Article Content
Abstract
Purpose
We quantified variation in the uptake of sodium-glucose co-transporter 2 inhibitors (SGLT2i) and glucagon-like peptide-1 receptor analogues (GLP-1RA) across sociodemographic, behavioural and clinical characteristics of people with type 2 diabetes (T2D) at high cardiovascular risk.
Methods
We used the 45 and Up Study survey data (2018–2020) linked to dispensing and service claims for 10,171 people with T2D (56% male, median age of 72 years, median diabetes duration of 11 years). We calculated the prevalence of GLP-1RA and SGLT2i use within 1 year and used logistic regressions to assess associations with each participant characteristic.
Results
We found that 2270 (22.3%) people with T2D used SGLT2i and 679 (6.7%) used GLP-1RA. Use of these medicines was higher in people diagnosed with diabetes for a longer period, a high number of comorbidities and survey year, decreased in older people, and varied by sex. After adjusting for these factors, utilisation of these medicines was lower among people who consume alcohol (versus non-drinkers) and higher among those with overweight or obesity. SGLT2i use was also higher in people who were less physically active or had established cardiovascular disease and lower in people with anxiety or depression. GLP-1RA use was higher among people with poorer health and lower in people born outside Australia/New Zealand.
Conclusion
Prevalent use of SGLT2i and GLP-1RA was suboptimal and varied across clinical characteristics and behavioural risk factors. While some variation reflects complexities in prescribing for this older population, there remains opportunity for optimised prescribing within this high-risk population.
Introduction
In the last decade, we have learnt of the cardiorenal benefits of sodium-glucose cotransporter 2 inhibitors (SGLT2i) and glucagon-like peptide-1 receptor agonists (GLP-1RA) in people with type 2 diabetes (T2D). These medicines have shifted the focus of T2D management to early therapy intensification to reduce risk of cardiovascular disease (CVD), cardiovascular events and chronic kidney disease, irrespective of improvements in glycemia [1, 2]. SGLT2i and GLP1-RA are now recommended for use in all people with T2D with atherosclerotic CVD or at high risk of CVD, heart failure or chronic kidney disease, irrespective of glycated haemoglobin (HbA1c) or metformin use [3].
Despite their clinical benefits, SGLT2i and GLP-1RA are being underutilised among people with T2D [4,5,6,7]. Identifying populations with suboptimal use of these medicines is key to informing promotion strategies and ensuring optimal uptake — particularly among high-risk individuals who would benefit the most from treatment. For example, older populations with established CVD for which SGLT2i or GLP1-RA medicines have multiple benefits in reducing both the risk of CVD events and chronic kidney disease while reducing other risk factors such as body fat mass, blood pressure and improving lipid profiles [1]. Studies have found uptake differences by sex [5, 6], socioeconomic status [5,6,7] and geographic remoteness [6, 8]. However, there are many barriers to optimal medicine use, including other sociodemographic, behavioural and clinical factors influencing prescribing and medicine use [9, 10]- for example, clinicians might be hesitant to prescribe for people with increased risk of harms (e.g. alcohol users [11], people with comorbidities[12]). There is limited understanding of how SGLT2i or GLP-1RA use varies across these characteristics, because most prior studies examining the uptake of these medicines have used clinical registries or claims-based datasets which lack information on detailed personal socioeconomic, behavioural and lifestyle factors [6, 7].
Therefore, we leveraged survey data linked to dispensing and service claims data to estimate the prevalence of uptake of SGLT2i and GLP-1RA across a broad range of demographic, socioeconomic, lifestyle and clinical characteristics of people living with T2D at high risk of CVD.
Methods
Setting and data source
New South Wales (NSW) is Australia’s most populous state, with approximately 8 million population. The Sax Institute’s 45 and Up Study is a population-based cohort study of NSW residents aged 45 years and over. Participants were randomly sampled from Australia’s universal healthcare system (Services Australia Medicare enrolment database), with an oversampling of people aged 80 + years and residents of rural and remote areas. A total of 267,357 people participated in the study, responding to a baseline survey (2005–2009) and two follow-up surveys (2012–2015 and 2018–2020) and provided consent for their data to be used for research. This corresponded to approximately 11% of NSW population aged 45 years and over (response rate: 19%). Survey data was linked to national dispensing claims from the Pharmaceutical Benefits Scheme (PBS) and to service claims from the Medicare Benefits Schedule (MBS) supplied by Services Australia. Further details of the 45 and Up Study, including participant recruitment, consent and data collection, are described elsewhere [13, 14].
Medicines of interest
Within Australia’s universal healthcare system, citizens and permanent residents are entitled to subsidised medicines through the PBS. PBS data captures information on PBS-listed medicines dispensed in community pharmacies, private hospitals and on discharge from public hospitals in most states (NSW excluded) [15]. In this study, we examined receipts of SGTL2i and GLP-1RA that are available via the PBS. SGLT2i currently PBS-listed including dapagliflozin, empagliflozin and ertugliflozin and GLP-1RA PBS-listed include exenatide, dulaglutide and semaglutide. During the study period, SGLT2i were subsidised on the PBS for the treatment of T2D if a patient had a glycated haemoglobin (HbA1c) measurement of > 7% (53 mmol/mol) despite treatment with metformin. GLP-1 RA were subsidised on the PBS for the treatment of T2D as add-on therapy to metformin if a patient had a HbA1c > 7% (53 mmol/mol) and had a contraindication to or was intolerant of a sulfonylurea (Supplementary Tables 1–2) [16, 17].
