Article Content
Introduction
Gout is a chronic inflammatory disease characterized by elevated serum urate (SU) levels, subsequent monosodium urate (MSU) crystal deposits, tophi, and gout flares [1, 2]. SU remains above 6.0 mg/dL in patients who either have oral urate-lowering therapy (ULT) underdosing or who are refractory to/intolerant of oral ULTs. This puts patients at risk for frequent flares (≥ 2 flares/year), tophi development/enlargement, and gout-related bone erosions [1, 3]. As a result, the American College of Rheumatology recommends that all gout patients be treated with oral ULTs in a treat-to-target SU < 6 mg/dL fashion [4]. Pegloticase is a recombinant PEGylated uricase that lowers SU in patients with uncontrolled gout who showed inefficacy, intolerance, or contraindication to oral urate-lowering treatments. In the pivotal MIRROR randomized controlled trial, the co-administration of methotrexate, a disease-modifying antirheumatic drug, with pegloticase attenuated anti-drug antibody formation, leading to higher response rates and markedly reduced infusion reaction risk compared with pegloticase + placebo [5,6,7]. Thus, methotrexate co-therapy can result in better treatment adherence by preventing premature pegloticase discontinuation due to loss of efficacy. However, further research is needed for monitoring and managing SU levels and optimizing pegloticase therapy duration [8].
With the increased use of pegloticase, dual-energy computed tomography (DECT) has been reliably used to image MSU deposition and subsequent bone erosions, showing a close physical relationship between MSU crystal deposits and bone erosions [9]. DECT imaging from pegloticase clinical trials demonstrated MSU depletion and bone erosion remodeling with pegloticase therapy [10, 11]. Diagnosis of gout via ultrasound imaging and DECT was included as a part of the 2015 collaborative guidelines by the American College of Rheumatology (ACR)/European League Against Rheumatism (EULAR) [12]. The role of both these imaging modalities has been further highlighted and confirmed in the 2018 EULAR evidence-based recommendations for the diagnosis of gout [13]. However, DECT is not widely available and is limited by cost; thus, better understanding gout abnormalities using more accessible imaging modalities such as the easily accessible and less expensive ultrasound would be of use. Ultrasound imaging has good specificity and sensitivity for detecting gout-related abnormalities, including synovitis, tophi, bone erosions, and the double-contour sign [14,15,16,17]. Additional advantages, including absence of radiation exposure and the ability to detect structural damage, inflammatory changes, and MSU deposits, have been highlighted in previous studies [18]. Further, comprehensive scanning protocols for the visualization and monitoring of MSU deposits for SU lowering and gout management have been published [18]. Here, for the first time, we characterized joint abnormalities on ultrasound in patients with severe uncontrolled gout, identified by pegloticase use. When available, ultrasound images during or after pegloticase treatment were used to preliminarily examine the ability of ultrasound to monitor indicators of treatment response not visible on physical exam.
Materials and methods
This was a retrospective deidentified medical record study reviewed and approved by the WIRB-Copernicus Group (WCG) international review board (IRB; Puyallup, WA). The study was assigned exempt status via WCG IRB, waiving the requirement of informed consent. The study does not include study-related procedures or examinations and was limited to the collection and analysis of deidentified standard-of-care data (including patient images) already present in the electronic medical record. The manuscript largely presents aggregate data of ten patients, and no individual patient’s identifying information is included. Hence, a consent to publish declaration was not applicable. All study conduct adhered to the tenets of the Declaration of Helsinki.
This retrospective observational study was conducted in a community clinic setting, outside of a research facility, to reflect real-world clinical practice. Therefore, elements of the Outcome Measures in Rheumatoid Arthritis Clinical Trials (OMERACT) guidelines were followed, but adherence to the full protocol cannot be guaranteed. Deidentified medical record data from patients who began pegloticase therapy between 2018 and 2022 and who underwent ultrasound joint imaging before treatment were included. Medical record and imaging data obtained through 1 May 2023 were included in analyses. All patients were seen at a single site and cared for by the same physician (B.F.). Pegloticase was administered according to the product label (8 mg infusion with a target interval of 2 weeks). Ultrasound exams were performed at the same clinical center with the same ultrasound equipment Sonosite M-Turbo (15 MHz transducer, FUJIFILM, Sonosite, Bothell, WA, USA). Images were obtained in two perpendicular planes. All scans were performed by the same experienced sonographer for consistency. A standardized ultrasound imaging reporting template was completed by the technologist performing the scan. Imaging regions were performed on the basis of standard-of-care practices of a single community rheumatologist (B.F.) and included the ankles, feet, hands, and/or wrists.
An independent third party (The Lockwood Group, Stamford, CT, USA) retrospectively collected the deidentified patient data, including patient characteristics, gout characteristics, available laboratory values, and ultrasound reports. Available ultrasound images (anonymous and deidentified) were also obtained, including static and cine loops for the body parts scanned.
