Article Content
Abstract
I present two arguments concerning the metaphysics of the chemical bond—the interatomic state underlying the structure of matter—by examining its structural and energetic conceptions. First, I argue that these conceptions of the chemical bond are plagued by an inherent vagueness owing to their sorites-susceptibility. Second, I argue that such vagueness can be remedied through infinite-valued semantics via a compatibility function that assigns degrees of chemical bonding to any pair of atoms, depending on their spatial arrangement. I support these arguments through quantum-mechanical calculations of the archetypal hydrogen molecule.
Explore related subjects
Discover the latest articles and news from researchers in related subjects, suggested using machine learning.
- Chemical Bonding
- Density Functional Theory
- Hydrogen Bonding
- Philosophy of Chemistry
- Structure And Bonding
- Valence-Bond Theory
1 To Bond or Not to Bond?
The chemical bond is a ubiquitous interatomic interaction that shapes the structure and reactivity of matter at nanoscopic and macroscopic scales. The making and breaking of chemical bonds is a fundamental process at the heart of any chemical reaction, with implications extending from biology to physics. Notwithstanding the abundance of classical and quantum theories of the chemical bond that have flourished over the past century, its ontological status, if any, has remained enigmatic (Bhushan and Rosenfeld 2000; Needham 2014; Seifert 2024).Footnote1 This is largely due to the pragmatic stance adopted in the chemical sciences, which prioritizes the explanatory and predictive power of bonding theories over a sharp ontological understanding.
In the pursuit to elucidate the metaphysical nature of the chemical bond, Robin Findlay Hendry has introduced in the philosophical debate two now-classic accounts—the structural conception and the energetic conception (Hendry 2008). On the structural conception, chemical bonds constitute a material part of the molecular system and are regarded as spatially localized relationships between atomic centers. This underwrites the spirit of the ‘ball-and-stick’ models routinely deployed in rationalizing chemical phenomena. Michael Weisberg has further refined this view by identifying such interatomic relationship with the electron density (Weisberg 2008; Hettema 2008). Conversely, the energetic conception regards chemical bonds as the lowering in energy that occurs when a set of otherwise isolated atoms agglomerate. Although my aim is not to favor one conception over the other, it is important to emphasize that the energetic conception is more comprehensive than the structural conception due to its agnosticism toward distinctions such as intermolecular versus intramolecular interactions, focusing on the universal feature across the diverse varieties of chemical bonding, i.e., energy stabilization. As phrased by Hendry, “facts about chemical bonding are just facts about energy changes” (Hendry 2008, 919).
In essence, the structural view holds that the hallmark of the chemical bond is the presence of a finite electron density residing between atoms, while the energetic conception holds that the hallmark is a decrease in energy that a collection of atoms experiences upon aggregation. Both conceptions are generally interpreted as equally viable,Footnote2 with the former favored in (bio)chemistry and the latter in physics (Scerri 2023). At the operative level, each account sheds light on specific chemical aspects (Seifert 2023). For example, the structural view can aid a schematic understanding of reaction mechanisms in organic chemistry while the energetic view can facilitate the quantification of reaction barriers and ensuing reaction rates in chemical kinetics.
Altogether, the structural and energetic conceptions seek to assess whether or not a set of atoms is chemically bonded. By translating these conceptions on a quantitative basis, I devise a simple demarcation criterion to distinguish between bonded and non-bonded pairs of atoms. On the one hand, according to the structural conception, the indicator of the occurrence of a chemical bond is the electron density accumulating in the region between atoms A and B at a given interatomic distance r, . On the other hand, according to the energetic conception, such an indicator is the energy lowering that a set of atoms acquires through their aggregation. The latter can be quantified by the binding energy, , which compares the energy of the aggregate of atoms to the isolated atomic constituents as , where is the total energy of the aggregate AB at interatomic distance r, while and are the total energies of the isolated atoms. The demarcation criterion can be formulated as follows:
Structural conception. Given a pair of atoms A and B at an interatomic distance r, a chemical bond is (i) present if the interatomic electron density is positive, ; and (ii) absent otherwise, .
Energetic conception. Given a pair of atoms A and B at an interatomic distance r, a chemical bond is (i) present if the binding energy is negative, ; and (ii) absent otherwise, .
