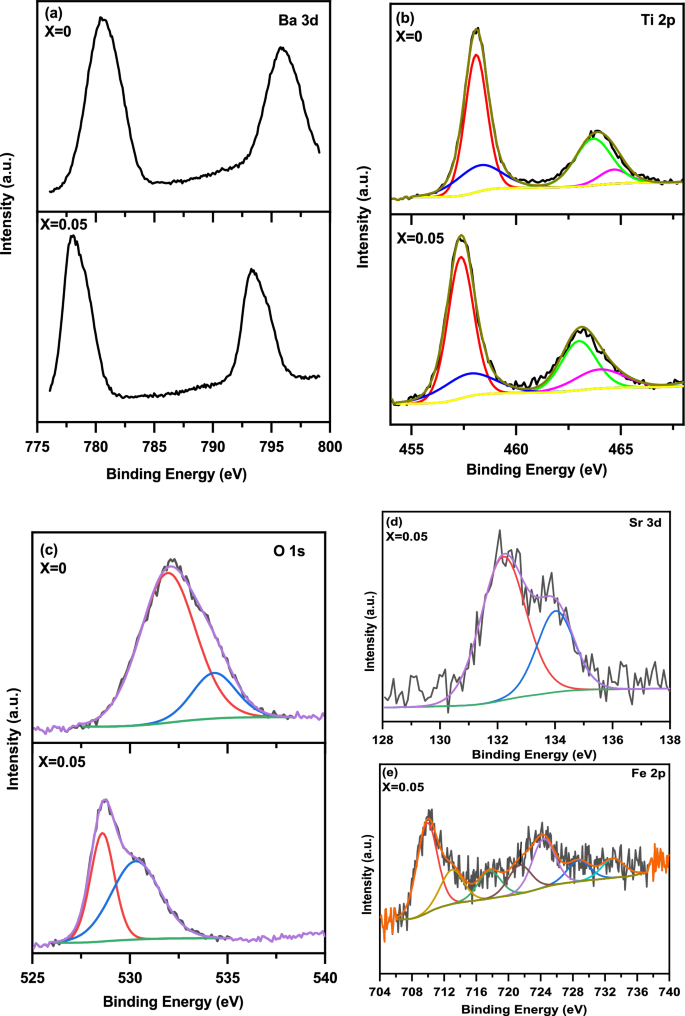
(a–e) The XPS spectra of Ba 3d, Ti 2p, O 1 s, Sr 3d, and Fe 2p core levels of the (1-x) BaTiO3-δ − xSrFeO3-δ (where x = 0 and 0.05) samples, respectively
Figure 9 shows the DSC and the derivative of DSC curves of the (1-x) BaTiO3-δ − xSrFeO3-δ (where x = 0 and 0.05) samples in the temperature range of 25–850 °C. From the shape of the DSC curves, it is expected the presence of two endothermic peaks blow 25 °C for x = 0 and x = 0.05 which represent the phase transition of BaTiO3-δ nanoparticle samples from orthorhombic to tetragonal [22, 38]. Additionally, the DSC curves exhibit two endothermic peaks near 130 and 140 °C for x = 0 and x = 0.05, respectively, which are attributed to the phase transition of BaTiO3-δ nanoparticle samples from tetragonal to cubic. This result is confirmed by subsequent dielectric results and is similar to those reported for BaTiO3 [35, 39]. Frey and Payne [39] showed that the BaTiO3 ceramics exhibit two endothermic peaks near 5 and 125 °C which are due to the first order transformations from orthorhombic to tetragonal and from tetragonal to cubic, respectively. As the temperature increase, the DSC curve at x = 0 exhibits broad exothermic peak in the 300 − 800 °C temperature range. Further, the DSC curve at x = 0.05 exhibits two broad exothermic peaks in the 300 − 500 °C and 500 − 800 °C temperature ranges. The broad exothermic peak in the derivative of DSC curve at x = 0 exhibits two exothermic peaks while the derivative of DSC curve at 0.05 exhibits three exothermic peaks. The first exothermic peak at x = 0 and x = 0.05 may be due to the oxygen loss [17]. The second exothermic peak at x = 0 may be due to the crystallization of BaTiO3-δ [40]. The second and third exothermic peaks at x = 0.05 may be due to the formation of the expected 0.95BaTiO3-δ-0.05SrFeO3-δ perovskite phase [5]. The change in the phase transition temperatures with introducing SrFeO3-δ (x = 0.05) may be attributed to the different of the ionic radii between host and dopant ions, the displacements of Ti ions, and the lattice distortion [25].