Data analysis
Patient and clinical characteristics were examined. Ultrasound imaging studies were interpreted according to a standard reporting template and included tophi, bone erosions, joint effusions, extra-articular deposits, synovitis, and double-contour signs. Findings of this retrospective, observational study are presented using mean (± SD) for continuous variables and n (%) for categorical parameters.
Results
Patients
A total of ten patients (70% male; age: 65.2 ± 15.3 years; body mass index [BMI]: 28.6 ± 4.9 kg/m2) met study criteria and were included. Patients had a gout duration of 7.8 ± 7.5 years, 90% had visible tophi, and mean SU was 7.2 ± 2.6 mg/dL [2.9–11.1 mg/dL] (Table 1). Patients had received an average of eight infusions over a mean of 17.0 ± 10.2 weeks. Following pegloticase discontinuation, eight patients (80.0%) began allopurinol (highest dose: 600 mg/day; initiated 15.4 ± 14.7 weeks after last pegloticase dose).
Gout-related imaging findings
Pretreatment ultrasound images revealed ≥ 1 gout-related bone and/or soft tissue abnormality in nine of ten patients (90%). The remaining patient had an amputation at the first metatarsophalangeal (MTP) joint of the imaged foot. In the nine patients with detected abnormalities, these included tophi (100%), bone erosions (67%), joint effusion (44%), synovitis (44%), and double-contour sign (33%; Fig. 1). A median of three types of ultrasound abnormalities (range 2–5) per image were noted.
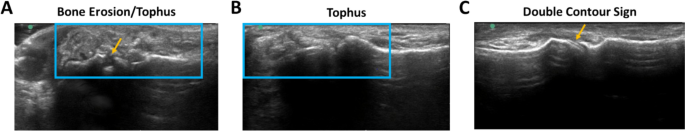
Ultrasound images with gout-related abnormalities in patients with uncontrolled gout. A Bone erosion (yellow arrow) and tophus (blue box) were observed in the right first MTP. B A tophus was visible in the left first MTP. C A double-contour sign was apparent in the right first MTP. MTP metatarsophalangeal
Imaging findings with pegloticase treatment
Two patients had follow-up imaging of the same joint after pegloticase initiation. Patient 1 was a 55-year-old male patient with a 20-year history of gout, visible tophi, and allopurinol inefficacy. Prior to pegloticase treatment, SU was 6.3 mg/dL, and palpable lumps/tophi were noted on physical examination on the right olecranon, right first MTP, and left Achilles tendon. Prior to initiating treatment, ultrasound imaging of the feet revealed tophi (bilateral MTP), double-contour sign (right metatarsal head), and a bone erosion (right first MTP, medial aspect). A large hyperechoic deposit was also identified in the soft tissues overlying the left Achilles tendon, along with a bone spur at the right Achilles tendon insertion. The patient received a total of four pegloticase infusions over 16 weeks (methotrexate co-therapy initiated 28 days after first infusion). The patient had a treatment interruption after infusion 2, with an infusion spacing of five and nine weeks for infusions 3 and 4, respectively. Then, 11 days following pegloticase discontinuation (oral ULT had not been re-initiated), repeat ultrasound revealed resolution of the double-contour sign (Fig. 2A) with persistence of all other ultrasound abnormalities. MTP, metatarsophalangeal, ULT, urate-lowering therapy.
Patient 2 was a 65-year-old male patient who had been diagnosed with gout approximately 1 year prior. The patient had visible tophi, allopurinol inefficacy, and a pretreatment SU of 8.4 mg/dL. Prior to initiating pegloticase, ultrasound showed a left tibiotalar joint effusion, bilateral first MTP tophi, and a right first MTP bone erosion. The patient received 18 pegloticase infusions over 40 weeks (methotrexate co-therapy initiated 7 days before first infusion), maintaining SU < 1.5 mg/dL for the full treatment duration. Repeat ultrasound at treatment week 26 (11 infusions received) revealed bilateral first MTP tophi improvement (resolution or size decrease; Fig. 2B).