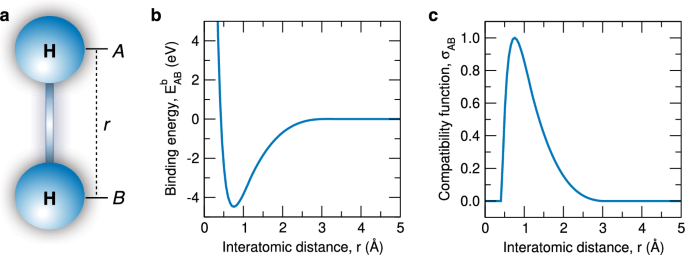
a, Ball-and-stick model of the hydrogen molecule, in which the two hydrogen atoms A and B are separated by the interatomic distance r. b, Evolution of the binding energy () of a pair of hydrogen atoms as a function of r, as obtained from quantum-mechanical calculations. c, Compatibility function of the chemical bond () between a pair of hydrogen atoms as a function r
To show the effectiveness of this demarcation criterion underpinning the structural and energetic conceptions of the chemical bond, I consider the representative example of a pair of hydrogen atoms at different interatomic distances r, as schematically depicted in Fig. 1a. By means of quantum-mechanical calculations,Footnote3 I examine two atomic configurations corresponding to interatomic distances either matching the equilibrium distance, Å, or a tenfold larger value, Å. Through the energetic conception, I obtain eV at Å and eV at Å. Through the structural conception, I obtain |e| at Å and |e| at Å, where Mulliken overlap populations are used as a proxy for the interatomic electron density. Hence, both conceptions consistently signal the presence of a chemical bond when the hydrogen atoms are located at their equilibrium distance, and the absence of a bond when the atoms are far apart, reflecting chemical intuition.
2 Vagueness of the Chemical Bond
By resorting to this demarcation criterion, I next argue that the notion of chemical bond is prone to soriticality and examine the resulting implications. The sorites paradox is a puzzle that underscores the tension arising between smooth changes and abrupt consequences (Hyde and Raffman 2018). The most celebrated version of the paradox is constructed as a recursive instance of modus ponens and centers on the constant decrease (increase) in the number of grains and the dissolution (formation) of a heap:
1,000,000,000 grains form a heap.
If 1,000,000,000 grains form a heap,
then 999,999,999 grains form a heap.
So 999,999,999 grains form a heap.
If 999,999,999 grains form a heap,
then 999,999,998 grains form a heap.
So 999,999,998 grains form a heap.
If
then
So 1 grain forms a heap.
The series can equivalently flow in the opposite direction,
1 grain does not form a heap.
If 1 grain does not form a heap,
then 2 grains do not form a heap.
So 2 grains do not form a heap.
If 2 grains do not form a heap,
then 3 grains do not form a heap.
So 3 grains do not form a heap.
If
then
So 1,000,000,000 grains do not form a heap.
This argument is paradoxical in that iterating flawless reasoning from flawless premises leads to flawed conclusions, viz., no number of grains is sufficiently large to form a heap, and no number of grains is sufficiently small to not form a heap. More generally, a sorites paradox can be contrived for any pair of predicates designating opposite states that are connected through a gradual variation of their ordering dimensions, with the latter being typically provided by real numbers. Examples include the predicates ‘tall’, ‘hot’, and ‘bald’ along with their respective ordering dimensions of ‘height’, ‘temperature’, and ‘number of hairs’.
In view of this, soritic reasoning can be applied to the predicate ‘chemical bond’ through the ordering dimension of ‘interatomic distance’. This can be exemplified in the case of a pair of hydrogen atoms. Both the energetic and structural conceptions hold that two hydrogen atoms form a chemical bond if their interatomic distance is 0.74 Å and do not form a chemical bond if their interatomic distance is increased to 7.40 Å. A soritic paradox employing the chemical bond can be constructed by altering the interatomic distance by an infinitesimally small amount:
Two hydrogen atoms at a distance of 0.740000 Å form a chemical bond.
If two hydrogen atoms at a distance of 0.740000 Å form a chemical bond,
then two hydrogen atoms at a distance of 0.740001 Å form a chemical bond.Footnote4
So two hydrogen atoms at a distance of 0.740001 Å form a chemical bond.
If two hydrogen atoms at a distance of 0.740001 Å form a chemical bond,
then two hydrogen atoms at a distance of 0.740002 Å form a chemical bond.Footnote5
So two hydrogen atoms at a distance of 0.740002 Å form a chemical bond.
If
then
So two hydrogen atoms at a distance of 7.40000 Å form a chemical bond.
The latter statement is false, as both the structural and energetic conception hold that a pair of hydrogen atoms at a distance of 7.40 Å does not form a chemical bond. In a similar vein, the argument can run in the opposite direction:
Two hydrogen atoms at a distance of 7.40000 Å do not form a chemical bond.
If two hydrogen atoms at a distance of 7.40000 Å do not form a chemical bond,
then two hydrogen atoms at a distance of 7.39999 Å do not form a chemical bond.Footnote6
So two hydrogen atoms at a distance of 7.39999 Å do not form a chemical bond.