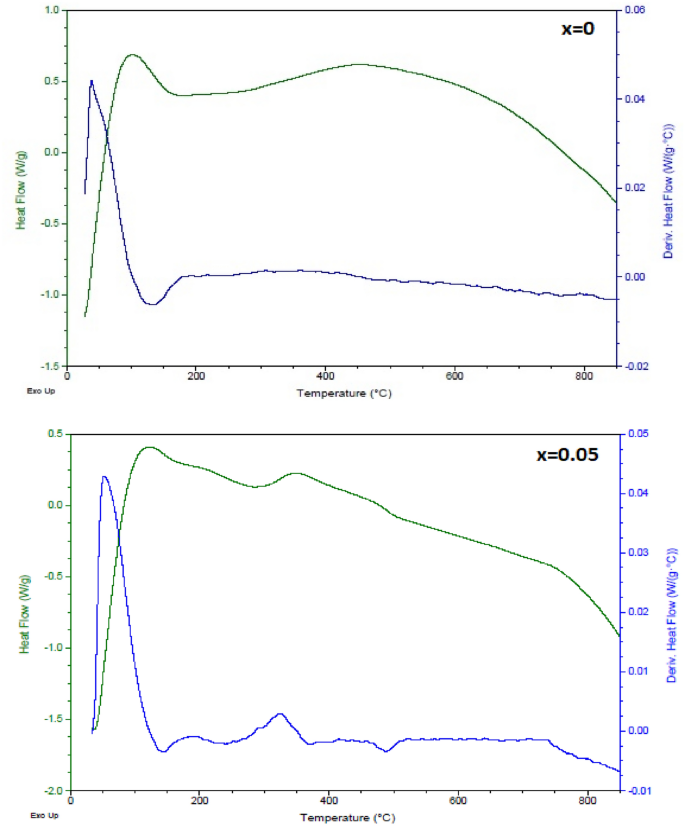
The DSC and the first derivative of DSC curves of the (1-x) BaTiO3-δ − xSrFeO3-δ samples
Figure 10 shows the UV–Vis. absorption spectra of the (1-x) BaTiO3-δ − xSrFeO3-δ (where x = 0 and 0.05) samples in the wavelength range of 200–800 nm. These spectra exhibit absorption peak around 294 nm and a shoulder at 362 nm in the UV range. Further, these spectra exhibit absorption peak around 704 nm in the Vis. Range (as shown inset of Fig. 10) besides the broad absorption peak around 502 nm for x = 0.05. The presence of absorption peak and a shoulder in the UV range can be due to the charge transfer transition between O 2p level to Ti 3d for x = 0 and between O 2p level to Ti 3d/Fe 3d orbital for x = 0.05 [14, 41, 42]. The appearance of the absorption peak around 502 nm for x = 0.05 may be due to the d-d transition between Fe center and Ti site [29, 43]. The absorption peak around 704 nm in the Vis. can be due to the overlapping of the electronic transition in the present lattice [14]. From Fig. 10, it observed that the absorption edges are 450 and 460 nm for x = 0 and x = 0.05, respectively, consistent to those reported for BaTiO3 by Bantawal et al. [34]. The error in values of the absorption edges is ± 1 nm. The absorption edges increases and wider with introducing SrFeO3-δ (x = 0.05) which indicate the existence of an inter-transition band of Fe cation in the crystal field [11]. The broad absorption peak around 502 nm in Vis range is attributed to the charge transfer between O 2p and Ti 3d/Fe 3d [14, 34]. Further, the absorption of the present samples increased in the visible region with introducing SrFeO3-δ (x = 0.05). The band gap energy (Eg) of these samples can be determined by using the following Kubelka–Munk function of reflectance equation [14].
where F(R) is Kubelka–Munk (K–M) function, R is the diffuse reflectance value of the present samples. It is known that the absorption coefficient α is directly proportional to F(R) and hence the Eg values of the (1-x) BaTiO3-δ − xSrFeO3-δ (where x = 0 and 0.05) samples have been determined from the following Wood and Tauc’s equation [14, 44].