Study sample
Of the 97,302 respondents of the 45 and Up Study Wave 3 survey, data from 97,200 were available for this research. Using survey responses and algorithms to ascertain T2D [4, 18, 19], we identified 11,195 people self-reporting diabetes (Has a doctor ever told you that you have diabetes? Answer options: type 1 diabetes (T1D), T2D or unknown, gestational diabetes or not informed), of whom 9900 self-reported having T2D. We then examined PBS dispensing of oral anti-hyperglycaemic agents (OHA) and insulins [4] (Supplementary Table 2) to further identify people with T2D who may have mistakenly self-reported having T1D. We defined T2D among people self-reporting T1D if people had been dispensed OHA but not insulin in the year before the survey (Fig. 1 and Supplementary Fig. 1). Finally, we identified 10,171 people with T2D.
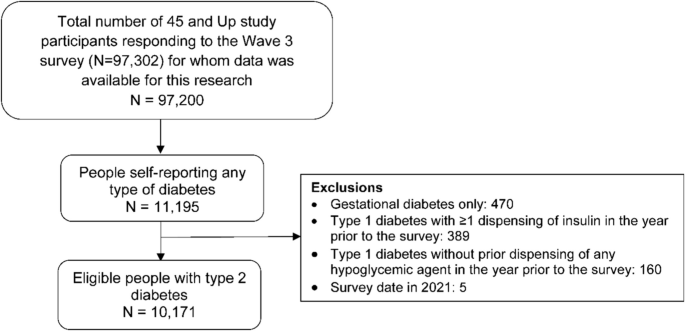
Diagram of study population selection
Study design
We used a cross-sectional study design (Supplementary Fig. 2) to estimate annual rates of prevalent use of medicines among people with T2D according to survey year (2018–2020).
Outcome measures
We considered people receiving the medicines if they had a dispensing of SGLT2i or GLP-1RA any time from the survey date to 365 days. We presented annual prevalence by demographic, socioeconomic, behavioural, clinical and medicine-related characteristics.
Covariates
Covariates included survey variables from the second follow-up survey (2018–2020) and baseline survey and variables derived from the administrative datasets (Supplementary Tables 2, 3, 4 and 5). We classified covariates according to the following participant characteristics: sociodemographic (e.g. sex, age group, country of birth, language spoken at home, highest education qualification, region of residence, socioeconomic status), behavioural (e.g. alcohol drinking status, smoking status, physical activity status [defined as moderate to vigorous physical activity (MVPA) [20]]), clinical (e.g. diabetes duration, number of comorbidities [based on medicines dispensed in the prior year [21]], CVD risk [based on age, smoking status, high blood pressure and high cholesterol [22]], established CVD based on self-report, procedures performed and medicines dispensed in the prior year [22], heart failure, depression or anxiety, overweight or obese) and medicine-related (e.g. other anti-hyperglycaemic agent dispensed in the prior year).
Statistical analysis
We measured use of SGLT2i and GLP-1RA separately, with people dispensed medicines from both classes contributing to both analyses. We conducted descriptive analyses of included participants and characteristics of those dispensed SGLT2i and GLP-1RA by sociodemographic, behavioural, clinical and medicine-related characteristics using proportions for categorical variables and median values and interquartile range (IQR) for quantitative variables.
We stratified annual prevalence by interested covariates and presented as proportions of selected subgroups. For example, we calculated annual prevalence of GLP-1RA use among females as the number of females receiving GLP-1RA by the total number of females with T2D and presented as percentages. We then performed a series of multivariable logistic regressions to test associations of uptake of SGLT2i and GLP-1RA for each participant characteristic. Given a causal framework to inform model selection in this topic is not available and many sociodemographic and clinical variables may be correlated, we fit separate models for each variable adjusted by key factors influencing utilisation. First models were adjusted by age, sex and survey year (Stage 1). Second models were further adjusted for clinical complexity, using participant’s self-reported duration of diabetes and number of comorbidities (Rx-Risk Comorbidity Index[21], measured on medicines dispensed in the year prior to survey) (Stage 2). Therefore, we fit two separate adjusted models for each variable of interest. Missing values were excluded from the regression models.
Results
Cohort characteristics
Our study cohort was older (median age 72 years [IQR: 66, 78]), with a large proportion aged 65–74 years (41.6%). Over half were male, approximately three-quarters were born in Australia or New Zealand and less than 10% spoke languages other than English at home. Participants were highly educated (57% with higher education degree), and half lived in major cities.
Approximately half self-reported having T2D for 11 years or more. Most people had three or more comorbidities (86%), had a high CVD risk profile (96%), had overweight or obesity (76%) and one third had established CVD. Moreover, over half self-reported drinking every week, and over a third did not perform 150 min or more of moderate to vigorous physical activity per week.
Most people had a PBS-dispensing of metformin in the year prior to the survey (71.5%), with variable rates of use of other anti-hyperglycaemic agents. More details are shown in Table 1.
Annual prevalence of SGLT2i use
A total of 2270 (22,3%) people with T2D were dispensed any SGLT2i within 1 year from the survey, most of which (75.2%) also had an SGLT2i dispensing in the year prior. SGLT2i utilisation was higher among males than females (25.4% vs 18.4%, p value < 0.001) and lower with increasing age (from 30.2% in people aged 55–64 years to 5.8% among those aged ≥ 85 years, p value < 0.001). Use was higher in more recent survey years (from 19.4% in 2018 to 24.0% in 2020, p value < 0.001).
After adjusting for these key sociodemographic characteristics (age, sex and survey year; Fig. 2, stage 1), SGLT2i use was higher in people living in most disadvantaged areas than least disadvantaged areas. Regarding behavioural characteristics, SGLT2i use was lower in alcohol drinkers than non-drinkers and less physically active people than physically active people. Regarding clinical characteristics, SGLT2i use was higher with longer diabetes duration and more comorbidities. Use was also higher in people who are overweight or obese and people who had established CVD and self-reported fair/poor health. Use was lower among people who self-reported anxiety or depression.