If two hydrogen atoms at a distance of 7.39999 Å do not form a chemical bond,
then two hydrogen atoms at a distance of 7.39998 Å do not form a chemical bond.Footnote7
So two hydrogen atoms at a distance of 7.39998 Å do not form a chemical bond.
If
then
So two hydrogen atoms at a distance of 0.740000 Å do not form a chemical bond.
Likewise, the latter statement is false, as both the structural and energetic conception hold that a pair of hydrogen atoms at a distance of 0.74 Å does form a chemical bond. Hence, starting from true premises, according to which a chemical bond is present at the interatomic distance of 0.74 Å and absent at 7.40 Å, soritic reasoning yields the opposite conclusion that a chemical bond is present at the interatomic distance of 7.40 Å and absent at 0.74 Å. Although I have formulated the paradox for the case of a pair of hydrogen atoms, it is evident that such an argument is general, regardless of the nature of the chemical species, the interatomic distances, and whether a molecular or extended system is involved in devising the soritic series.
Owing to its susceptibility to the sorites paradox, I claim that the notion of chemical bond, as described by the structural and energetic conceptions, is vague (Sorensen 2023). The vagueness originates from the indeterminate character that spans over an ample range of interatomic distances. It is widely accepted that an intertwinement between vagueness and soriticality exists. The explicit connection between vagueness and soriticality traces back to Charles Sanders Peirce, according to whom “vagueness is an indeterminacy in the applications of an idea, as to how many grains of sand are required to make a heap, and the like” (Peirce 1892, 167). More recently, Otávio Bueno and Mark Colyvan have elaborated a general definition of the vagueness of a predicate that leverages its sorites-susceptibility, that is, “a predicate is vague just in case it can be employed to generate a sorites argument” (Bueno and Colyvan 2012, 29). A similar view is held by Kirk Ludwig and Greg Ray, who maintain that “a sorites argument is a symptom of the vagueness of the predicate with which it is constructed” (Ludwig and Ray 2002, 420).
Depending on the relationship between the predicate and the target entity in the reality that the predicate seeks to designate, it is customary to distinguish between epistemological, linguistic, and ontological taxonomies of vagueness (Pelletier 2017). Epistemological vagueness stems from the insufficient epistemological ability that prevents the adequate access to reality that would be necessary to dissolve the indeterminate character of the predicate. It is thus a mismatch between knowledge and reality. Linguistic vagueness results from the intrinsic limitations of language, that is, its inability to capture accurately the fine features of reality, prompting an indeterminacy on whether or not the predicate designates the target entity. It is thus a mismatch between language and reality. Finally, ontological vagueness is an inherent trait of reality, that is, the indeterminacy is not a consequence of the limitation of either knowledge or language, but is constitutive to the target entity that the predicate attempts to designate.
On the basis of this classification, I suggest that the vagueness affecting the chemical bond is primarily of a linguistic nature. As demonstrated by the sorites paradox, the indeterminacy of the chemical bond persists across the values of interatomic distances that neither match the equilibrium distance nor exceed the one at which any interaction (energy stabilization or interatomic electron density, depending on the conception considered) has ceased. That is, for an infinite number of intermediate cases, it is not possible to establish that the predicate (i.e., ‘chemical bond’) designates the target entity (i.e., the interatomic state). This perspective highlights the multifaceted nature of the chemical bond and calls for a reconsideration of its understanding beyond a binary classification in terms of presence and absence.
3 Fuzziness of the Chemical Bond
I propose many-valued logic as an effective strategy to remedy the challenges posed by the soriticality and vagueness of the chemical bond. At odds with classical Boolean logic, which dictates that a statement is either true or false, many-valued logic allows for truth-values that are intermediate between definite truth and definite falsehood (Sorensen 2023). The simplest form of many-valued logic is three-valued logic, according to which a statement can be true (e.g., ‘two hydrogen atoms at a distance of 0.74 Å form a chemical bond’), false (e.g., ‘two hydrogen atoms at a distance of 0.74 Å do not form a chemical bond’), or indeterminate (e.g., ‘two hydrogen atoms at a distance of 2.0 Å form a chemical bond’). This approach can be generalized by assigning degrees of truth to a statement via an infinite spectrum of truth-values quantified by real numbers ranging from 0 for definite falsehood to 1 for definite truth (Goguen 1969; Zadeh 1975). Infinite-valued, or fuzzy, logic is optimally suited to address soritic arguments, especially involving paradoxes that entail continuous ordering dimensions, such as the case of the evolution of interatomic distances bridging the limiting cases of presence and absence of the chemical bond. Fuzzy logic can be readily applied to vague predicates through fuzzy semantics (Hyde 2008; Varzi 2003). Analogously to the degree of truth employed in fuzzy logic, fuzzy semantics relies on the degree of membership, as quantified by the compatibility function (Zadeh 1975). The latter is obtained by mapping the vague predicate onto a continuum-valued interval ranging from 0 to 1. The closer to 1 is, the more the argument is a member of the set.