where hν is the photon energy, C is a constant that depends on the types of transitions, and n is the kind of transition where n = ½ and 2 for direct and indirect transitions, respectively [14, 44]. The direct and indirect band gap energy (Eg1 and Eg2) of these samples were estimated from a plot of [F(R) hν]2 and [F(R) hν]1/2 versus (hν) as shown in Fig. 11 (a and b), respectively. The Eg1 and Eg2 values have been determined from the intercept of the tangents lines with [F(R) hν]2 = 0 and [F(R) hν]½ = 0, respectively. The Eg1 and Eg2 values of these samples are 3.70 and 2.75 eV at x = 0 while at x = 0.05 these values are 3.65 and 1.13 eV, respectively. The error in the values of Eg is ± 0.01 eV. The values of Eg1 and Eg2 are similar to those observed for BaTiO3 ceramics which reported by Reddy et al. [43]. The small values of Eg1 and Eg2 of these samples can be attributed to the presence of defects (oxygen vacancies) near the conduction band which lead to the formation of additional energy levels [14, 15]. The decrease of the Eg values with introducing SrFeO3-δ (x = 0.05) can be attributed to the increase of the oxygen vacancies, consistent with XPS results [11]. Additionally, the decrease in Eg values can be attributed to the random distribution of Fe and Sr cations in host lattice of BaTiO3-δ [11]. This random distribution of the cations leads to the presence of new local state near the valence band [11]. Based on the above results, the optical properties of BaTiO3-δ were enhanced with introducing SrFeO3-δ (x = 0.05) as a result of the reduction of the band gap energies and the increase of the absorption coefficient in Vis. Range [7]. Therefore, these results confirmed that the introducing of SrFeO3-δ (x = 0.05) into host lattice of BaTiO3-δ makes it a suitable material for optoelectronic applications [7].
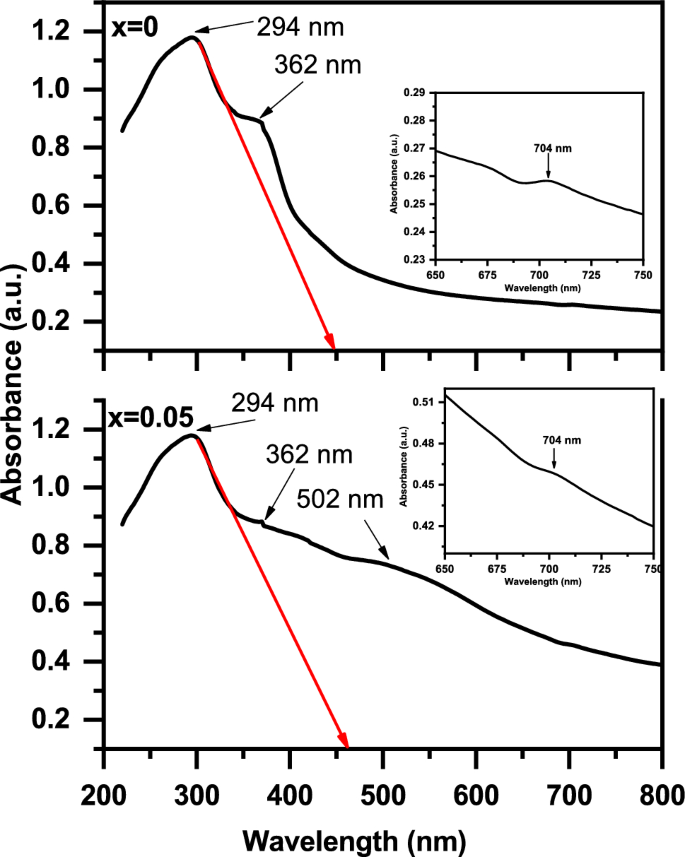
The UV–visible absorption spectra of the (1-x) BaTiO3-δ − xSrFeO3-δ samples in the wavelength range of 200–800 nm
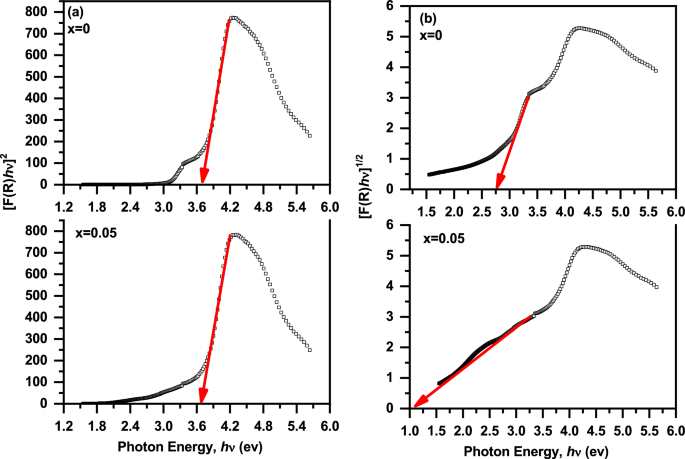
(a, b) The plot of [F(R) hν]2 and [F(R) hν]1/2 versus (hν) of the (1-x) BaTiO3-δ − xSrFeO3-δ samples, respectively
Figure 12 shows the magnetic hysteresis (M-H) loops of the (1-x) BaTiO3-δ − xSrFeO3-δ samples at room temperature. The shape of the M-H loops indicated that these samples exhibit ferromagnetic behavior and reach saturation at the applied magnetic field (20 kOe) [8, 9]. The ferromagnetic behavior in these samples, is like that reported by Mangalam et al. and Kumari et al. for the nanocrystalline BaTiO3 and Sm doped BaTiO3 ceramics, respectively [8, 9]. It is known the bulk BaTiO3 exhibits diamagnetic behavior in the magnetic hysteresis loop [8]. Therefore, the origin of ferromagnetism in present BaTiO3-δ sample is due to the exchange interactions between spin moments of unpaired electrons which arise from oxygen vacancies on the surface of the nanoparticles [45]. These results are similar to those observed for BaTiO3 which reported by Cao et al. [46]. In order to balance the oxygen vacancies in BaTiO3-δ sample, some