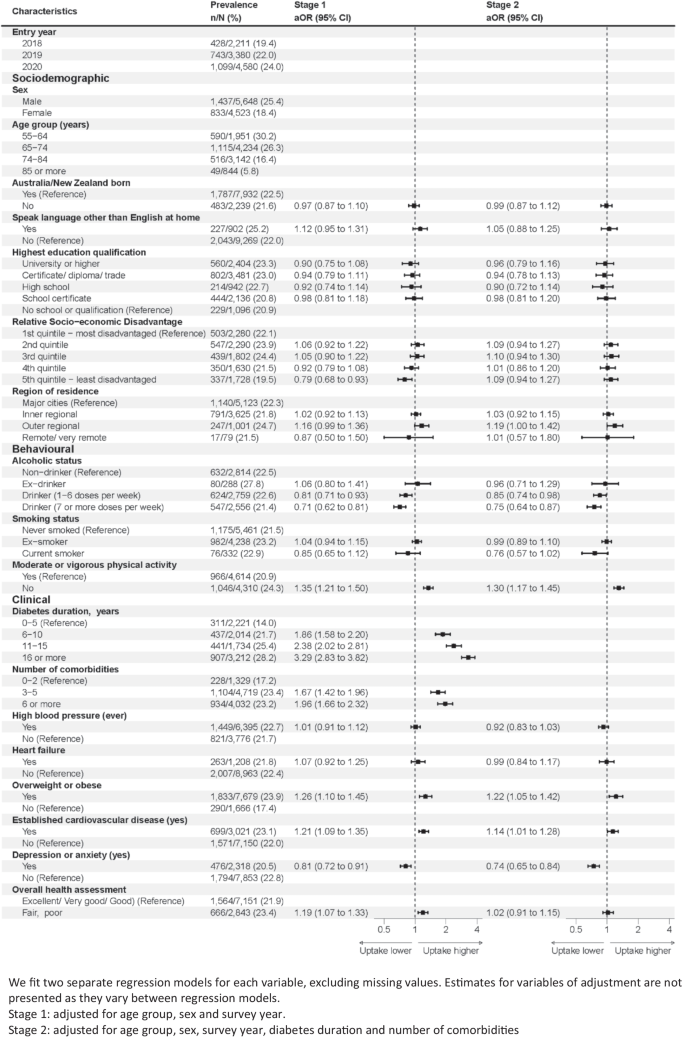
Annual prevalence of SGLT2i use and adjusted odds ratios (aOR) by participant characteristics (2018–2021)
After further adjusting for key markers of clinical complexity (number of comorbidities and diabetes duration; Fig. 2, stage 2), there was no statistically significant difference in utilisation of SGLT2i by socioeconomic status. However, use remained lower in alcohol drinkers, less physically active people and people who self-reported anxiety or depression. It remained higher in people who were overweight or obese and people who had established CVD.
Annual prevalence of GLP-1RA use
A total of 679 (6.7%) people with T2D were dispensed GLP-1RA within 1 year from the survey, most of whom (n = 425) also had a GLP-1RA dispensing in the year prior. GLP-1RA utilisation was higher among females than males (7.8% vs 5.8%, p value < 0.001) and was lower with increasing age (from 9.6% in people aged 55–64 years to 1.8% among those aged ≥ 85 years, p value < 0.001). Utilisation was higher in more recent survey years (from 5.1% in 2018 to 8.2% in 2020, p value < 0.001).
After adjusting for these key sociodemographic characteristics (Fig. 3, stage 1), use of GLP-1RA was lower in people born outside of Australia or New Zealand (than people born within these countries), as well as in people living in outer regional areas than people living in major cities. Regarding behavioural characteristics, use of GLP-1RA was lower in drinkers with higher levels of alcohol consumption and higher among ex-smokers than never smokers. Regarding clinical characteristics, use of GLP-1RA was higher with longer diabetes duration and more comorbidities. It was also higher in people who self-reported having high blood pressure, people who were overweight or obese or had reported fair or poor health.
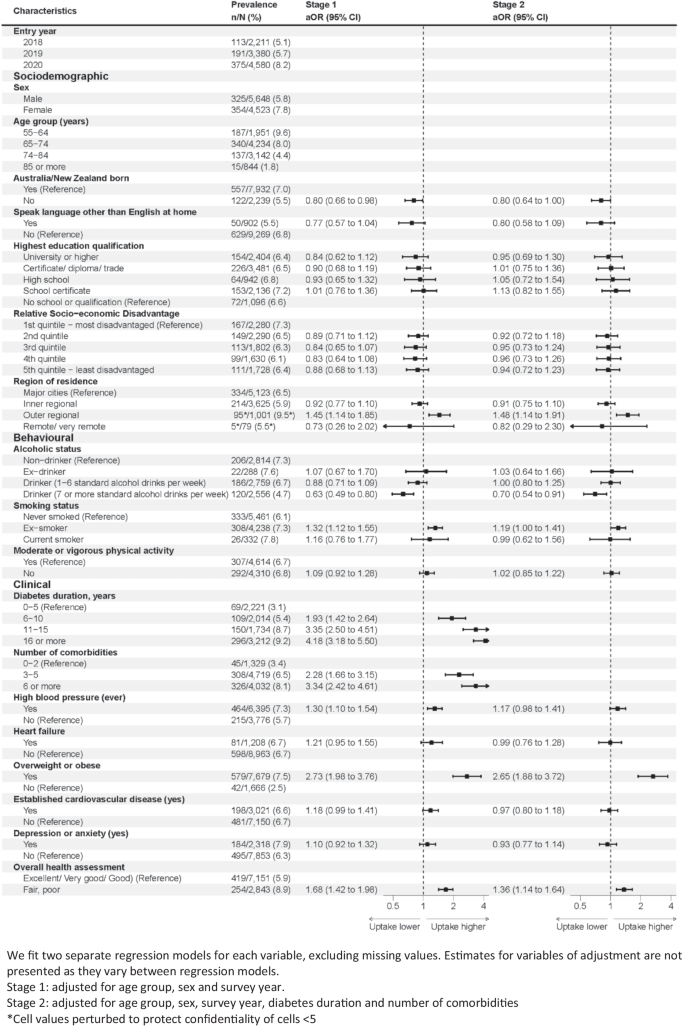
Annual prevalence of GLP-1RA use and adjusted odds ratios (aOR) by participant characteristics (2018–2021)
After further adjusting for key markers of clinical complexity (Fig. 3, stage 2), there were few statistically significant associations. Use of GLP-1RA was marginally lower in people born in countries other than Australia or New Zealand. Use was also higher in people living in outer regional areas than people living in major cities, people who were overweight/obese and people with fair/poor health. Use was lower in people with higher levels of alcohol consumption.