I advocate for the fuzzification of the structural and energetic accounts of the chemical bond to tame their vagueness. Specifically, I argue that, for any given pair of atoms, whether or not a chemical bond exists is a matter of degrees, leading to the following fuzzified conceptions of the chemical bond:
Structural conception. Given a pair of atoms A and B at an interatomic distance r, a chemical bond is (i) definitely present ( = 1) at the value of at which the interatomic electron density attains its maximum; (ii) definitely absent ( = 0) at values of r at which ; and (iii) partially present (0 < < 1) at all other values of r.
Energetic conception. Given a pair of atoms A and B at an interatomic distance r, a chemical bond is (i) definitely present ( = 1) at the value of at which the binding energy attains its minimum; (ii) definitely absent ( = 0) at values of r at which ; and (iii) partially present (0 < < 1) at all other values of r.
On the energetic conception of the chemical bond, a simple analytic expression for , which quantifies the degree of chemical bond between a pair of atoms A and B as a function of their interatomic distance r and resulting binding energy , can be introduced as,
where is the minimum value of binding energy attained at the equilibrium distance between atoms and is the step function,
A similar expression can be derived for the structural conception, based on instead of .
I illustrate this fuzzification through the example of the pair of hydrogen atoms. I determine of a pair of hydrogen atoms as a function of r through quantum-mechanical calculations. The result is shown in Fig. 1b. Three observations can be drawn. First, attains its minimum at = 0.74 Å, at which the chemical bond is definitely present ( = 1). Second, at 0.43 Å and 3.0 Å. In the latter cases, the chemical bond is definitely absent ( = 1). Third, at values of r spanning the intervals 0.43 Å < r < 0.74 Å, and 0.74 Å < r < 3.0 Å, the notion of chemical bond is fuzzy (0 < < 1). Combining the results given in Fig. 1b with the expression given in Eq. 1, in Fig. 1c I quantify the resulting for a pair of hydrogen atoms.
4 Conclusions
Understanding the metaphysics of the chemical bond, a vital interaction that governs the nature of matter, endures challenges despite a century of empirically successful theories. In this Essay, I have argued that the recently proposed structural and energetic conceptions of the chemical bond are prone to soriticality due to their inherent vagueness. I have shown that this vulnerability can be addressed through semantic fuzzification, advancing a simple compatibility function that quantifies the degree of chemical bonding between any pair of atoms. Using quantum-mechanical calculations, I have illustrated the strength of these arguments for the paradigmatic example of molecular hydrogen. While this work focuses on the hydrogen molecule as a simplified model, I envision this framework as a starting point for exploring more realistic bonding scenarios, including those impacted by factors such as temperature, pressure, environmental, and non-classical effects. Overall, the present Essay provides a multidisciplinary insight into the complex nature of the chemical bond, resonating with the famous quote of Robert Mulliken: “The chemical bond is not so simple as some people seem to think” (Weisberg 2008, 933).
Notes
-
For example, recent analyses have offered accounts of molecular systems in terms of Dennett’s real patterns (Seifert 2023; Dennett 1991) or processes in the framework of process ontology (Stein 2004; 2006).
-
There are, however, cases where the two conceptions conflict. For example, it is likely for a pair of atoms to retain a non-vanishing interatomic electron density (signaling the presence of a chemical bond, according to the structural conception) even at values of interatomic distances at which the atoms experience a repulsive interaction, with their energy upon association being larger than their isolated counterpart (signaling the absence of a chemical bond, according to the energetic conception). Additionally, electron-density difference maps obtained from X-ray crystallography have revealed that the accumulation of electron density in the internuclear region does not necessarily accompany bond formation (Dunitz and Seiler 1983), questioning the generality of the structural conception.
-
Quantum-mechanical calculations rely on spin-polarized density-functional theory, adopting the PBE gradient-corrected approximation to the exchange-correlation functional. Kohn-Sham orbitals are represented as a linear combination of atom-centered basis functions of double- plus single polarization quality in conjunction with a mesh cutoff of 400 Ry. Hydrogen atoms are hosted in a cubic box 25 Å-long to avoid spurious interactions between periodic replicas. Calculations are conducted with siesta (Soler et al. 2002).