of the Ti4+ ions were transformed to Ti3+ ions. Therefore, the unpaired electron spins in Ti3+ ions are responsible for ferromagnetism in this sample [34]. The values of the saturation magnetization (Ms), coercive field (Hc), and remnant magnetization (Mr) of the present samples are listed in Table 2. The Ms and Mr values increase with introducing 0.05SrFeO3-δ into BaTiO3-δ host. This increase can be due to the increase of the oxygen vacancies which leads to creation of the magnetic moments at the surface of this sample [8, 46]. The origin of magnetism in 0.95 BaTiO3-δ − 0.05 SrFeO3-δ sample may be due to the random distribution of Sr and Fe cations in BaTiO3-δ host where the magnetic Fe ions interact via the oxygen vacancies [11]. This result confirmed that the introducing of SrFeO3-δ (x = 0.05) into host lattice of BaTiO3-δ lead to the enhancement the ferromagnetic ordering [11]. Therefore, the enhancement of ferromagnetic ordering of this sample makes it a promising material for magneto-electric applications [11]. Moreover, the Hc value decreases with introducing 0.05SrFeO3-δ which may be due to the increase in the average particle size [47].
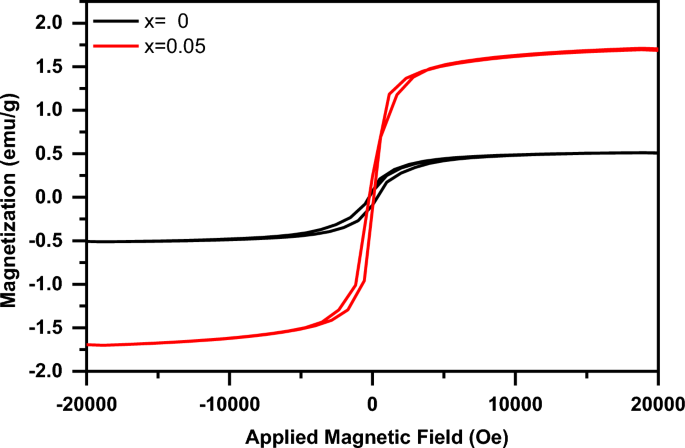
The magnetic hysteresis (M-H) loops of the (1-x) BaTiO3-δ − xSrFeO3-δ samples at room temperature
3.3 Dielectric constant, ferroelectric, and Piezoelectric studies
Figure 13 shows the dielectric constant (εr) as a function of temperature for the (1-x) BaTiO3-δ − xSrFeO3-δ samples at different frequencies in 0.120–100 kHz range. It can be seen that the sample at x = 0 exhibits two dielectric anomaly peaks at 313 and around 425 K which can be attributed to the phase transition temperature (Tc) of the BaTiO3 phase, consistent with DSC results. The error in the value of Tc is ± 1 K. From Fig. 13, it is noticed that the first sharp peak and the second broad peak does not depend on the frequency. The second broad peaks in the samples indicate the presence of diffuse type of the phase transition [3]. In accordance with Xiao et al., BaTiO3 ceramics have structural transitions between hexagonal, cubic, tetragonal, orthorhombic and rhombohedral at phase transition temperatures 1432, 130, 5, and -90 ◦C, respectively [48]. Therefore, the phase transition temperatures in the present samples at Tc = 313 and 425 K are accompanied by structural transitions from orthorhombic to tetragonal and from tetragonal to cubic, respectively [49]. The shift in the Tc values of the sample at x = 0 may be attributed to the grain size effect of BaTiO3-δ [49]. The values of ɛr are larger at lower frequencies and decrease with increasing frequency over all the temperature range. This result can be attributed to the presence of the hopping phenomenon and the space charges at low frequency while the dipoles cannot follow the Ac applied electric field at higher frequencies [50, 51]. The polarization at lower frequencies may be due to the charge carriers hopping of Ti4+ → Ti3+ and Fe4+ → Fe3+ which are aligned with the direction of electric field [51]. Moreover, the