Discussion
We found uptake of SGLT2i and GLP-1RA was suboptimal among older adults living with T2D in NSW, with less than one quarter (22.3%) or people using an SGLT2i, and 1 in 15 (6.7%) people using a GLP-1RA. Given the high risk of cardiovascular events often experienced in older populations, continued investigation into health system barriers for prescribing and access is needed to ensure optimal use of these medicines in people who would benefit the most. Encouragingly, we found utilisation rates increased in more recent years and did not observe variation in uptake by sociodemographic factors. While we also found higher levels of use among people with clinical factors which aligned with need for therapy—such as people with more comorbidities, cardiovascular risk factors or behaviours (such as insufficient physical activity) and longer diabetes duration—the use of these medicines was still low within these population groups.
Suboptimal use in the Australian context
Multiple studies have demonstrated increasing use of SGLT2i and GLP-1RA among people with T2D in Australia and internationally, particularly after the cardio-renal benefits of these medicines have been increasingly evident in the last decade.[4, 5, 23,24,25,26]. However, given the potential benefits, overall utilisation remains low, with less than one in four people living with T2D receiving these medicines [SGLT2i (~ 20%) and GLP-1RA (~ 6%) as of 2019] [5, 24]. This is consistent with other population-level studies finding low use among older populations [8].
With regard to the clinical appropriateness of the use of these medicines, a Swedish study based on national registry data estimated around 50–80% of people with T2D would be eligible to receive either SGLT2i or GLP1-RA based on 2019 clinical guideline recommendations [27]. While our study population was primarily older, and we could not assess eligibility to receive these medicines, the high levels of cardiovascular risk factors and CVD indicate that a large proportion of our study population could benefit from receiving SGLT2i or GLP-1RA—and hence this was a potential missed opportunity. During the observation period of our study, these medicines were recommended as preferred add-on therapies to prevent further risk of progression and cardiovascular and renal complication in both international (2019) and local guidelines (2020) [27, 28].
During the study period, the PBS required people to meet specific criteria for subsidy of SGLT2i or GLP1-RA, such as use in combination with metformin and having a HbA1c ≥ 7% despite present treatment. Data from a clinical audit in Australia found two in three patients with T2D had an HbA1c ≥ 7% and 73% were using metformin. Thus, approximately 70% of people with T2D would be eligible for second-line therapy according to PBS reimbursement criteria required during the observation period of our study [29]. Taken altogether, use of SGLT2i and GLP-1RA was suboptimal even if we accounted for clinical and PBS reimbursement indications.
Use according to behavioural and clinical factors
Despite the low use across the board, we found behavioural and clinical variation in the use of SGLT2i and GLP1-RA aligned with evidence-based practice, with targeted use in specific populations most likely to benefit from these medicines. For example, we observed higher utilisation among people who were overweight or obese, as well as people with low physical activity, aligning with the weight loss benefits of these medicines, particularly for GLP1-RA [5, 25]. We also found lower utilisation among alcohol drinkers, which might be due to concern about an increased risk of the rare serious side effect of SGLT2i diabetic ketoacidosis [30] and a potentially higher risk of pancreatitis with GLP1-RA. Interestingly, GLP-1 RA is being explored as a potential treatment for alcohol use disorders [31].
We also found higher utilisation of SGLT2is in people with established CVD, diverging from previous research in Australia, suggesting a non-selective uptake by the presence of CVD [5, 26]. This might reflect the fact we analysed more recent data and that this is a rapidly evolving area. There is increasing prescriber awareness of the use of SGLT2i for preventing heart failure hospitalisations and kidney disease reflecting recent guideline recommendations from diabetes and cardiac societies [32, 33]. Similarly, the higher association with number of comorbidities and diabetes duration may reflect increased prescribing in high-risk populations for such secondary prevention. However, we also found lower rates of SGLT2i use among people reporting anxiety or depression. The reason for this difference is unclear.
Use according to sociodemographic factors
Similar to other studies [34, 35], we found lower use of SGLT2i and GLP1-RA therapies with increasing age which may reflect prescriber concern about side effects in the older age population. This is despite increased prevalence of cardiorenal disease in this population and other treatments for T2D having potentially serious risks, for example sulfonylureas and hypoglycaemia.