-
Using quantum-mechanical calculations, I explicitly verified that increasing the distance between the two hydrogen atoms from 0.740000 Å to 0.740001 Å does not alter the binding energy and interatomic electron density.
-
Using quantum-mechanical calculations, I explicitly verified that increasing the distance between the two hydrogen atoms from 0.740001 Å to 0.740002 Å does not alter the binding energy and interatomic electron density.
-
Using quantum-mechanical calculations, I explicitly verified that decreasing the distance between the two hydrogen atoms from 7.40000 Å to 7.39999 Å does not alter the binding energy and interatomic electron density.
-
Using quantum-mechanical calculations, I explicitly verified that decreasing the distance between the two hydrogen atoms from 7.39999 Å to 7.39998 Å does not alter the binding energy and interatomic electron density.
References
-
Bhushan, N., and S. Rosenfeld. 2000. Of minds and molecules: new philosophical perspectives on chemistry. Oxford: Oxford University Press.
-
Bueno, O., and M. Colyvan. 2012. Just what is vagueness? Ratio 25 (1): 19–33.
-
Dennett, D.C. 1991. Real patterns. Journal of Philosophy 88 (1): 27–51.
-
Dunitz, J.D., and P. Seiler. 1983. The absence of bonding electron density in certain covalent bonds as revealed by x-ray analysis. Journal of the American Chemical Society 105 (24): 7056–7058.
-
Goguen, J.A. 1969. The logic of inexact concepts. Synthese 19 (3): 325–373.
-
Hendry, R.F. 2008. Two conceptions of the chemical bond. Philosophy of Science 75 (5): 909–920.
-
Hettema, H. 2008. A note on Michael Weisberg’s: challenges to the structural conception of chemical bonding. Foundations of Chemistry 10 (2): 135–142.
-
Hyde, D. 2008. Vagueness, logic and ontology. London: Routledge.
-
Hyde, D., and D. Raffman. 2018. Sorites paradox. In The stanford encyclopedia of philosophy, ed. E.N. Zalta. Metaphysics Research Lab: Stanford: Stanford University.
-
Ludwig, K., and G. Ray. 2002. Vagueness and the sorites paradox. Noûs 36 (s16): 419–461.
-
Needham, P. 2014. The source of chemical bonding. Studies in History and Philosophy of Science, Part A 45: 1–13.
-
Peirce, C.S. 1892. Review of Alfred Sidgwick’s distinction and criticism of belief. The Nation 27: 167.
-
Pelletier, F.J. 2017. The logic for metaphysical conceptions of vagueness. IfCoLog Journal of Logics and their Applications 4 (4): 1333–1350.
-
Scerri, E.R. 2023. A commentary on Weisberg’s critique of the ‘structural conception’ of chemical bonding. Foundations of Chemistry 25 (2): 253–264.
-
Seifert, V.A. 2023. The chemical bond is a real pattern. Philosophy of Science 90 (2): 269–287.
-
Seifert, V.A. 2024. Chemistry’s metaphysics. Elements in metaphysics. Cambridge: Cambridge University Press.
-
Soler, J.M., E. Artacho, J.D. Gale, et al. 2002. The SIESTA method for ab initio order-N materials simulation. Journal of Physics: Condensed Matter 14 (11): 2745.
-
Sorensen, R. 2023. Vagueness. In The stanford encyclopedia of philosophy, ed. E.N. Zalta and U. Nodelman. Metaphysics Research Lab: Stanford: Stanford University.
-
Stein, R.L. 2004. Towards a process philosophy of chemistry. HYLE – International Journal for Philosophy of Chemistry 10 (1): 5–22.
-
Stein, R.L. 2006. A process theory of enzyme catalytic power – the interplay of science and metaphysics. Foundations of Chemistry 8 (1): 3–29.
-
Varzi, A.C. 2003. Vagueness. In Encyclopedia of cognitive science, vol. 4, ed. L. Nadel, 459–464. Nature Publishing Group.
-
Weisberg, M. 2008. Challenges to the structural conception of chemical bonding. Philosophy of Science 75 (5): 932–946.
Ethics declarations
Conflict of interest
The author has no Conflict of interest to declare.
Additional information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Reprints and permissions
About this article
Cite this article
Pizzochero, M. The Fuzzy Logic of the Chemical Bond. J Gen Philos Sci (2025). https://doi.org/10.1007/s10838-024-09714-4
- Accepted
- Published
- DOI https://doi.org/10.1007/s10838-024-09714-4
Keywords
- Vagueness
- Fuzzy Logic
- Chemical Bond
- Quantum Chemistry