maximum values of εr at Tc can be due to the presence of high ionic and electronic polarizability [3]. The decrease in the values of εr with increasing temperature beyond the second broad peaks can be due to the decrease in the dipolar polarization because of the random orientation of dipoles [3, 17]. With introducing SrFeO3-δ (x = 0.05), the εr curve at 0.120 kHz exhibits broad dielectric anomaly at 307 and small dielectric anomaly near 499 K (as shown inset of Fig. 13) which can be attributed to the phase transition temperature (Tc) of the BaTiO3 phase, consistent with DSC results. The presence of tetragonal to cubic phase transition at 499 K instead of 425 K for x = 0.05, is like that reported by Tomar et al. for Sn doped BaTiO3 [52]. In addition, the small dielectric anomaly may be due to the motion of oxygen vacancies which is similar to that observed in ferroelectric perovskite containing BaTiO3 [53, 54]. The values of εr increase with increasing the temperature beyond the dielectric anomaly which can be attributed to the orientational polarization related to thermal motion of dipoles [49]. Further the values of εr increase with introducing SrFeO3-δ (x = 0.05) which can be attributed to the increase of oxygen vacancies, consistent with XPS results [3, 50]. Therefore, the dielectric properties of BaTiO3-δ were enhanced with introducing SrFeO3-δ (x = 0.05) which makes it a promising material for electronic applications [8].
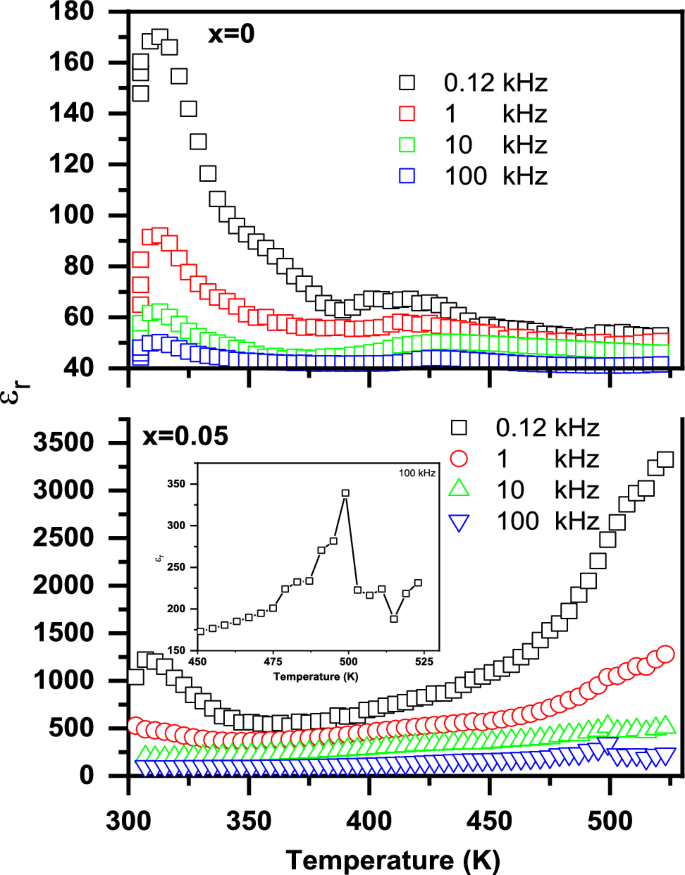
The dielectric constant (εr) as a function of temperature for the (1-x) BaTiO3-δ − xSrFeO3-δ samples at different frequencies in 0.120–100 kHz range and the inset shows the dielectric anomaly peak at 499 K
Figure 14 shows the polarization versus electric field (P-E) loops of the (1-x) BaTiO3-δ − xSrFeO3-δ samples at various electric fields. The P-E loops showed that the samples have ferroelectric nature. In addition, the P-E loop of the present samples doesn’t saturate even at electric field of 50 μC/cm2, which may be attributed to the incomplete domain reorientation. This result is similar to that observed for BaTiO3 ceramics by Rubavathi et al. [55]. The gap in the P-E loops (as shown in Fig. 14) means the presence of lossy nature of the present samples, consistent with that reported for the BaTiO3 family [17, 56]. The ferroelectric nature in these samples can be attributed to the off-centering of the Ti ions [8]. In addition, the strain induced in the lattice is due to the reduction of Ti4+ to Ti3+ at x = 0 beside the reduction of Fe4+ to Fe3+ at x = 0.05 [17]. The slightly pinched hysteresis (P-E) loop with introducing SrFeO3-δ (x = 0.05) can be due to the decrease of the tetragonal distortion, consistent with XRD results [57]. The presence of pinched hysteresis at x = 0.05 indicates the coexistence of ferroelectric and nonpolar phases [58]. As the applied electric field increases from 15 kV/cm to 50 kV/cm, the polarization of these samples increases which can be attributed to the activation of oxygen vacancies [17]. The activation of oxygen vacancies