Although we did not find statistically significant differences in utilisation across a range of other sociodemographic factors, we did observe some differences indicative of potential disparities. For example, we found lower use of GLP-1RA in people born outside of Australia and New Zealand. A previous study in Australia reported general practitioners in training to prescribe GLP-1RA or SGLT2i less often to people from non-English-speaking background [36], possibly due to communication challenges between prescribers and their clients. While we did not find this association when assessing language spoken at home, this association was borderline significant (potentially due to the small subgroup size with < 10% of our sample speaking languages other than English).
Lower utilisation of SGLT2i and GLP1-RA in remote areas has been reported in Australia when these medicines were first introduced in PBS [6]. Although we did not find utilisation decreased with increasing remoteness, similar to a recent study using 45 and Up data [5], the lowest utilisation rates were observed in people with T2D living in remote areas. We also had limited power to analyse this group, given the small proportion of people living in remote areas. However, we found people living in outer regional areas were more likely to receive GLP-1RA compared to people living in major cities even after adjusting for age, sex, number of comorbidities, and diabetes duration, with a marginal effect for SGLT2i. These findings may reflect an increasing dissemination of evidence of the benefits of these medicines among clinicians practising in those areas. It is well known that regional areas have lower availability of specialists, and general practitioners in outer regions may have an expanded scope of practice compared with those in major cities, possibly being more confident in prescribing add-on therapies for T2D management than their major city counterparts [12]. This result may also reflect a higher prevalence of specific cardiovascular conditions and risk factors that underpin the indication for prescription of these medicines in regional areas compared to major cities. It may also relate to outreach programs rolled out to increase the uptake of these medicines in these areas [37]—with a recent population-level study finding highly localised patterns of increased GLP-1RA use in regional areas [8].
Implications for clinical practice and policy
The generally low level of use of SGLTT2i and GLP-1RA suggests that addressing broader health system barriers may help improve uptake of these medicines[38]—such as prescriber education initiatives, as well as medicine subsidisation. In Australia, several reasons for suboptimal use of newer cardiometabolic medicines have been identified, such as lack of confidence in prescribing SGLT2i [12, 39], under-appreciation of their cardio-renal benefits [12, 39] and concerns with potential adverse effects and prescribing among people with comorbidities [12], which may be common concerns when prescribing amongst older populations. While costs of medicines have not been raised by clinicians as a barrier to prescribing [12], monthly therapy with GLP1-RA and SGLT2i is substantially costlier than older add-on options such as sulfonylureas to general PBS beneficiaries (AUD$30 vs. around $10 in July 2023) [40]. Utilisation of SGLT2i is likely to increase in more recent years, as the PBS glycaemic requirement for subsidised access for the treatment of T2D was lifted in December 2024 to facilitate access among people with high cardiovascular risk [41]. However, for GLP-1RA, there have been more reimbursement restrictions since March 2023, that only those contraindicate or intolerant to SGLT2i are eligible to reimburse GLP-1RA for treating T2D [42].
Limitations
Our study has several limitations. We used a highly selective cohort of people with T2D participating from the 45 and Up Study. This population is unlikely to be representative of the whole NSW T2D population in terms of age, health status and sociodemographics. While population-level extrapolation of utilisation rates should be performed with caution, internal relative risk estimates from this data have been found to be comparable with those from population health surveys [43]. We used self-reported information and dispensing data to identify the T2D population. While consistent with previous studies identifying people with T2D [4, 18], there is potential for misclassification due to recall bias and private dispensings not captured from PBS data. Similarly, there may be misclassification in other self-reported variables. We also lacked data on key indications and contraindications, such as HbA1c (a key biomarker of diabetes control and indicator for eligibility for SGLT2i and GLP1-RA via the PBS) and further potential factors influencing uptake (e.g. prescriber preferences). This means we could not assess appropriateness of prescribing and may be underestimating the prevalence of indicated use within our study population. Despite those limitations, the availability of survey data containing other key sociodemographic, clinical and behavioural characteristics linked to PBS data enabled a deeper understanding of a broader range of factors related to the utilisation of these critical medicines. During the study period, there were shortages of SGLT2i and GLP1-RA, although all shortages were classified as non-critical and possibly had a limited impact on our analysis [23]. Further research from different health systems and population groups is needed to confirm the generalisability of these findings and to unpack the role of specific associations where there may be common factors influencing medicine use (e.g. obesity and physical exercise; prescriber preferences).
In conclusion, the uptake of SGLT2i and GLP-1RA in a cohort of older and comorbid people living with T2D was suboptimal. Despite variation in the uptake of these medicine classes by sociodemographic, behavioural and clinical characteristics of people with T2D, further systemic interventions to promote prescribing within high-risk populations may be needed to increase utilisation of these highly efficacious medicines.
Data Availability
The data were provided by the Sax Institute. Access to the data by other individuals or authorities is not permitted without the express permission of the approving human research ethics committees and data custodians.