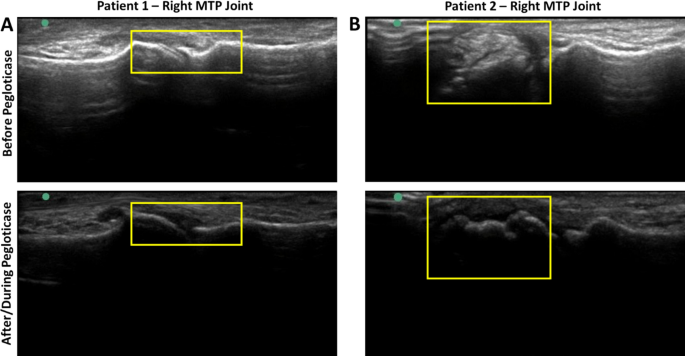
A Serial ultrasound imaging in a patient who received four pegloticase infusions but prematurely discontinued treatment due to SU rise. Pretreatment ultrasound imaging of the feet showed tophi (bilateral MTP joints), a double-contour sign (right metatarsal head), and a bone erosion (right first MTP, medial aspect). Following pegloticase discontinuation, the double-contour sign was no longer apparent. B Serial ultrasound imaging in a patient who received 18 pegloticase infusions. SU was maintained < 1.5 mg/dL during the 40-week treatment course. Pretreatment ultrasound imaging of the feet showed tophi (both MTP joints) and bone erosions (right first MTP adjacent to tophus). At treatment week 26, repeat ultrasound of the feet revealed marked decrease in tophi size. Boxes indicate areas of focus. MTP metatarsophalangeal
Discussion
The utility of ultrasonography in diagnosing and monitoring gout has been previously reported in literature [19,20,21,22,23,24,25]. DECT and ultrasound provide distinct signatures that can be used for gout diagnosis, including tophus detection using DECT, and tophus, double-contour sign, and aggregate detection using ultrasound; however, how these abnormalities represent changes in MSU deposits and their exact relevance in follow-up of MSU deposits is not clear. A prior study showed that changes in DECT and ultrasound gout lesions (e.g., double-contour sign resolution, tophus volume reduction) after 6 months of conventional ULT can be correlated with lowering of SU levels [24]. In the light of these previous studies, the intent of the current study design was to evaluate real-world improvement in joint crystal burden with pegloticase treatment, using tools and conditions that mirror standard community practice. To that effect, ultrasound imaging was used in this retrospective observational study to characterize joint and bone abnormalities in patients with uncontrolled gout. When possible, matched before/after treatment images were also examined to monitor the response of gout-affected joints. Pre-therapy ultrasound revealed the presence of ≥ 1 ultrasound abnormality in 90% of patients, including tophi, double-contour sign, synovitis, joint effusion, and/or bone erosions. These findings suggest that better understanding of gout-related changes on ultrasound before and after treatment could be important for overall clinical assessment, monitoring of SU-lowering treatment effects, and making therapy decisions such as optimal response and length of therapy.
Two patients who had matched follow-up imaging during/after pegloticase treatment showed improvement in ≥ 1 ultrasound abnormality. One patient had resolution of a double-contour sign overlying the right metatarsal head after receiving four pegloticase infusions over 16 weeks (tophi and bone erosions persisted). The other patient had marked bilateral reduction in tophi size at the first MTPs (with possible resolution) after intensive SU lowering (< 1.5 mg/dL) at 26 weeks (11 pegloticase infusions received). These findings are in agreement with prior DECT imaging studies, showing an initial deposited MSU volume reduction (approximately 15%–20% at 4 weeks) and approximately 90% depletion by week 24 of therapy [10, 11]. Those studies, along with a prior DECT study during allopurinol treatment [26], have shown that DECT imaging can be a useful tool in diagnosing gout and monitoring therapeutic response to SU-lowering treatment.
The limitations of the current study include its smaller sample size and retrospective data collection. In particular, varying timing of serial images and pegloticase therapy duration (which was based on patient clinical characteristics) prevented systematic analysis. Further, although this community practice has standardized ultrasound protocols, a single sonographer and a single reader analyzed the ultrasound images. As a result, the findings of the current study are preliminary, and a more systematic study on a larger number of patients with uncontrolled gout is needed.
The preliminary findings of the current study further support the use of ultrasound in gout as a readily available and economical imaging modality. To our knowledge, this is the first study to demonstrate the utility of ultrasound for monitoring pegloticase treatment response in patients with uncontrolled gout, who typically have intolerance of or are refractory to oral ULTs. It is important to note that this study was conducted in a community clinic setting, outside of a research facility, to reflect real-world clinical practice. The ultrasound images are representative of the imaging typically performed in the majority of rheumatology practices where pegloticase is most often administered, which enhances the generalizability and translational value of the findings.
The current study suggests that ultrasound may be an effective imaging modality to monitor tophus and other joint abnormalities during pegloticase treatment. Because optimal pegloticase treatment duration has not yet been established but may be related to tophus resolution [7], ultrasound could be a useful tool for guiding treatment duration in a typical clinical setting. With increased use of pegloticase and increased response rates due to methotrexate co-administration, ultrasound can be an effective and simpler imaging modality to monitor treatment in clinical settings where DECT or other comprehensive assessments may not be available. Further, our findings agree with a prior study that also observed double-contour sign resolution with oral ULT-induced normalization of urate levels (allopurinol [n = 3], probenecid [n = 1]) [27]. Of note, the present study showed double-contour sign resolution within weeks of initiating pegloticase therapy versus several months with oral ULT [27].
Conclusion
In conclusion, this small, retrospective study shows that ultrasound is a useful and readily accessible diagnostic imaging tool that can detect abnormalities associated with uncontrolled gout, including tophi, double-contour signs, and bone erosions. Of note, this study adds evidence that ultrasound may also be useful for monitoring treatment response to both oral and biologic ULTs. Although further study is needed in a larger number of patients, ultrasound imaging seems to be a useful tool for monitoring MSU crystal deposits, which could help inform therapy decisions.