leads to the enhancement of the polarization of the dipoles [17]. The change in the ferroelectricity of BaTiO3-δ with introducing SrFeO3-δ (x = 0.05) can be due to the change of ferroelectric domains and grain sizes [13]. The enhancement of ferroelectric behaviour of BaTiO3-δ with introducing SrFeO3-δ (x = 0.05) is due to the increase of the defect dipoles and the reduction of the leakage current [13, 58]. The increase of the defect dipoles with introducing SrFeO3-δ (x = 0.05) is due to the increase of oxygen vacancies which confirmed by the aforementioned XPS results [9, 58]. In addition, the reduction of the leakage current with introducing SrFeO3-δ (x = 0.05) is due to the increase of the porous which confirmed by the aforementioned SEM results [13]. The ferroelectric parameters of the P-E loops for these samples at various electric fields such as the maximum polarization (Pmax), the remnant polarization (Pr), and the coercive field (Ec) are listed in Table 3. From Table 3, the increase in the ferroelectric parameters (Pmax, Pr and Ec) of the P-E loops with increasing the electric field may be due to the dielectric charging effect [35, 59]. The Pmax value of BaTiO3-δ increases with introducing SrFeO3-δ (x = 0.05) expect at 50 kV/cm which is due to the increase of the oxygen vacancies [9]. The Pr value of BaTiO3-δ increases with introducing SrFeO3-δ (x = 0.05) when the applied electric field increases from 15 kV/cm to 20 kV/cm. Thereafter the Pr value of BaTiO3-δ decreases with increasing the applied electric field from 20 kV/cm to 50 kV/cm. This decrease with introducing SrFeO3-δ (x = 0.05) is due to the increase of the oxygen vacancies which rise the pinning effect on the domain wall [55]. The low values of Pr and Ec in these samples can be attributed to the low internal electrical polarizability and the large grain structure, respectively [3]. Figure 15 shows the bipolar strain versus electric field (S–E) loops of the (1-x) BaTiO3-δ − xSrFeO3-δ samples at electric field of 25 kV/cm. It is noticed that the bipolar S-E loops of the samples display typical butterfly-shaped hysteresis loops and indicate the presence of piezoelectric nature of these samples [13, 60]. The shape of the S–E loop changed with introducing SrFeO3-δ (x = 0.05) content. The induced field strain at x = 0.05 may be due to the phase transition from the ferroelectric to non-polar while it may be due to the ferroelectric-domain switching at x = 0 [57, 61]. The value of the strain of BaTiO3-δ increases with introducing SrFeO3-δ (x = 0.05) which is due to the increase of the number of local nanoparticles [13, 60]. Further, the value of negative strain (i.e., the difference between the zero-field strain and the lowest strain) of BaTiO3-δ decreases with introducing SrFeO3-δ (x = 0.05) which indicates the presence of deviation from that of the typical ferroelectric materials [61, 62]. Therefore, the piezoelectric properties of BaTiO3-δ were enhanced with introducing SrFeO3-δ (x = 0.05) which makes it a promising material for electromechanical sensors [13].