References
-
Kristensen SL, Rørth R, Jhund PS, Docherty KF, Sattar N, Preiss D, Køber L, Petrie MC, McMurray JJV (2019) Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet Diabetes Endocrinol 7(10):776–785. https://doi.org/10.1016/s2213-8587(19)30249-9
-
Nuffield Department of Population Health Renal Studies Group; SGLT2 inhibitor Meta-Analysis Cardio-Renal Trialists’ Consortium (2022) Impact of diabetes on the effects of sodium glucose co-transporter-2 inhibitors on kidney outcomes: collaborative meta-analysis of large placebo-controlled trials. Lancet (London, England) 400(10365):1788–1801. https://doi.org/10.1016/s0140-6736(22)02074-8
-
American Diabetes Association Professional Practice Committee (2024) 9 Pharmacologic approaches to glycemic treatment: standards of care in diabetes—2025. Diabetes care 48(1):181–206. https://doi.org/10.2337/dc25-S009
-
Greiver M, Havard A, Bowles JK, Kalia S, Chen T, Aliarzadeh B, Moineddin R, Sherlock J, Hinton W, Sullivan F, O’Neill B, Pow C, Bhatt A, Rahman F, Meza-Torres B, Litchfield M, de Lusignan S (2021) Trends in diabetes medication use in Australia, Canada, England, and Scotland: a repeated cross-sectional analysis in primary care. Bri J Gen Pract : J Royal College Gen Pract 71(704):e209–e218. https://doi.org/10.3399/bjgp20X714089
-
Lee CMY, Gibson AA, Humphries J, Nassar N, Colagiuri S (2023) Sex and age differences in the use of medications for diabetes and cardiovascular risk factors among 25,733 people with diabetes. PLoS ONE 18(10):e0287599. https://doi.org/10.1371/journal.pone.0287599
-
Morton JI, Ilomӓki J, Magliano DJ, Shaw JE (2021) The association of socioeconomic disadvantage and remoteness with receipt of type 2 diabetes medications in Australia: a nationwide registry study. Diabetologia 64(2):349–360. https://doi.org/10.1007/s00125-020-05304-3
-
Ofori-Asenso R, Liew D, Lalic S, Mazidi M, Magliano DJ, Ademi Z, Bell JS, Ilomaki J (2019) Adherence, persistence, and switching among people prescribed sodium glucose co-transporter 2 inhibitors: a nationwide retrospective cohort study. Adv Ther 36(11):3265–3278. https://doi.org/10.1007/s12325-019-01077-3
-
de Oliveira CJ, Lin J, Milder TY, Greenfield JR, Day RO, Stocker SL, Neuen BL, Havard A, Pearson SA, Falster MO (2024) Geographic variation in sodium-glucose cotransporter 2 inhibitor and glucagon-like peptide-1 receptor agonist use in people with type 2 diabetes in New South Wales. Diabetes Obes Metab, Australia. https://doi.org/10.1111/dom.15597
-
Mahtta D, Ramsey DJ, Lee MT, Chen L, Al Rifai M, Akeroyd JM, Vaughan EM, Matheny ME, Santo K, Navaneethan SD, Lavie CJ, Birnbaum Y, Ballantyne CM, Petersen LA, Virani SS (2022) Utilization rates of SGLT2 inhibitors and GLP-1 receptor agonists and their facility-level variation among patients with atherosclerotic cardiovascular disease and Type 2 diabetes: insights from the Department of Veterans Affairs. Diabetes Care 45(2):372–380. https://doi.org/10.2337/dc21-1815
-
Warren JR, Falster MO, Fox D, Jorm L (2013) Factors influencing adherence in long-term use of statins. Pharmacoepidemiol Drug Saf 22(12):1298–1307. https://doi.org/10.1002/pds.3526
-
Zhao Z, Zhao F, Zhang Y, Hu X, Li J, Tian C, Jin P, Liu D (2023) Risk factors of dapagliflozin-associated diabetic ketosis/ketoacidosis in patients with type 2 diabetes mellitus: a matched case-control study. Diabetes Res Clin Pract 196:110236. https://doi.org/10.1016/j.diabres.2023.110236
-
Milder TY, Stocker SL, Baysari M, Day RO, Greenfield JR (2021) Prescribing of SGLT2 inhibitors in primary care: a qualitative study of General Practitioners and Endocrinologists. Diabetes Res Clin Pract 180:109036. https://doi.org/10.1016/j.diabres.2021.109036
-
Bleicher K, Summerhayes R, Baynes S, Swarbrick M, Navin Cristina T, Luc H, Dawson G, Cowle A, Dolja-Gore X, McNamara M (2022) Cohort profile update: the 45 and Up Study. Int J Epidemiol 52(1):e92–e101. https://doi.org/10.1093/ije/dyac104
-
The Sax Institute’s 45 and Up Study. 5 Dec 2022. Cited 13 May 2025. Available from: www.saxinstitute.org.au/solutions/45-and-up-study/
-
Mellish L, Karanges EA, Litchfield MJ, Schaffer AL, Blanch B, Daniels BJ, Segrave A, Pearson SA (2015) The Australian Pharmaceutical Benefits Scheme data collection: a practical guide for researchers. BMC Res Notes 8:634. https://doi.org/10.1186/s13104-015-1616-8
-
NPS MedicineWise (2015) SGLT2 inhibitor listings: indications and combinations. Accessed 1 Jul 2022. Available from: https://www.nps.org.au/radar/articles/sglt2-inhibitor-listings-indications-and-combinations
-
NPS MedicineWise (2010) Exenatide (Byetta) for type 2 diabetes mellitus. Accessed 1 Jul 2022. Available from: https://www.nps.org.au/radar/articles/exenatide-byetta-for-type-2-diabetes-mellitus
-
Comino EJ, Tran DT, Haas M, Flack J, Jalaludin B, Jorm L, Harris MF (2013) Validating self-report of diabetes use by participants in the 45 and Up Study: a record linkage study. BMC Health Serv Res 13:481. https://doi.org/10.1186/1472-6963-13-481
-
Sax Institute (2021) The 45 and Up Study Wave 3 Data Book Second Follow-Up, 2018–2020. Cited 13 May 2025. Available from: https://www.saxinstitute.org.au/wp-content/uploads/W3-databook-May2021.pdf
-
Australian Insitute of Health and Welfare (2023) The Active Australia Survey: a guide and manual for implementation, analysis and reporting. Accessed 1 Jul 2022. Available from: https://www.aihw.gov.au/reports/physical-activity/active-australia-survey/summary
-
Pratt NL, Kerr M, Barratt JD, Kemp-Casey A, KalischEllett LM, Ramsay E, Roughead EE (2018) The validity of the Rx-Risk Comorbidity Index using medicines mapped to the Anatomical Therapeutic Chemical (ATC) Classification System. BMJ Open 8(4):e021122. https://doi.org/10.1136/bmjopen-2017-021122
-
Liu Z, Moorin R, Worthington J, Tofler G, Bartlett M, Khan R, Zuo Y (2016) Using large-scale linkage data to evaluate the effectiveness of a National Educational Program on antithrombotic prescribing and associated stroke prevention in primary care. J Am Heart Assoc 5(10) https://doi.org/10.1161/jaha.116.003729
-
Lin J, Pearson S-A, Greenfield JR, Park KH, Havard A, Brieger D, Day RO, Falster MO, de Oliveira CJ (2023) Trends in use of sodium-glucose co-transporter 2 inhibitors (SGLT2i) and glucagon-like peptide-1 receptor agonists (GLP-1RA) in Australia in the era of increased evidence of their cardiovascular benefits (2014–2022). Eur J Clin Pharmacol 79(9):1239–1248. https://doi.org/10.1007/s00228-023-03539-8
-
Morton JI, Ilomӓki J, Magliano DJ, Shaw JE (2022) Persistent disparities in diabetes medication receipt by socio-economic disadvantage in Australia. Diabet Med : J Bri Diabet Assoc 39(9):e14898. https://doi.org/10.1111/dme.14898
-
Arnold SV, Tang F, Cooper A, Chen H, Gomes MB, Rathmann W, Shimomura I, Vora J, Watada H, Khunti K, Kosiborod M (2022) Global use of SGLT2 inhibitors and GLP-1 receptor agonists in type 2 diabetes. Result Dis BMC Endocrine Disord 22(1):111. https://doi.org/10.1186/s12902-022-01026-2
-
Khera R, Dhingra LS, Aminorroaya A, Li K, Zhou JJ, Arshad F, Blacketer C, Bowring MG, Bu F, Cook M, Dorr DA, Duarte-Salles T, DuVall SL, Falconer T, French TE, Hanchrow EE, Horban S, Lau WC, Li J, Liu Y, Lu Y, Man KK, Matheny ME, Mathioudakis N, McLemore MF, Minty E, Morales DR, Nagy P, Nishimura A, Ostropolets A, Pistillo A, Posada JD, Pratt N, Reyes C, Ross JS, Seager S, Shah N, Simon K, Wan EY, Yang J, Yin C, You SC, Schuemie MJ, Ryan PB, Hripcsak G, Krumholz H, Suchard MA (2023) Multinational patterns of second line antihyperglycaemic drug initiation across cardiovascular risk groups: federated pharmacoepidemiological evaluation in LEGEND-T2DM. BMJ Medicine 2(1):e000651. https://doi.org/10.1136/bmjmed-2023-000651
-
Lim CE, Pasternak B, Eliasson B, Danaei G, Ueda P (2023) Use of sodium-glucose co-transporter 2 inhibitors and glucagon-like peptide-1 receptor agonists according to the 2019 ESC guidelines and the 2019 ADA/EASD consensus report in a national population of patients with type 2 diabetes. Eur J Prev Cardiol 30(8):634–643. https://doi.org/10.1093/eurjpc/zwac315
-
Australian Diabetes Society (2020) Australian type 2 diabetes glycaemic management algorithm 2020. Cited 13 May 2025. Available from: https://www.diabetessociety.com.au/20200908%20T2D%20Management%20Algorithm%2003092020.pdf
-
Australian National Diabetes Audit Annual Report 2022. (2023) Monash University, School of Public Health and Preventive Medicine. Report No 15, 91 pages
-
Hamblin PS, Wong R, Ekinci EI, Fourlanos S, Shah S, Jones AR, Hare MJL, Calder GL, Epa DS, George EM, Giri R, Kotowicz MA, Kyi M, Lafontaine N, MacIsaac RJ, Nolan BJ, O’Neal DN, Renouf D, Varadarajan S, Wong J, Xu S, Bach LA (2019) SGLT2 inhibitors increase the risk of diabetic ketoacidosis developing in the community and during hospital admission. J Clin Endocrinol Metab 104(8):3077–3087. https://doi.org/10.1210/jc.2019-00139
-
Lähteenvuo M, Tiihonen J, Solismaa A, Tanskanen A, Mittendorfer-Rutz E, Taipale H (2025) Repurposing semaglutide and liraglutide for alcohol use disorder. JAMA Psychiat 82(1):94–98. https://doi.org/10.1001/jamapsychiatry.2024.3599
-
ElSayed NA, Aleppo G, Aroda VR, Bannuru RR, Brown FM, Bruemmer D, Collins BS, Hilliard ME, Isaacs D, Johnson EL, Kahan S, Khunti K, Leon J, Lyons SK, Perry ML, Prahalad P, Pratley RE, Seley JJ, Stanton RC, Gabbay RA, Association obotAD, (2022) 9 Pharmacologic approaches to glycemic treatment: standards of care in diabetes—2023. Diabetes Care 46(1):140–157. https://doi.org/10.2337/dc23-S009
-
Khan MS, Fonarow GC, McGuire DK, Hernandez AF, Vaduganathan M, Rosenstock J, Handelsman Y, Verma S, Anker SD, McMurray JJV, Kosiborod MN, Butler J (2020) Glucagon-like peptide 1 receptor agonists and heart failure: the need for further evidence generation and practice guidelines optimization. Circulation 142(12):1205–1218. https://doi.org/10.1161/circulationaha.120.045888
-