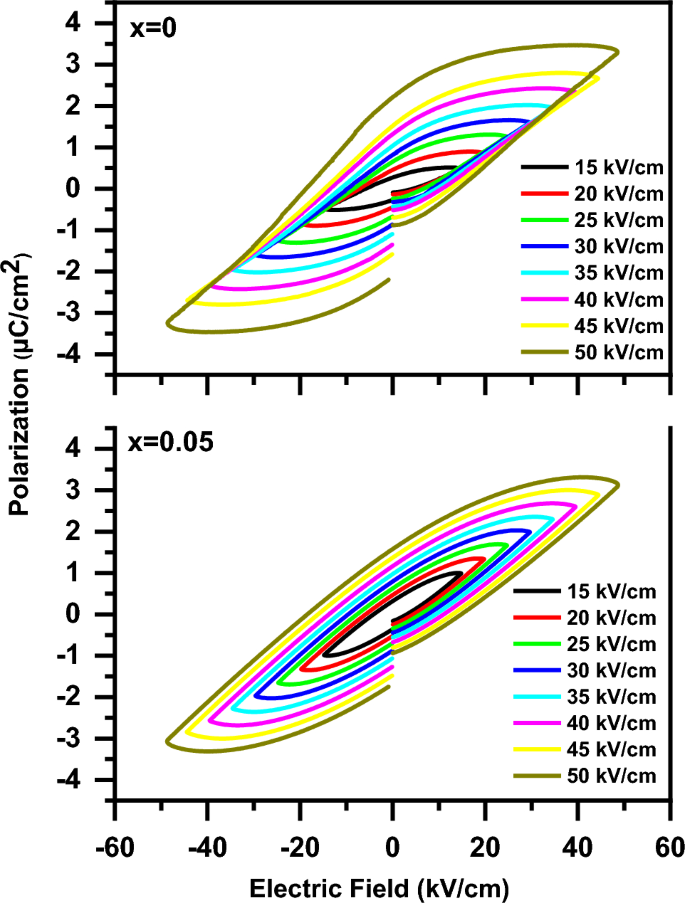
The polarization versus electric field (P–E) loops of the (1-x) BaTiO3-δ − xSrFeO3-δ samples at various electric field
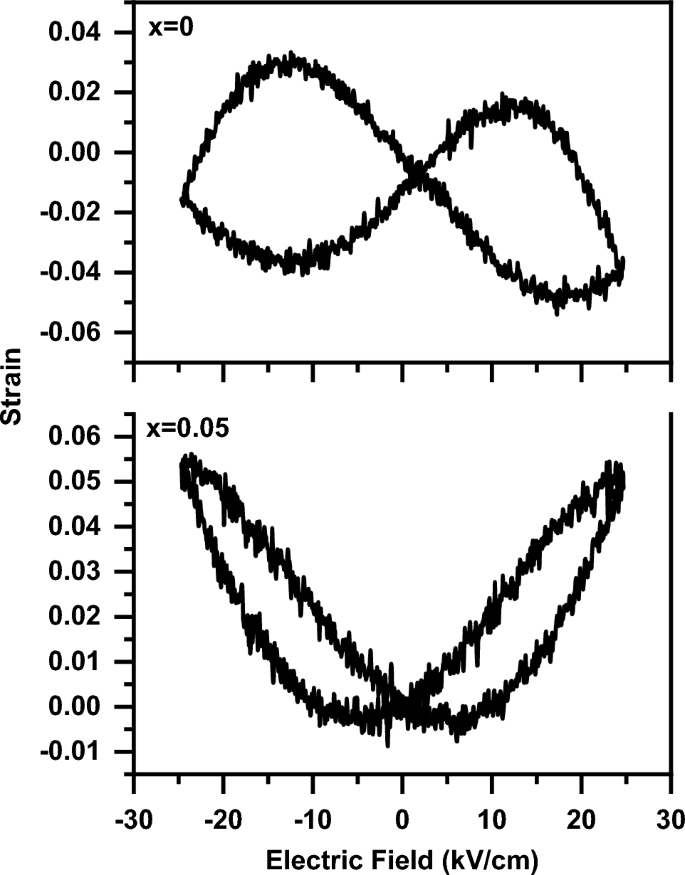
The bipolar strain versus electric field (S–E) loops of the (1-x) BaTiO3-δ − xSrFeO3-δ samples at electric field of 25 kV/cm
3.4 Pulse-echo response study of the 0.95 BaTiO3-δ − 0.05 SrFeO3-δ sample
The performance of the piezoceramic of the 0.95 BaTiO3-δ − 0.05 SrFeO3-δ sample to be acting as an active element for the ultrasonic transducer was evaluated by the pulse- echo response method [63,64,65,66,67]. Figure 16a shows the pulse-echo waveform of this sample which obtained on the Flaw detector screen (as shown Fig. 16b). This figure shows a short pulse duration time that means a relatively high damping. Recent studies reported that the thickness of piezoceramic element influenced the frequency of the transducer. Therefore, the decrease in the thickness will increase the frequency as well as the increase in the damping will decrease the amplitude [68, 69]. Owing to the high damping in our sample, the amplitude of echoes decreases and to overcome this obstacle the gain of echoes was increased. Further, the uncertainty of measurements was carried out on the frequency readings to obtain accurate value for the frequency. From Fig. 16a, it noticed that the measured nominal frequency of this sample is approximately 5 MHz with an expanded uncertainty of 0.04 under confidence level 95% (where the coverage factor k = 2).