Johansson KS, Bülow C, Jimenez-Solem E, Petersen TS, Christensen MB (2023) Age disparities in glucose-lowering treatment for Danish people with type 2 diabetes: a cross-sectional study between 2019 and 2020. Lancet Healthy Longevity 4(12):e685–e692. https://doi.org/10.1016/S2666-7568(23)00210-6
-
Milder TY, Lin J, Pearson S-A, Greenfield JR, Day RO, Stocker SL, Neuen BL, Falster MO, de Oliveira CJ (2025) Use of, time to, and type of first add-on anti-hyperglycaemic therapy to metformin in Australia, 2018–2022. Br J Clin Pharmacol 91(1):117–126. https://doi.org/10.1111/bcp.16231
-
Patsan I, Tapley A, Davoren P, Fielding A, Holliday E, Ball J, Davey A, van Driel M, Turner R, Mulquiney K, Spike N, FitzGerald K, Magin P (2023) Temporal trends in, and associations of, early-career general practitioner prescriptions of second-line Type 2 Diabetes medications, 2010–2018. PLoS ONE 18(1):e0280668. https://doi.org/10.1371/journal.pone.0280668
-
Acharya S, Philcox AN, Parsons M, Suthers B, Luu J, Lynch M, Jones M, Attia J (2019) Hunter and New England Diabetes Alliance: innovative and integrated diabetes care delivery in general practice. J Australian J Prim Health. 25(3):219–243. https://doi.org/10.1071/PY18179
-
Ndumele CE, Rangaswami J, Chow SL, Neeland IJ, Tuttle KR, Khan SS, Coresh J, Mathew RO, Baker-Smith CM, Carnethon MR, Despres JP, Ho JE, Joseph JJ, Kernan WN, Khera A, Kosiborod MN, Lekavich CL, Lewis EF, Lo KB, Ozkan B, Palaniappan LP, Patel SS, Pencina MJ, Powell-Wiley TM, Sperling LS, Virani SS, Wright JT, Rajgopal Singh R, Elkind MSV (2023) Cardiovascular-kidney-metabolic health: a presidential advisory from the American Heart Association. Circulation 148(20):1606–1635. https://doi.org/10.1161/cir.0000000000001184
-
Milder TY, Stocker SL, Baysari MT, Day RO, Greenfield JR (2024) ‘We are somehow fixated on this being a diabetes drug’: a qualitative study exploring the views of cardiologists and nephrologists about sodium-glucose cotransporter 2 inhibitor initiation. Intern Med J 54(4):559–567. https://doi.org/10.1111/imj.16203
-
Department of Health and Aged Care (2023) Pharmaceutical benefits: fees, patient contributions and safety net thresholds. Cited 13 Nov 2023. Available from: https://www.pbs.gov.au/info/healthpro/explanatory-notes/front/fee
-
Department of Health and Aged Care (2024) Public Summary Document – July 2024 PBAC Meeting. Cited 21 Jan 2025. Available from: https://m.pbs.gov.au/reviews/pbs-restriction-changes-to-type-2-diabetes-mellitus/SGLT2-inhibitors-for-T2DM-PBAC-PSD-07-2024.PDF
-
Department of Health and Aged Care (2023) March 2023 PBAC Meeting. Cited 11 Nov 2023. Available from: https://www.pbs.gov.au/info/industry/listing/elements/pbac-meetings/agenda/march-2023-pbac-meeting
-
Mealing NM, Banks E, Jorm LR, Steel DG, Clements MS, Rogers KD (2010) Investigation of relative risk estimates from studies of the same population with contrasting response rates and designs. BMC Med Res Methodol 10:26. https://doi.org/10.1186/1471-2288-10-26
Acknowledgements
This research was completed using data collected through the 45 and Up Study (www.saxinstitute.org.au). The 45 and Up Study is managed by the Sax Institute in collaboration with major partner Cancer Council NSW and partners the Heart Foundation and the NSW Ministry of Health. We thank the many thousands of people participating in the 45 and Up Study. Medicare claims and PBS data were provided by Services Australia. Linkage of the 45 and Up Study cohort data to these datasets is facilitated by the Sax Institute using a unique identifier and deterministic matching. Secure data access was provided through the Sax Institute’s Secure Unified Research Environment (SURE).
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. This research is supported by the Heart Foundation 2022 45 & Up Grant (ID: 107048), the National Health and Medical Research Council (NHMRC) Ideas Grants (ID: 2002889; ID: 1183273) and NHMRC Centre of Research Excellence in Medicines Intelligence (ID: 1196900). BLN is supported by an NHMRC Emerging Leader Investigator Grant (ID: 2026621) and a Ramaciotti Foundation Health Investment Grant (ID: 2023HIG69). AAG is supported by an NHMRC Emerging Leader Investigator Grant (ID: APP1173784). MOF is supported by a Heart Foundation Future Leader Fellowship (ID: 105609) and the NHMRC Centre of Research Excellence in Medicines Intelligence (ID: 1196900).
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (DOCX 193 KB)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Reprints and permissions
About this article
Cite this article
de Oliveira Costa, J., Lin, J., Milder, T.Y. et al. Variation in uptake of sodium glucose cotransporter 2 inhibitors and glucagon-like peptide-1 receptor analogues in adults with type 2 diabetes at high cardiovascular risk. Eur J Clin Pharmacol (2025). https://doi.org/10.1007/s00228-025-03870-2
- Received
- Accepted
- Published
- DOI https://doi.org/10.1007/s00228-025-03870-2
Keywords
- Pharmacoepidemiology
- Type 2 diabetes
- Sodium -glucose cotransporter 2 inhibitor
- Glucagon-like peptide-1 receptor agonists