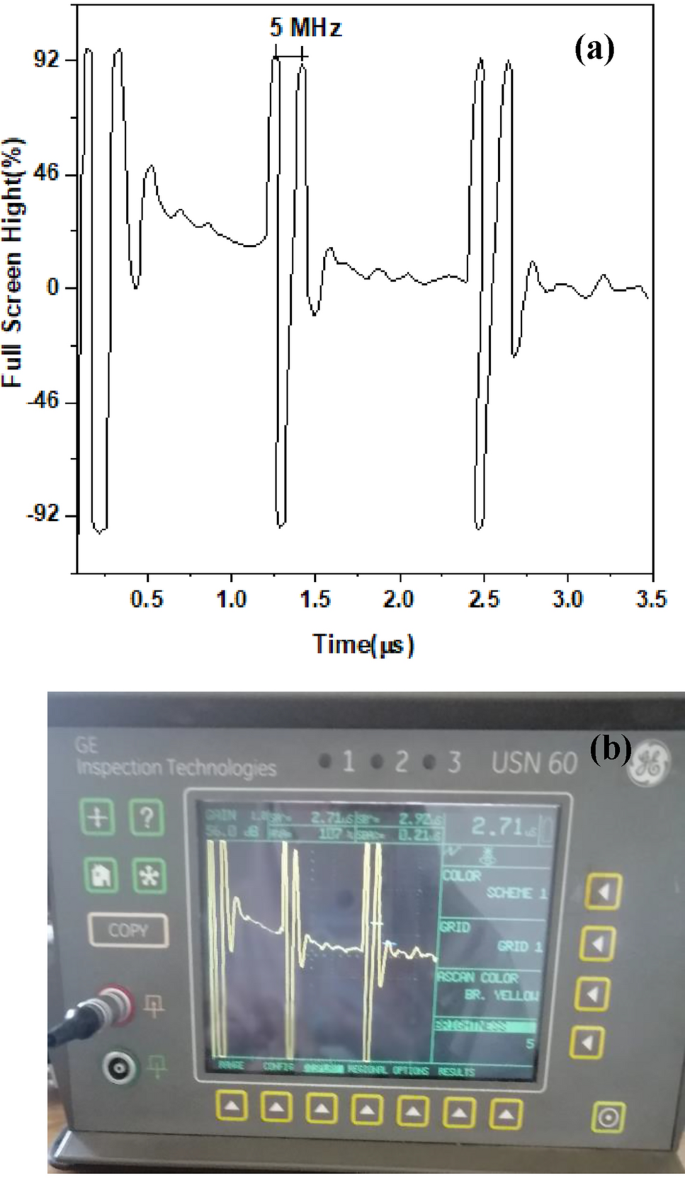
(a, b) The pulse-echo waveform of 0.95 BaTiO3-δ − 0.05 SrFeO3-δ sample and the Flaw detector screen, respectively
4 Conclusion
In summary, (1-x) BaTiO3-δ − xSrFeO3-δ (where x = 0 and 0.05) perovskite samples were successfully prepared. XRD analysis revealed the tetragonal phase (P4mm) of the present samples. Morphological investigation showed that the average particle size of BaTiO3-δ increases with introducing SrFeO3-δ (x = 0.05). The XPS results indicate that the presence of oxygen vacancies and mixed valence states of Ti at x = 0, beside mixed valence states of Fe ions at x = 0.05. DSC analysis of these samples confirmed the presence of the phase transitions of BaTiO3-δ nanoparticle. The optical properties of BaTiO3-δ were enhanced with introducing SrFeO3-δ (x = 0.05) as a result of the reduction of the band gap energies and the increase of the absorption coefficient in Vis. Range. The magnetic measurements elucidated an enhancement in the ferromagnetic ordering with introducing SrFeO3-δ (x = 0.05). The dielectric measurements showed an enhancement in the dielectric constant (εr) with introducing SrFeO3-δ (x = 0.05). The P-E and the bipolar S-E loops measurements elucidated an enhancement in the polarization and strain of BaTiO3-δ with introducing SrFeO3-δ (x = 0.05), respectively. The sample at x = 0.05 showed good ability to generate ultrasonic waves with nominal frequency of proximately 5 MHz. Our results pave a way to enhancement the properties of BaTiO3-δ by doping with SrFeO3-δ which makes it a promising material for optoelectronic, magneto-electric, electronic, electromechanical sensors, and ultrasonic transducers applications.
